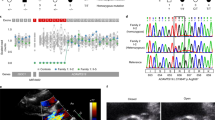
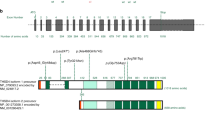
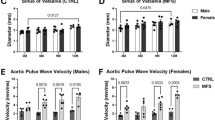
Abstract
Aortic aneurysm and dissection account for about 2% of all deaths in industrialized countries; they are also components of several genetic diseases, including Marfan syndrome (MFS)1. The vascular phenotype of MFS results from mutations in fibrillin-1 (FBN1), the major constituent of extracellular microfibrils2,3. Microfibrils, either associated with or devoid of elastin, give rise to a variety of extracellular networks in elastic and non-elastic tissues3. It is believed that microfibrils regulate elastic fibre formation by guiding tropo-elastin deposition during embryogenesis and early post-natal life4. Hence, vascular disease in MFS is thought to result when FBN1 mutations preclude elastic fibre maturation by disrupting microfibrillar assembly. Here we report a gene-targetting experiment in mice that indicates that fibrillin-1 microfibrils are predominantly engaged in tissue homeostasis rather than elastic matrix assembly. This finding, in turn, suggests that aortic dilation is due primarily to the failure by the microfibrillar array of the adventitia to sustain physiological haemodynamic stress, and that disruption of the elastic network of the media is a secondary event.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dietz, H., Ramirez, F. & Sakai, L. Marfan syndrome and other microfibrillar diseases. in Advances in Human Genetics, vol. 22 (eds Harris, H. & Hirschhorn, K.) 153–186 (Plenum, New York, (1994).
Dietz, H. & Pyeritz, R. Mutations in the human gene for fibrillin-1 (FBN1) in the Marfan syndrome and related disorders. Hum. Mol. Genet. 4, 1799–1809 (1995).
Sakai, L., Keene, D.R. & Engvall, E. Fibrillin, a new 350-kD glycoprotein, is a component of extracellular microfibrils. J. Cell Biol. 103, 2499–2509 (1986).
Mecham, R.P. & Davis, E. Elastic fiber structure and assembly, in Extracellular Matrix Assembly and Structure (eds Yurchenco, P. O., Birk, D.E. & Mecham, R.P.) 281–314 (Academic Press, New York, (1994).
Pereira, L. et al. Genomic organization of the sequence coding for fibrillin, the defective gene product in Marfan syndrome. Hum. Mol. Genet. 2, 961–968 (1993).
Kainulainen, K., Karttunen, L., Puhakka, L., Sakai, L. & Peltonen, L. Mutations in the fibrillin gene responsible for dominant ectopia lentis and neonatal Marfan syndrome. Nature Genet. 6, 64–69 (1994).
Ramirez, F. Fibrillin mutations in Marfan syndrome and related phenotypes. Curr. Opin. Genet. Dev. 6, 309–315 (1996).
Hogan, B., Beddington, R., Constantini, F. & Lacy, E. Manipulating the Mouse Embryo: A Laboratory Manual, 2nd ed. (Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, 1994).
Zhang, H. et al. Structure and expression of fibrillin-2, a novel microfibrillar component preferentially located in elastic matrices. J. Cell Biol. 124, 855–863 (1994).
Fiering, S. et al. Targeted deletion of 5′ HS2 of the murine b-globin LCR reveals that it is not essential for proper regulation of the b-globin locus. Genes Dev. 9, 2203–2213 (1995).
Pham, C.T.N., Maclvor, D.M., Hug, B.A., Heusel, J.W. & Ley, T.J. Long-range disruption of gene expression by a selectable marker cassette. Proc. Natl. Acad. Sci. USA 93, 13090–13095 (1996).
Zhang, H., Hu, W. & Ramirez, F. Developmental expression of fibrillin genes suggests heterogeneity of extracellular microfibrils. J. Cell Biol. 123, 1165–1176 (1995).
Davis, E.C. Smooth muscle cell to elastic lamina connections in developing mouse aorta: role in aortic medial organization. Lab. Invest. 68, 89–97 (1993).
