Abstract
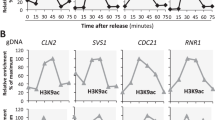
Genes located in chromosomal regions near telomeres are transcriptionally silent, whereas those located in regions away from telomeres are not. Here we show that there is a gradient of acetylation of histone H4 at lysine 16 (H4–Lys16) along a yeast chromosome; this gradient ranges from a hypoacetylated state in regions near the telomere to a hyperacetylated state in more distant regions. The hyperacetylation is regulated by Sas2p, a member of the MYST-type family of histone acetylases, whereas hypoacetylation is under the control of Sir2p, a histone deacetylase. Loss of hyperacetylation is accompanied by an increase in localization of the telomere protein Sir3p and the inactivation of gene expression in telomere-distal regions. Thus, the Sas2p and Sir2p function in concert to regulate transcription in yeast, by acetylating and deacetylating H4–Lys16 in a mechanism that may be common to all eukaryotes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gottschling, D.E., Aparicio, O.M., Billington, B.L. & Zakian, V.A. Position effect at S. cerevisiae telomeres: reversible repression of Pol II transcription. Cell 63, 751–762 (1990).
Louis, E.J. The chromosome ends of Saccharomyces cerevisiae. Yeast 11, 1553–1573 (1995).
Zakian, V.A. Structure, function, and replication of Saccharomyces cerevisiae telomeres. Annu. Rev. Genet. 30, 141–172 (1996).
Vega-Palas, M.A., Venditti, S. & Di Mauro, E. Telomeric transcriptional silencing in a natural context. Nat. Genet. 15, 232–233 (1997).
Loo, S. & Rine, J. Silencing and heritable domains of gene expression. Annu. Rev. Cell. Dev. Biol. 11, 519–548 (1995).
Guarente, L. Diverse and dynamic functions of the Sir silencing complex. Nat. Genet. 23, 281–285 (1999).
Grunstein, M. Yeast heterochromatin: regulation of its assembly and inheritance by histones. Cell 93, 325–328 (1998).
Hecht, A., Laroche, T., Strahl-Bolsinger, S., Gasser, S.M. & Grunstein, M. Histone H3 and H4 N-termini interact with SIR3 and SIR4 proteins: a molecular model for the formation of heterochromatin in yeast. Cell 80, 583–592 (1995).
Martin, S.G., Laroche, T., Suka, N., Grunstein, M. & Gasser, S.M. Relocalization of telomeric Ku and SIR proteins in response to DNA strand breaks in yeast. Cell 97, 621–633 (1999).
Mills, K.D., Sinclair, D.A. & Guarente, L. MEC1-dependent redistribution of the Sir3 silencing protein from telomeres to DNA double-strand breaks. Cell 97, 609–620 (1999).
Renauld, H. et al. Silent domains are assembled continuously from the telomere and are defined by promoter distance and strength, and by SIR3 dosage. Genes Dev. 7, 1133–1145 (1993).
Wyrick, J.J. et al. Chromosomal landscape of nucleosome-dependent gene expression and silencing in yeast. Nature 402, 418–421 (1999).
Hecht, A., Strahl-Bolsinger, S. & Grunstein, M. Spreading of transcriptional repressor SIR3 from telomeric heterochromatin. Nature 383, 92–96 (1996).
Cockell, M. & Gasser, S.M. Nuclear compartments and gene regulation. Curr. Opin. Genet. Dev. 9, 199–205 (1999).
Strahl, B.D. & Allis, C.D. The language of covalent histone modifications. Nature 403, 41–45 (2000).
Braunstein, M., Sobel, R.E., Allis, C.D., Turner, B.M. & Broach, J.R. Efficient transcriptional silencing in Saccharomyces cerevisiae requires a heterochromatin histone acetylation pattern. Mol. Cell. Biol. 16, 4349–4356 (1996).
Braunstein, M., Rose, A.B., Holmes, S.G., Allis, C.D. & Broach, J.R. Transcriptional silencing in yeast is associated with reduced nucleosome acetylation. Genes Dev. 7, 592–604 (1993).
de Bruin, D., Kantrow, S.M., Liberatore, R.A. & Zakian, V.A. Telomere folding is required for the stable maintenance of telomere position effects in yeast. Mol. Cell. Biol. 20, 7991–8000 (2000).
