Abstract
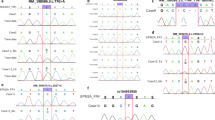
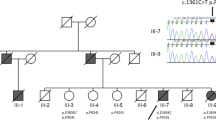
Lafora's disease (LD; OMIM 254780) is an autosomal recessive form of progressive myoclonus epilepsy characterized by seizures and cumulative neurological deterioration. Onset occurs during late childhood and usually results in death within ten years of the first symptoms1,2. With few exceptions, patients follow a homogeneous clinical course despite the existence of genetic heterogeneity3. Biopsy of various tissues, including brain, revealed characteristic polyglucosan inclusions called Lafora bodies4–8, which suggested LD might be a generalized storage disease6,9. Using a positional cloning approach, we have identified at chromosome 6q24 a novel gene, EPM2A, that encodes a protein with consensus amino acid sequence indicative of a protein tyrosine phosphatase (PTP). mRNA transcripts representing alternatively spliced forms of EPM2A were found in every tissue examined, including brain. Six distinct DNA sequence variations in EPM2A in nine families, and one homozygous microdeletion in another family, have been found to cosegregate with LD. These mutations are predicted to cause deleterious effects in the putative protein product, named laforin, resulting in LD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Berkovic, S.F., Andermann, F., Carpenter, S. & Wolfe, L.S. Progressive myoclonus epilepsies: specific causes and diagnosis. New Eng. J. Med. 315, 296–305 (1986).
Van Heycop Ten Ham, M.W. Lafora disease, a form of progressive myoclonus epilepsy. Handbook of Clinical Neurology 15, 382–422 (1974).
Minassian, B.A., Sainz, J., Bohlega, S., Sakamoto, L.M. & Delgado-Escueta, A.V. Genetic heterogeneity in Lafora's disease . Epilepsia 37, 126 ( 1996).
Lafora, G.R. Uber das vorkommen amyloider korperchen im innern der ganglienzellen; zugleich ein beitrag zum studium der amyloiden substanz im nervensystem. Virchows. Arch. Path. Anat. 205, 295– 303 (1911).
Harriman, D.G. & Millar, J.H.D. Progressive familial myoclonic epilepsy in 3 families: its clinical features and pathological basis. Brain 78, 325–349 ( 1955).
Schwarz, G.A. & Yanoff, M. Lafora's disease, distinct clinico-pathologic form of Unverricht's syndrome. Arch. Neurol. 12, 172–188 (1965).
Sakai, M., Austin, J., Witmer, F. & Trueb, L. Studies in myoclonus epilepsy (Lafora body form). Neurol. 20, 160–176 (1970).
Carpenter, S. & Karpati, G. Sweat gland duct cells in Lafora disease: diagnosis by skin biopsy. Neurol. 31, 1564–1568 (1981).
Carpenter, S., Karpati, G., Andermann, F., Jacob, J.C. & Andermann, E. Lafora's disease: peroxisomal storage in skeletal muscle. Neurol. 24, 531–538 (1974).
Serratosa, J. et al. The gene for progressive myoclonus epilepsy of the Lafora type maps to chromosome 6q. Hum. Molec. Genet. 9, 1657–1663 (1995).
Sainz, J. et al. Lafora progressive myoclonus epilepsy: narrowing the chromosome 6q24 locus by recombinations and homozygosities. Am. J. Hum. Genet . 61, 1205–1209 ( 1997).
Kozak, M. Interpreting cDNA sequences: some insights from studies on translation. Mamm. Genome 7, 563–574 (1996).
Yuvaniyama, J., Denu, J.M., Dixon, J.E. & Saper, M.A. Crystal structure of the dual specificity protein phosphatase VHR. Science 272, 1328–1331 (1996).
Denu, J.M., Stuckey, J.A., Saper, M.A. & Dixon, J.E. Form and function in protein dephosphorylation. Cell 87, 361–364 (1996).