Hollister, D.W., Godfrey, M., Sakai, L.Y. & Pyeritz, R.E. Marfan syndrome: immunohistologic abnormalities of the microfibrillar fiber system. N. Engl. J. Med. 323, 935–939 (1990).
Tilson, D.M., Elefteriades, J. & Brophy, C.M. Tensile strength and collagen in abdominal aortic aneurysm disease, in The Cause and Management of Aneurysms (eds Greenhalgh, M. & Mannick, J.A.) 97–104 (Latimer Trend, Plymouth UK, (1990).
Davis, E.C. Immunolocalization of microfibril and microfibril-associated proteins in the subendothelial matrix of the developing mouse aorta. J. Cell Sci. 107, 727–736 (1994).
Putnam, E.A., Zhang, H., Ramirez, F. & Milewicz, D.M. Fibrillin-2 (FBN2) mutations result in the Marfan-like disorder, congenital contractural arachnodactyly. Nature Genet. 11, 456–458 (1995).
Ewart, A.K., Jin, W., Atkinson, D.L., Morris, C.A. & Keating, M.T. Supravalvular aortic stenosis associated with a deletion disrupting the elastin gene. J. Clin. Invest. 93, 1071–1077 (1994).
Li, E., Bestor, T.H. & Jaenisch, R. Targeted mutation of the DNA methyltransferase gene results in embryonic lethality. Cell 63, 915–926 (1992).
Andrikopoulos, K., Liu, X., Keene, R.D., Jaenisch, R. & Ramirez, F. Targeted mutation in the col5a2 gene reveals a regulatory role for type V collagen during matrix assembly. Nature Genet. 9, 31–36 (1995).
Sambrook, E., Fritsch, E.F. & Maniatis, T., Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, 1989).
Gause, W.C. & Adamovicz, J. Use of PCR to quantitate relative differences in gene expression, in PCR Primer: A Laboratory Manual (eds Dieffenbach, C. W. & Dveksler, G.S.) 293–338 (Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, (1989).
Reinhardt, D.P. et al. Fibrillin 1: organization in microfibrils and structural properties. J. Mol. Biol. 258, 104–115 (1996).
Keene, D.R. et al. Fibrillin-1 in human cartilage: developmental expression and formation of special banded fibrils. J. Histochem. Cytochem. (in the press).
Hurle, J.M. et al. Elastin exhibits a distinctive temporal and spatial pattern of distribution in the developing chick limb in association with the establishment of the cartilaginous skeleton J. Cell Sci. 107, 2623–2634 (1994).
Sakai, L.Y., Keene, D.R., Glanville, R.A. & Bächinger, H.P. Purification and partial characterization of fibrillin, a cystine-rich structural component of connective tissue microfibrils. J. Biol. Chem. 266, 14763–14770 (1991).
Sakai, L.Y. & Keene, D.R. Fibrillin: monomers and microfibrils. Methods Enzymol. 245, 29–52 (1994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pereira, L., Andrikopoulos, K., Tian, J. et al. Targetting of the gene encoding fibrillin–1 recapitulates the vascular aspect of Marfan syndrome. Nat Genet 17, 218–222 (1997). https://doi.org/10.1038/ng1097-218
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1097-218
This article is cited by
-
The mechanism and therapy of aortic aneurysms
Signal Transduction and Targeted Therapy (2023)
-
Genetics and mechanisms of thoracic aortic disease
Nature Reviews Cardiology (2023)
-
Fibroblast-Secreted Phosphoprotein 1 Mediates Extracellular Matrix Deposition and Inhibits Smooth Muscle Cell Contractility in Marfan Syndrome Aortic Aneurysm
Journal of Cardiovascular Translational Research (2022)
-
Marfan syndrome
Nature Reviews Disease Primers (2021)
-
Effects of Simulated Microgravity on Wild Type and Marfan hiPSCs-Derived Embryoid Bodies
Cellular and Molecular Bioengineering (2021)