Suka, N., Suka, Y., Carmen, A.A., Wu, J. & Grunstein, M. Highly specific antibodies determine histone acetylation site usage in yeast heterochromatin and euchromatin. Mol. Cell 8, 473–479 (2001).
Megee, P.C., Morgan, B.A., Mittman, B.A. & Smith, M.M. Genetic analysis of histone H4: essential role of lysines subject to reversible acetylation. Science 247, 841–845 (1990).
Johnson, L.M., Kayne, P.S., Kahn, E.S. & Grunstein, M. Genetic evidence for an interaction between SIR3 and histone H4 in the repression of the silent mating loci in Saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 87, 6286–6290 (1990).
Aparicio, O.M., Billington, B.L. & Gottschling, D.E. Modifiers of position effect are shared between telomeric and silent mating-type loci in S. cerevisiae. Cell 66, 1279–1287 (1991).
Carmen, A.A., Milne, L. & Grunstein, M. Acetylation of the yeast histone H4 N terminus regulates its binding to heterochromatin protein SIR3. J. Biol. Chem. 277, 4778–4781 (2002).
Imai, S., Armstrong, C.M., Kaeberlein, M. & Guarente, L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature 403, 795–800 (2000).
Smith, E.R. et al. ESA1 is a histone acetyltransferase that is essential for growth in yeast. Proc. Natl Acad. Sci. USA 95, 3561–3565 (1998).
Kimura, A. & Horikoshi, M. Tip60 acetylates six lysines of a specific class in core histones in vitro. Genes Cells 3, 789–800 (1998).
Hilfiker, A., Hilfiker-Kleiner, D., Pannuti, A. & Lucchesi, J.C. mof, a putative acetyl transferase gene related to the Tip60 and MOZ human genes and to the SAS genes of yeast, is required for dosage compensation in Drosophila. EMBO J. 16, 2054–2060 (1997).
Reifsnyder, C., Lowell, J., Clarke, A. & Pillus, L. Yeast SAS silencing genes and human genes associated with AML and HIV-1 Tat interactions are homologous with acetyltransferases. Nat. Genet. 14, 42–49 (1996).
Ehrenhofer-Murray, A.E., Rivier, D.H. & Rine, J. The role of Sas2, an acetyltransferase homologue of Saccharomyces cerevisiae, in silencing and ORC function. Genetics 145, 923–934 (1997).
Thompson, J.S., Ling, X. & Grunstein, M. Histone H3 amino terminus is required for telomeric and silent mating locus repression in yeast. Nature 369, 245–247 (1994).
Meijsing, S.H. & Ehrenhofer-Murray, A.E. The silencing complex SAS-I links histone acetylation to the assembly of repressed chromatin by CAF-I and Asf1 in Saccharomyces cerevisiae. Genes. Dev. 15, 3169–3182 (2001).
Clarke, A.S., Lowell, J.E., Jacobson, S.J. & Pillus, L. Esa1p is an essential histone acetyltransferase required for cell cycle progression. Mol. Cell. Biol. 19, 2515–2526 (1999).
Yamamoto, T. & Horikoshi, M. Novel substrate specificity of the histone acetyltransferase activity of HIV-1-Tat interactive protein Tip60. J. Biol. Chem. 272, 30595–30598 (1997).
Osada, S. et al. The yeast SAS (something about silencing) protein complex contains a MYST-type putative acetyltransferase and functions with chromatin assembly factor ASF1. Genes Dev. 15, 3155–3168 (2001).
Yan, Y., Barlev, N.A., Haley, R.H., Berger, S.L. & Marmorstein, R. Crystal structure of yeast Esa1 suggests a unified mechanism for catalysis and substrate binding by histone acetyltransferases. Mol. Cell 6, 1195–1205 (2000).
Takechi, S. & Nakayama, T. Sas3 is a histone acetyltransferase and requires a zinc finger motif. Biochem. Biophys. Res. Commun. 266, 405–410 (1999).
Ikura, T. et al. Involvement of the TIP60 histone acetylase complex in DNA repair and apoptosis. Cell 102, 463–473 (2000).
Rundlett, S.E., Carmen, A.A., Suka, N., Turner, B.M. & Grunstein, M. Transcriptional repression by UME6 involves deacetylation of lysine 5 of histone H4 by RPD3. Nature 392, 831–835 (1998).
