Abstract
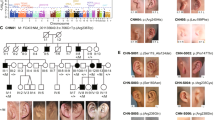
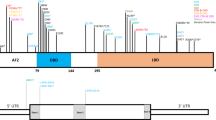
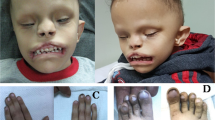
Autosomal recessive Meckel syndrome (OMIM 249000) (MES), first described in 1822 by Johann F. Meckel1, is a major monogenic malformation syndrome with a neural tube defect leading to death of the fetus in utero or shortly after birth. The hallmarks of the syndrome are occipital meningo-encephalocele, very large kidneys with multicystic dysplasia, cystic and fibrotic changes of the liver and polydactyly2,3 (Fig. 1). Other typical malformations for MES are cleft lip and palate, urinary tract anomalies, ambiguous genitals in the males and club feet. Although MES has been reported worldwide, reports on the true birth prevalence of MES in different populations are scarce. In Finland MES is effectively screened and relatively frequent with a birth prevalence of 1:9,000 and a disease gene frequency of 0.01 (ref. 4) which is of the same order of magnitude as that of the most common recessive diseases belonging to the ‘Finnish disease heritage’, that is genetic disorders enriched or only encountered in Finland. However, in MES, comparable or even higher incidences are also reported from other populations4–6. Here, we report the assignment of the MES locus to chromosome 17q21–q24 in the 13 cM region, and exclude some of the potential candidate genes located in this critical chromosomal region.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Meckel, J.F. Beschreibung zweier, durch sehr ähnliche Bildungsabweichungen entstellter Geschwister. Dtsch. Arch. Physiol. 7, 99–172 (1822).
Mecke, S. & Passarge, E. Encephalocele, polycystic kidneys, and polydactyly as an autosomal recessive trait simulating certain other disorders: the Meckel syndrome. Ann. Génét. 14, 97–103 (1971).
Salonen, R. The Meckel syndrome: clinicopathological findings in 67 patients. Am. J. med. Genet. 18, 671–689 (1984).
Salonen, R. & Norio, R. The Meckel's syndrome in Finland; epidemiologic and genetic aspects. Am. J. med. Genet. 18, 691–698 (1984).
Young, I.D., Rickett, A.B. & Clarke, M. High incidence of Meckel's syndrome in Gujarati Indians. J. med. Genet. 22, 301–304 (1985).
Teebi, A.S., Al-Saleh, Q.A. & Odeh, H. Meckel syndrome and neural tube defects in Kuwait. J. med. Genet. 29, 140 (1992).
Polymeropoulos, M.H., Rath, D.S., Xiao, H. & Merril, C.R. A simple sequence repeat polymorphism at the human growth hormone locus. Nucl. Acids Res. 19, 689 (1991).
Ott, J. Analysis of Human Genetic Linkage rev. edn. 203–216 (Johns Hopkins Univ. Press, Baltimore, 1991).
Savukoski, M. et al. Defined chromosomal assignment of CLN5 demonstrates that at least four genetic loci are involved in the pathogenesis of human ceroid lipofuscinoses. Am. J. hum. Genet. 55, 695–701 (1994).
Schleutker, J. et al. Linkage disequilibrium utilized to establish a refined genetic position of the Salla disease locus on 6q14–q15. Genomics 27, 286–292 (1995).
Phillips, J.A. III & Cogan, J.D. Genetic basis of endocrine disease 6: molecular basis of familial human growth hormone deficiency. J. Clin. Endocr. Metab. 78, 11–16 (1994).
Gilbert, S.F. Developmental Biology, 3rd edn. (Sinauer Associates, Inc., Sunderland, Massachusetts, 1991).
Kaur, S. et al. Dominant mutation of the murine Hox-2.2 gene results in developmental abnormalities. J. exp. Zool. 264, 323–336 (1992).