Laporte, J. et al. A gene mutated in X-linked myotubular myopathy defines a new putative tyrosine phosphatase family conserved in yeast. Nature Genet. 13, 175–182 ( 1996).
Cui, X. et al. Association of SET domain and myotubularin-related proteins modulates growth control. Nature Genet. 18, 331– 337 (1998).
McLaughlin, S. & Dixon, J.E. Alternative splicing gives rise to a nuclear protein tyrosine phosphatase in Drosophila. J. Biol. Chem. 268, 6839–6842 (1993).
Tonks, N.K. & Neel, B.G. From form to function: signaling by protein tyrosine phosphatases. Cell 87, 365–368 (1996).
Wang, Q.M., Fiol, C.J., DePaoli-Roach, A.A. & Roach, P.J. Glycogen synthase kinase-3ß is a dual specificity kinase differentially regulated by tyrosine and /threonine phosphorylation. J. Biol. Chem. 20, 14566–14574 ( 1994).
Gurd, J.W. & Bissoon, N. The N-methyl-D-aspartate receptor subunits NR2A and NR2B bind to the SH2 domains of phospholipase C-gamma. J. Neurochem. 69, 623–630 (1997).
Llinas, R., Moreno, H., Sugimori, M., Mohammadi, M. & Schlessinger, J. Differential pre- and postsynaptic modulation of chemical transmission in the squid giant synapse by tyrosine phosphorylation. Proc. Natl Acad. Sci. USA 94, 1990– 1994 (1997).
Lopes-Cendes, I. et al. Searching for the gene causing Lafora body disease. Epilepsia 36, 6 (1995).
Acharya, J.N., Satishchandra, P. & Shankar, S.K. Lafora's disease in south India: a clinical, electrophysiologic, and pathologic study. Epilepsia 34, 476– 487 (1993).
Fong, C.G. et al. Lafora's progressive myoclonus epilepsy: Italian families narrow the chromosome 6q24 locus to less than 1cM. Epilepsia 39, 4–5 (1998).
Acknowledgements
We thank J. Rommens, L. Osborne, S. Beck, K. Thorpe, Q. Zhang and the Bloorview Epilepsy Program. Supported by grants from the Hospital for Sick Children (HSC) Foundation and the Medical Research Council of Canada (MRC) to S.W.S. A.J.M. and I.D. are funded by the Wellcome Trust. A.V.D. was supported by NIH grant 5P01-NS21908 (UCLA Comprehensive Epilepsy Program) and contributions from A. Malenfant (Quebec Lafora Society) and V. Faludi (Sweden Lafora effort). S.W.S. is a Scholar of the MRC, L.-C.T. is a Senior Scientist of the MRC and Sellers Chair in Cystic Fibrosis Research at HSC and International Scholar of the Howard Hughes Medical Institute. S.W.S., L.-C.T. and G.R. are members of the Canadian Genetic Disease Network.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Minassian, B., Lee, J., Herbrick, JA. et al. Mutations in a gene encoding a novel protein tyrosine phosphatase cause progressive myoclonus epilepsy. Nat Genet 20, 171–174 (1998). https://doi.org/10.1038/2470
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/2470
This article is cited by
-
Lafora progressive myoclonus epilepsy: Disease mechanism and therapeutic attempts
Journal of Biosciences (2024)
-
Gys1 Antisense Therapy Prevents Disease-Driving Aggregates and Epileptiform Discharges in a Lafora Disease Mouse Model
Neurotherapeutics (2023)
-
Beneficial Effect of Fingolimod in a Lafora Disease Mouse Model by Preventing Reactive Astrogliosis-Derived Neuroinflammation and Brain Infiltration of T-lymphocytes
Molecular Neurobiology (2023)
-
Progressive Myoklonusepilepsien
Clinical Epileptology (2023)
-
Early Treatment with Metformin Improves Neurological Outcomes in Lafora Disease
Neurotherapeutics (2023)