Strahl-Bolsinger, S., Hecht, A., Luo, K. & Grunstein, M. SIR2 and SIR4 interactions differ in core and extended telomeric heterochromatin in yeast. Genes Dev. 11, 83–93 (1997).
Pryde, F.E. & Louis, E.J. Limitations of silencing at native yeast telomeres. EMBO J. 18, 2538–2550 (1999).
Suka, N., Luo, K. & Grunstein, M. Sir2p and Sas2p opposingly regulate acetylation of yeast histone H4 lysine 16 and spreading of heterochromatin. Nature Genet. 32 (2002); advance online publication, 15 October 2002 (doi:10.1038/ng1017).
Donze, D. & Kamakaka, R.T. RNA polymerase III and RNA polymerase II promoter complexes are heterochromatin barriers in Saccharomyces cerevisiae. EMBO J. 20, 520–531 (2001).
Turner, B.M., Birley, A.J. & Lavender, J. Histone H4 isoforms acetylated at specific lysine residues define individual chromosomes and chromatin domains in Drosophila polytene nuclei. Cell 69, 375–384 (1992).
Kimura, A. & Horikoshi, M. How do histone acetyltransferases select lysine residues in core histones? FEBS Lett. 431, 131–133 (1998).
Edmondson, D.G., Smith, M.M. & Roth, S.Y. Repression domain of the yeast global repressor Tup1 interacts directly with histones H3 and H4. Genes Dev. 10, 1247–1259 (1996).
Dhalluin, C. et al. Structure and ligand of a histone acetyltransferase bromodomain. Nature 399, 491–496 (1999).
Lachner, M., O'Carroll, D., Rea, S., Mechtler, K. & Jenuwein, T. Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins. Nature 410, 116–120 (2001).
Bannister, A.J. et al. Selective recognition of methylated lysine 9 on histone H3 by the HP1 chromo domain. Nature 410, 120–124 (2001).
Nakayama, J., Rice, J.C., Strahl, B.D., Allis, C.D. & Grewal, S.I. Role of histone H3 lysine 9 methylation in epigenetic control of heterochromatin assembly. Science 292, 110–113 (2001).
Knop, M. et al. Epitope tagging of yeast genes using a PCR-based strategy: more tags and improved practical routines. Yeast 15, 963–972 (1999).
Wodicka, L., Dong, H., Mittmann, M., Ho, M.H. & Lockhart, D.J. Genome-wide expression monitoring in Saccharomyces cerevisiae. Nat. Biotechnol. 15, 1359–1367 (1997).
Acknowledgements
We thank D. Gottschling, K. Nasmyth and J. Rine for plasmids and yeast strains; H. Araki, M. Grunstein, C. L. Peterson and K. Struhl for the protocols for the ChrIP assay; S. Harashima and Y. Ohya for tools and advice on yeast manipulation; K. Hasegawa, Y. Ikejiri, K. Matsubara, S. Okano, T. Sakuno and S. Yoshihara for technical assistance; and T. Suzuki, M. Yamaki and all members of our laboratory for discussions and comments on the manuscript. A.K. is a Research Fellow of the Japan Society for the Promotion of Science. This work was supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan, and by Exploratory Research for Advanced Technology of the Japan Science and Technology Corporation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Kimura, A., Umehara, T. & Horikoshi, M. Chromosomal gradient of histone acetylation established by Sas2p and Sir2p functions as a shield against gene silencing. Nat Genet 32, 370–377 (2002). https://doi.org/10.1038/ng993
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng993
This article is cited by
-
The histone methyltransferase DOT1L is a new epigenetic regulator of pulmonary fibrosis
Cell Death & Disease (2022)
-
SESAME-catalyzed H3T11 phosphorylation inhibits Dot1-catalyzed H3K79me3 to regulate autophagy and telomere silencing
Nature Communications (2022)
-
Metabolic regulation of telomere silencing by SESAME complex-catalyzed H3T11 phosphorylation
Nature Communications (2021)
-
Acetylation of PAX7 controls muscle stem cell self-renewal and differentiation potential in mice
Nature Communications (2021)
-
The SESAME complex regulates cell senescence through the generation of acetyl-CoA
Nature Metabolism (2021)