Abstract
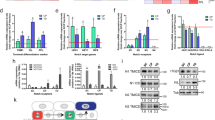
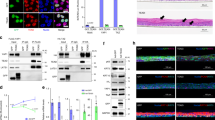
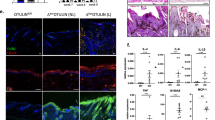
Notch proteins are important in binary cell-fate decisions and inhibiting differentiation in many developmental systems1, and aberrant Notch signaling is associated with tumorigenesis2,3,4,5. The role of Notch signaling in mammalian skin is less well characterized and is mainly based on in vitro studies, which suggest that Notch signaling induces differentiation in mammalian skin6,7. Conventional gene targeting is not applicable to establishing the role of Notch receptors or ligands in the skin because Notch1−/− embryos die during gestation8,9,10,11,12. Therefore, we used a tissue-specific inducible gene-targeting approach to study the physiological role of the Notch1 receptor in the mouse epidermis and the corneal epithelium of adult mice. Unexpectedly, ablation of Notch1 results in epidermal and corneal hyperplasia followed by the development of skin tumors and facilitated chemical-induced skin carcinogenesis. Notch1 deficiency in skin and in primary keratinocytes results in increased and sustained expression of Gli2, causing the development of basal-cell carcinoma–like tumors. Furthermore, Notch1 inactivation in the epidermis results in derepressed β-catenin signaling in cells that should normally undergo differentiation. Enhanced β-catenin signaling can be reversed by re-introduction of a dominant active form of the Notch1 receptor. This leads to a reduction in the signaling-competent pool of β-catenin, indicating that Notch1 can inhibit β-catenin-mediated signaling. Our results indicate that Notch1 functions as a tumor-suppressor gene in mammalian skin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Artavanis-Tsakonas, S., Rand, M.D. & Lake, R.J. Notch signaling: cell fate control and signal integration in development. Science 284, 770–776 (1999).
Ellisen, L.W. et al. TAN-1, the human homolog of the Drosophila notch gene, is broken by chromosomal translocations in T lymphoblastic neoplasms. Cell 66, 649–661 (1991).
Jhappan, C. et al. Expression of an activated Notch-related int-3 transgene interferes with cell differentiation and induces neoplastic transformation in mammary and salivary glands. Genes Dev. 6, 345–355 (1992).
Zagouras, P., Stifani, S., Blaumueller, C.M., Carcangiu, M.L. & Artavanis-Tsakonas, S. Alterations in Notch signaling in neoplastic lesions of the human cervix. Proc. Natl. Acad. Sci. USA 92, 6414–6418 (1995).
Capobianco, A.J., Zagouras, P., Blaumueller, C.M., Artavanis-Tsakonas, S. & Bishop, J.M. Neoplastic transformation by truncated alleles of human NOTCH1/TAN1 and NOTCH2. Mol. Cell. Biol. 17, 6265–6273 (1997).
Lowell, S., Jones, P., Le Roux, I., Dunne, J. & Watt, F.M. Stimulation of human epidermal differentiation by delta-notch signalling at the boundaries of stem-cell clusters. Curr. Biol. 10, 491–500 (2000).
Rangarajan, A. et al. Notch signaling is a direct determinant of keratinocyte growth arrest and entry into differentiation. EMBO J. 20, 3427–3436 (2001).
Conlon, R.A., Reaume, A.G. & Rossant, J. Notch1 is required for the coordinate segmentation of somites. Development 121, 1533–1545 (1995).
Swiatek, P.J., Lindsell, C.E., del Amo, F.F., Weinmaster, G. & Gridley, T. Notch1 is essential for postimplantation development in mice. Genes Dev. 8, 707–719 (1994).
Xue, Y. et al. Embryonic lethality and vascular defects in mice lacking the Notch ligand Jagged1. Hum. Mol. Genet. 8, 723–730 (1999).
Hrabe de Angelis, M., McIntyre, J. 2nd & Gossler, A. Maintenance of somite borders in mice requires the Delta homologue DII1. Nature 386, 717–721 (1997).
Hamada, Y. et al. Mutation in ankyrin repeats of the mouse Notch2 gene induces early embryonic lethality. Development 126, 3415–3424 (1999).
Pear, W.S. et al. Exclusive development of T-cell neoplasms in mice transplanted with bone marrow expressing activated Notch alleles. J. Exp. Med. 183, 2283–2291 (1996).
Uyttendaele, H. et al. Notch4/int-3, a mammary proto-oncogene, is an endothelial cell-specific mammalian Notch gene. Development 122, 2251–2259 (1996).
