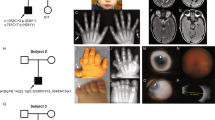
Abstract
Isolated strabismus affects 1–5% of the general population1. Most forms of strabismus are multifactorial in origin; although there is probably an inherited component, the genetics of these disorders remain unclear. The congenital fibrosis syndromes (CFS) represent a subset of monogenic isolated strabismic disorders that are characterized by restrictive ophthalmoplegia, and include congenital fibrosis of the extraocular muscles (CFEOM) and Duane syndrome (DURS)2. Neuropathologic studies indicate that these disorders may result from the maldevelopment of the oculomotor (nIII), trochlear (nIV) and abducens (nVI) cranial nerve nuclei3,4,5. To date, five CFS loci have been mapped (FEOM1, FEOM2, FEOM3, DURS1 and DURS2)6,7,8,9,10, but no genes have been identified. Here, we report three mutations in ARIX (also known as PHOX2A) in four CFEOM2 pedigrees. ARIX encodes a homeodomain transcription factor protein previously shown to be required for nIII/nIV development in mouse and zebrafish11,12. Two of the mutations are predicted to disrupt splicing, whereas the third alters an amino acid within the conserved brachyury-like domain13,14. These findings confirm the hypothesis that CFEOM2 results from the abnormal development of nIII/nIV (ref. 7) and emphasize a critical role for ARIX in the development of these midbrain motor nuclei13,14,15,16,17,18,19.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Engle, E. The genetics of strabismus: Duane, Moebius, and fibrosis syndromes. in Genetic Diseases of the Eye: a Textbook and Atlas. (ed. Traboulsi, E.) 477–512 (Oxford University Press, New York, 1998).
Brown, H.W. Congenital structural muscle anomalies. in Strabismus Ophthalmic Symposium (ed. Allen, J.H.) 205–236 (C. V. Mosby Co., St. Louis, 1950).
Engle, E.C. et al. Oculomotor nerve and muscle abnormalities in congenital fibrosis of the extraocular muscles. Ann. Neurol. 41, 314–325 (1997).
Hotchkiss, M.G., Miller, N.R., Clark, A.W. & Green, W.G. Bilateral Duane's retraction syndrome: A clinical-pathological case report. Arch. Ophthalmol. 98, 870–874 (1980).
Miller, N.R., Kiel, S.M., Green, W.R. & Clark, A.W. Unilateral Duane's retraction syndrome (type 1). Arch. Ophthalmol. 100, 1468–1472 (1982).
Engle, E.C. et al. Congenital fibrosis of the extraocular muscles (autosomal dominant congenital external ophthalmoplegia): genetic homogeneity, linkage refinement, and physical mapping on chromosome 12. Am. J. Hum. Genet. 57, 1086–1094 (1995).
Wang, S. et al. Congenital fibrosis of the extraocular muscles type 2 (CFEOM2), an inherited exotropic strabismus fixus, maps to distal 11q13. Am. J. Hum. Genet. 63, 517–525 (1998).
Doherty, E. et al. CFEOM3: a new extraocular congenital fibrosis syndrome that maps to 16q24.2-q24.3. Invest. Ophthalmol. Vis. Sci. 40, 1687–1694 (1999).
Vincent, C. et al. A proposed new contiguous gene syndrome on 8q consists of branchio-oto-renal (BOR) syndrome, Duane syndrome, a dominant form of hydrocephalus and trapeze aplasia; implications for the mapping of the BOR gene. Hum. Mol. Genet. 3, 1859–1866 (1994).
Appukuttan, B. et al. Localization of a gene for Duane retraction syndrome to chromosome 2q31. Am. J. Hum. Genet. 65, 1639–1646 (1999).
Pattyn, A., Morin, X., Cremer, H., Goridis, C. & Brunet, J.F. Expression and interactions of the two closely related homeobox genes Phox2a and Phox2b during neurogenesis. Development 124, 4065–4075 (1997).
Guo, S. et al. Development of noradrenergic neurons in the zebrafish hindbrain requires BMP, FGF8, and the homeodomain protein soulless/Phox2a. Neuron 24, 555–566 (1999).
Swanson, D.J., Adachi, M. & Lewis, E.J. The homeodomain protein Arix promotes protein kinase A–dependent activation of the dopamine β-hydroxylase promoter through multiple elements and interaction with the coactivator cAMP-response element–binding protein-binding protein. J. Biol. Chem. 275, 2911–2923 (2000).
