Abstract
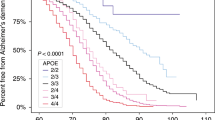
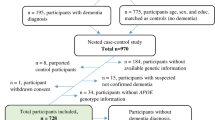
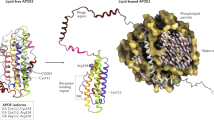
Genetic studies on Alzheimer's disease (AD), a devastating neurodegenerative disorder, have identified the apolipoprotein E (APOE) gene as a strong susceptibility marker for AD1–2. TheE*4 allele of APOE is a major risk factor for AD regardless of age of onset or family history3. However, the observation that the APOE*4 allele is neither necessary nor sufficient for the expression of AD emphasizes the involvement of other environmental or genetic elements that, either in conjunction with APOE*4 or alone, increase an individual's risk of developing AD. Among the candidate genes that may affect the risk of this multifactorial disease is the gene coding for α1 -antichymotrypsin (ACT). Like APOE protein, ACT binds to β-amyloid peptide (ApP) with high affinity in the filamentous deposits found in the AD brain4–10 and serves as a strong stimulatory factor in the polymerization of AβP into amyloid filaments10. In AD brains, ACT expression is enhanced, particularly in areas that develop amyloid plaques4–8, 11–12, suggesting that ACT may play an important role in the pathogenesis of AD. Here we show that a common polymorphism in the signal peptide of ACT13 confers a significant risk for AD. Furthermore, the APOE*4 gene dosage effect associated with AD risk is significantly modified by the ACT polymorphism. We have also identified a unique combination of the ACT/AA and APOE 4/4 genotypes as a potential susceptibility marker for AD, as its frequency was 1/17 in the AD group compared to 1/313 in the general population control. Our data show that ACT behaves as a modifier gene that alters the AD risk conventionally associated with the APOE*4 allele.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Strittmatter, W.J. et al. Apolipoprotein E: high avidity binding to β-amyloid and increased frequency of type 4 allele in late-onset familial Alzheirner disease. Proc. natn. Acad. Sci. U.S.A. 90, 1977–1981 (1993).
Saunders, A.M. et al. Association of apolipoprotein E allele ε4 with late-onset familial and sporadic Alzheimer's disease. Neurology 43, 1467–1472 (1993).
Kamboh, M.I. Apolipoprotein E polymorphism and susceptibility to Alzheimer's disease. Hum. Biol. 67, 195–215 (1995).
Abraham, C.R., Selkoe, D.J. & Potter, H. Immunochemical identification of the serine protease inhibitor α1-antichymotrypsin in the brain amyloid deposits of Alzheimer's disease. Cell 52, 487–501 (1988).
Abraham, C.R., Shirahama, T. & Potter, H. α1-antichymotrypsin is associated solely with amyloid deposits containing the β-protein: amyloid and cell localization of α1-antichymotrypsin. Neurobiol. Aging 11, 123–129 (1990).
Picken, M.M. et al. Distribution of the protease inhibitor α1 -antichymotrypsin in cerebral and systemic amyloid. J. Neuropathol. exp. Neurol. 49, 41–48 (1990).
Rozemuller, J.M. et al. Distribution pattern and functional state of α1-antichymotrypsin in plaques and vascular amyloid in Alzheimer's disease. Acta Neuropath. 82, 200–207 (1991).
Shoji, M. et al. Alpha 1-antichymotrypsin is present in diffuse senile plaques: a comparative study of β-protein and α1-antichymotrypsin immunostaining in the Alzheimer brain. Am. J. Path. 138, 247–257. (1991).
Fraser, P.E., Nguyen, J.T., McLachlan, D.R., Abraham, C.R. & Kirschner, D.A. α1-antichymotrypsin binding to Alzheimer Aβ peptides is sequence specific and induces fibril disaggregation in vitro. J. Neurochem. 61, 298–305 (1993).
Ma, J., Yee, A., Brewer, H.B. Jr., Das, S. & Potter, H. Amyloid-associated proteins α1-antichymotrypsin and apolipoprotein E promote assembly of Alzheimer β-protein into filaments. Nature 372, 92–94 (1994).