Milner, L.A. & Bigas, A. Notch as a mediator of cell fate determination in hematopoiesis: evidence and speculation. Blood 93, 2431–2448 (1999).
Missero, C., Di Cunto, F., Kiyokawa, H., Koff, A. & Dotto, G.P. The absence of p21Cip1/WAF1 alters keratinocyte growth and differentiation and promotes ras-tumor progression. Genes Dev. 10, 3065–3075 (1996).
Philipp, J., Vo, K., Gurley, K.E., Seidel, K. & Kemp, C.J. Tumor suppression by p27Kip1 and p21Cip1 during chemically induced skin carcinogenesis. Oncogene 18, 4689–4698 (1999).
Weinberg, W.C. et al. Genetic deletion of p21WAF1 enhances papilloma formation but not malignant conversion in experimental mouse skin carcinogenesis. Cancer Res. 59, 2050–2054 (1999).
Topley, G.I., Okuyama, R., Gonzales, J.G., Conti, C. & Dotto, G.P. p21(WAF1/Cip1) functions as a suppressor of malignant skin tumor formation and a determinant of keratinocyte stem-cell potential. Proc. Natl. Acad. Sci. USA 96, 9089–9094 (1999).
Callahan, C.A. & Oro, A.E. Monstrous attempts at adnexogenesis: regulating hair follicle progenitors through Sonic hedgehog signaling. Curr. Opin. Genet. Dev. 11, 541–546 (2001).
Dahmane, N. et al. The Sonic Hedgehog-Gli pathway regulates dorsal brain growth and tumorigenesis. Development 128, 5201–5212 (2001).
Grachtchouk, M. et al. Basal cell carcinomas in mice overexpressing Gli2 in skin. Nat. Genet. 24, 216–217 (2000).
Hennings, H. et al. Calcium regulation of growth and differentiation of mouse epidermal cells in culture. Cell 19, 245–254 (1980).
van Noort, M., Meeldijk, J., van der Zee, R., Destree, O. & Clevers, H. Wnt signaling controls the phosphorylation status of β-catenin. J. Biol. Chem. 277, 17901–17905 (2002).
Polakis, P. Wnt signaling and cancer. Genes Dev. 14, 1837–1851 (2000).
Hovanes, K. et al. β-catenin-sensitive isoforms of lymphoid enhancer factor-1 are selectively expressed in colon cancer. Nat. Genet. 28, 53–57 (2001).
Radtke, F. et al. Deficient T cell fate specification in mice with an induced inactivation of Notch1. Immunity 10, 547–558 (1999).
Indra, A.K. et al. Temporally-controlled site-specific mutagenesis in the basal layer of the epidermis: comparison of the recombinase activity of the tamoxifen-inducible Cre-ER(T) and Cre-ER(T2) recombinases. Nucleic Acids Res. 27, 4324–4327 (1999).
He, T.C. et al. A simplified system for generating recombinant adenoviruses. Proc. Natl. Acad. Sci. USA 95, 2509–2514 (1998).
Hui, C.C., Slusarski, D., Platt, K.A., Holmgren, R. & Joyner, A.L. Expression of three mouse homologs of the Drosophila segment polarity gene cubitus interruptus, Gli, Gli-2, and Gli-3, in ectoderm- and mesoderm-derived tissues suggests multiple roles during postimplantation development. Dev. Biol. 162, 402–413 (1994).
Acknowledgements
We thank G. Badic, E. Säuberly and J. Bamat for histological analysis; B. Sordat, M. Guitard and F. Sierro for technical help; P. Chambon and D. Metzger for the K5–cre–ERT transgenic mice; and T. Jacks and A. Trumpp for the Cdnk1a−/− mice. This work was supported by grants from the Swiss Cancer League/Oncosuisse (to F.R.), the National Cancer Institute of Canada (to C.C.H.) and the US National Institutes of Health (to G.P.D.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nicolas, M., Wolfer, A., Raj, K. et al. Notch1 functions as a tumor suppressor in mouse skin. Nat Genet 33, 416–421 (2003). https://doi.org/10.1038/ng1099
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1099
This article is cited by
-
Influence of copper(I) nicotinate complex on the Notch1 signaling pathway in triple negative breast cancer cell lines
Scientific Reports (2024)
-
miR-184 represses β-catenin and behaves as a skin tumor suppressor
Cell Death & Disease (2024)
-
The role of Hedgehog and Notch signaling pathway in cancer
Molecular Biomedicine (2022)
-
Turning the tide on Alzheimer’s disease: modulation of γ-secretase
Cell & Bioscience (2022)
-
Notch signaling pathway: architecture, disease, and therapeutics
Signal Transduction and Targeted Therapy (2022)