Adachi, M., Browne, D. & Lewis, E.J. Paired-like homeodomain proteins Phox2a/Arix and Phox2b/NBPhox have similar genetic organization and independently regulate dopamine β-hydroxylase gene transcription. DNA Cell. Biol. 19, 539–554 (2000).
Zellmer, E. et al. A homeodomain protein selectively expressed in noradrenergic tissue regulates transcription of neurotransmitter biosynthetic genes. J. Neurosci. 15, 8109–8120 (1995).
Morin, X. et al. Defects in sensory and autonomic ganglia and absence of locus coeruleus in mice deficient for the homeobox gene Phox2a. Neuron 18, 411–423 (1997).
Yang, C. et al. Paired-like homeodomain proteins, Phox2a and Phox2b, are responsible for noradrenergic cell–specific transcription of the dopamine β-hydroxylase gene. J. Neurochem. 71, 1813–1826 (1998).
Pattyn, A., Morin, X., Cremer, H., Goridis, C. & Brunet, J.F. The homeobox gene Phox2b is essential for the development of autonomic neural crest derivatives. Nature 399, 366–370 (1999).
Pattyn, A., Goridis, C. & Brunet, J.F. Specification of the central noradrenergic phenotype by the homeobox gene Phox2b. Mol. Cell. Neurosci. 15, 235–243 (2000).
Shapiro, M.B. & Senapathy, P. RNA splice junctions of different classes of eukaryotes: sequence statistics and functional implications in gene expression. Nucleic Acids Res. 15, 7155–7174 (1987).
Zhang, M.Q. Statistical features of human exons and their flanking regions. Hum. Mol. Genet. 7, 919–932 (1998).
Teraoka, S.N. et al. Splicing defects in the ataxia-telangiectasia gene, ATM: underlying mutations and consequences. Am. J. Hum. Genet. 64, 1617–1631 (1999).
Tullio-Pelet, A. et al. Mutant WD-repeat protein in triple-A syndrome. Nature Genet. 26, 332–335 (2000).
Valarche, I. et al. The mouse homeodomain protein Phox2 regulates Ncam promoter activity in concert with Cux/CDP and is a putative determinant of neurotransmitter phenotype. Development 119, 881–896 (1993).
Tiveron, M.C., Hirsch, M.R. & Brunet, J.F. The expression pattern of the transcription factor Phox2 delineates synaptic pathways of the autonomic nervous system. J. Neurosci. 16, 7649–7660 (1996).
Pattyn, A., Hirsch, M., Goridis, C. & Brunet, J.F. Control of hindbrain motor neuron differentiation by the homeobox gene Phox2b. Development 127, 1349–1358 (2000).
Robertson, D. et al. Dopamine β-hydroxylase deficiency. A genetic disorder of cardiovascular regulation. Hypertension 18, 1–8 (1991).
Engle, E.C., Castro, A.E., Macy, M.E., Knoll, J.H.M. & Beggs, A.H. A gene for isolated congenital ptosis maps to a 3 cM region within 1p32-p34.1. Am. J. Hum. Genet. 60, 1150–1157 (1997).
Osoegawa, K. et al. A bacterial artificial chromosome library for sequencing the complete human genome. Genome Res. 11, 483–496 (2001).
Ross, M., LaBrie, S., McPherson, J. & Stanton, J., VP. Screening large-insert libraries by hybridization. in Current Protocols in Human Genetics (ed. Dracopoli, N. et al.) 5.6.1–5.6.52 (Wiley, New York, 1999).
Acknowledgements
We thank the affected families for their participation in this study, W.-M. Chan, C. St. Hilaire and D. Southwell for technical assistance, C. Walsh for a subset of control DNA samples and J. Scharf for his critical review of the manuscript. This work was supported by National Eye Institute EY12498 and by the Children's Hospital Mental Retardation Research Center (P30 HD18655).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Nakano, M., Yamada, K., Fain, J. et al. Homozygous mutations in ARIX(PHOX2A) result in congenital fibrosis of the extraocular muscles type 2. Nat Genet 29, 315–320 (2001). https://doi.org/10.1038/ng744
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng744
This article is cited by
-
MRI evaluation of cranial nerve abnormalities and extraocular muscle fibrosis in duane retraction syndrome and congenital extraocular muscle fibrosis
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)
-
Clinical and genetic characteristics of Chinese patients with congenital cranial dysinnervation disorders
Orphanet Journal of Rare Diseases (2022)
-
Multi-omic analysis of selectively vulnerable motor neuron subtypes implicates altered lipid metabolism in ALS
Nature Neuroscience (2021)
-
Congenital fibrosis of the extra-ocular muscles (CFEOM) and the cranial dysinnervation disorders
Eye (2020)