Charité, J., de Graaff, W., Shen, S. & Deschamps, J. Ectopic expression of Hoxb-8 causes duplication of the ZPA in the forelimb and homeotic transformation of axial structures. Cell 78, 589–601 (1994).
Munke, M., Cox, D.R., Jackson, I.J., Hogan, B.L.M. & Francke, U. The murine Hox-2 cluster of homeo box containing genes maps distal on chromosome 11 near the tail-short (Ts) locus. Cytogenet. Cell Genet. 42, 236–240 (1986).
Winter, R.M. Malformation syndromes: a review of mouse/human homology. J. med. Genet. 25, 480–487 (1988).
Deinard, A.S., Ruano, G. & Kidd, K.K. A dinucleotide repeat polymorphism at the HOX2B locus. Nucl. Acids Res. 20, 1171 (1992).
Wilhelmsen, K.C., Lynch, T., Pavlou, E., Higgins, M. & Nygaard, T.G. Localization of disinhibition-dementia-parkinsonism-amyotrophy complex to 17q21–22. Am. J. hum. Genet. 55, 1159–1165 (1994).
Anderson, L.A. et al. High-density genetic map of the BRCA1 region of chromosome 17q12–q21. Genomics 17, 618–623 (1993).
Haines, J.L. et al. A genetic linkage map of chromosome 17. Genomics 8, 1–6 (1990).
Bentley, K.L., Ferguson-Smith, A.C., Miki, T., Kidd, K.K. & Ruddle, F.H. Physical linkage of Hox2.1 and nerve growth factor receptor. Cytogenet. Cell Genet. 51, A961 (1989).
Stoffel, M. & Bell, G.I. Microsatellite polymorphism in the human platelet glycoprotein Ilia gene (GP3A) on chromosome 17. Nucl. Acids Res. 20, 1172 (1992).
Polymeropoulos, M.H., Xiao, H., Rath, D.S. & Merril, C.R. Dinucleotide repeat polymorphism at the human gene of the light and heavy chains of myeloperoxidase glycoprotein (MPO). Nucl. Acids Res. 19, 1961 (1991).
Patau, K. et al. Multiple congenital anomaly caused by an extra chromosome. Lancet 1, 790 (1960).
Aaltonen, J. et al. Autoimmune polyglandular disease type I: exclusion map using amplifiable multiallelic markers in a microtiter well format. Eur. J. hum. Genet. 1, 164–171 (1993).
Lathrop, G.M., Lalouel, J.-M., Julier, C. & Ott, J. Strategies for multilocus linkage analysis in humans. Proc. natn. Acad. Sci. U.S.A. 81, 3443–3446 (1984).
Terwilliger, J.D. A powerful likelihood method for the analysis of linkage disequilibrium between trait loci and one or more polymorphic marker loci. Am. J. hum. Genet. 56, 777–787 (1995).
Wright, C., Healicon, R., English, C. & Burn, J. Meckel syndrome: what are the minimum diagnostic criteria?. J. med. Genet. 31, 482–485 (1994).
Guapay, G. et al. The 1993–94 Généthon human genetic linkage map. Nature Genet. 7, 246–339 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Paavola, P., Salonen, R., Weissenbach, J. et al. The locus for Meckel syndrome with multiple congenital anomalies maps to chromosome 17q21–q24. Nat Genet 11, 213–215 (1995). https://doi.org/10.1038/ng1095-213
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1095-213
This article is cited by
-
Molecular Genetics and Pathogenic Mechanisms for the Severe Ciliopathies: Insights into Neurodevelopment and Pathogenesis of Neural Tube Defects
Molecular Neurobiology (2011)
-
Molecular diagnostics of Meckel–Gruber syndrome highlights phenotypic differences between MKS1 and MKS3
Human Genetics (2007)
-
MKS1, encoding a component of the flagellar apparatus basal body proteome, is mutated in Meckel syndrome
Nature Genetics (2006)
-
The transmembrane protein meckelin (MKS3) is mutated in Meckel-Gruber syndrome and the wpk rat
Nature Genetics (2006)