Ishiguro, K. et al. Differential expression of α1 -antichymotrypsin in the aged human brain. Virchows Arch. B. Cell Path. 64, 221–227 (1993).
Wisniewski, K.E. & Kida, E. Proteinase inhibitor α 1-antichymotrypsin has different expression in various forms of neuronal ceroid lipfuscinosis. Exp. Neurol. 110, 121–126 (1990).
Faber, J.-P. et al. The molecular basis of α1 -antichymotrypsin deficiency in a heterozygote with liver and lung disease. J. Hepatol. 18, 313–321 (1993).
Strittmatter, W.J. et al. Hypothesis — microtubule instability and paired helical filament formation in the Alzheimer disease brain are related to apolipoprotein E genotype. Expl. Neurol. 125, 163–171 (1994).
Randall, L.L. & Hardy, S.J.S. Unity in function in the absence of consequence in sequence: role of leader peptides in export. Science 243, 1156–1159 (1989).
Matsubara, E. et al. Alpha 1-antichymotrypsin as a possible biochemical marker for Alzheimer-type dementia. Ann. Neurol. 28, 561–567 (1990).
Rayford, A., Rao, J.S. & Festoff, B.W. Characterization of the serpin, β1 -antichymotrypsin, in normal human cerebrospinal fluid. J. Neurochem. 58, 88–94 (1992).
Wisniewski, T., Castano, E.M., Golabek, A., Vogel, T. & Frangione, B. Acceleration of Alzheimer's fibril formation by apolipoprotein E in vitro. Am. J. Pathol. 145, 1030–1035 (1994).
Sanan, D.A. et al. Apolipoprotein E associates with Aβ amyloid peptide to form novel monofibrils: Isoform apoE4 associates more efficiently than apoES. J. Clin Invest. 94, 880–889 (1994).
DeKosky, S.T. et al. Alzheimer's disease and related dementias: report of follow-up on the first 1,000 cases from Pittsburgh. Neurology 44, A354–355 (1994).
McKhann, G. et al. Clinical diagnosis of Alzheimer's disease. Neurology 34, 939–944 (1984).
Diagnostic and Statistical Manual of Mental Disorders: DSM-III-R. (3rd revised edn) (Washington, D.C. American Psychiatric Association, (1987).
Khachaturian, Z. Diagnosis of Alzheimer's disease. Arch. Neurol. 42, 1097–1105 (1985).
Mirra, S.S. et al. The consortium to establish a registry for Alzheimer's disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer's disease. Neurology 41, 479–486 (1991).
Kamboh, M.I., Aston, C.E. & Hamman, R.F. The relationship of APOE polymorphism and cholesterol levels in normoglycemic and diabetic subjects in a biethnic population from the San Luis Valley, Colorado. Atherosclerosis 112, 145–159 (1995).
Eichner, J.E., Kuller, L.H., Ferrell, R.E., Meilahn, E.N. & Kamboh, M.I. Phenotypic effects of apolipoproteins structural variation on lipid profiles. III. The contribution of APOE to prediction of total cholesterol, apoB and LDL-C in the Healthy Women Study. Arteriosclerosis 10, 379–385 (1990).
Rights and permissions
About this article
Cite this article
Kamboh, M., Sanghera, D., Ferrell, R. et al. A4POE*4-associated Alzheimer's disease risk is modified by α1–antichymotrypsin polymorphism. Nat Genet 10, 486–488 (1995). https://doi.org/10.1038/ng0895-486
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0895-486
This article is cited by
-
Serpin Signatures in Prion and Alzheimer’s Diseases
Molecular Neurobiology (2022)
-
Polygenic resilience scores capture protective genetic effects for Alzheimer’s disease
Translational Psychiatry (2022)
-
Polymorphism of the APOE Gene and Markers of Brain Damage in the Outcomes of Severe Traumatic Brain Injury in Children
Neuroscience and Behavioral Physiology (2021)
-
Human and mouse single-nucleus transcriptomics reveal TREM2-dependent and TREM2-independent cellular responses in Alzheimer’s disease
Nature Medicine (2020)
-
Association between α1-antichymotrypsin signal peptide −15A/T polymorphism and the risk of Alzheimer’s disease: a meta-analysis
Molecular Biology Reports (2012)