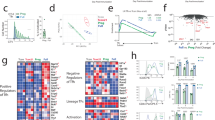
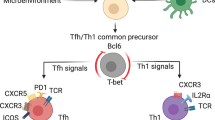
Abstract
Interleukin 2 (IL-2) promotes Foxp3+ regulatory T (Treg) cell responses, but inhibits T follicular helper (TFH) cell development. However, it is not clear how IL-2 affects T follicular regulatory (TFR) cells, a cell type with properties of both Treg and TFH cells. Using an influenza infection model, we found that high IL-2 concentrations at the peak of the infection prevented TFR cell development by a Blimp-1-dependent mechanism. However, once the immune response resolved, some Treg cells downregulated CD25, upregulated Bcl-6 and differentiated into TFR cells, which then migrated into the B cell follicles to prevent the expansion of self-reactive B cell clones. Thus, unlike its effects on conventional Treg cells, IL-2 inhibits TFR cell responses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Yuan, X., Cheng, G. & Malek, T.R. The importance of regulatory T-cell heterogeneity in maintaining self-tolerance. Immunol. Rev. 259, 103–114 (2014).
Lio, C.W. & Hsieh, C.S. A two-step process for thymic regulatory T cell development. Immunity 28, 100–111 (2008).
Burchill, M.A., Yang, J., Vogtenhuber, C., Blazar, B.R. & Farrar, M.A. IL-2 receptor beta-dependent STAT5 activation is required for the development of Foxp3+ regulatory T cells. J. Immunol. 178, 280–290 (2007).
Yao, Z. et al. Nonredundant roles for Stat5a/b in directly regulating Foxp3. Blood 109, 4368–4375 (2007).
Fontenot, J.D., Rasmussen, J.P., Gavin, M.A. & Rudensky, A.Y. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat. Immunol. 6, 1142–1151 (2005).
D'Cruz, L.M. & Klein, L. Development and function of agonist-induced CD25+Foxp3+ regulatory T cells in the absence of interleukin 2 signaling. Nat. Immunol. 6, 1152–1159 (2005).
Cheng, G., Yu, A., Dee, M.J. & Malek, T.R. IL-2R signaling is essential for functional maturation of regulatory T cells during thymic development. J. Immunol. 190, 1567–1575 (2013).
de la Rosa, M., Rutz, S., Dorninger, H. & Scheffold, A. Interleukin-2 is essential for CD4+CD25+ regulatory T cell function. Eur. J. Immunol. 34, 2480–2488 (2004).
Sadlack, B. et al. Ulcerative colitis-like disease in mice with a disrupted interleukin-2 gene. Cell 75, 253–261 (1993).
León, B., Bradley, J.E., Lund, F.E., Randall, T.D. & Ballesteros-Tato, A. FoxP3+ regulatory T cells promote influenza-specific Tfh responses by controlling IL-2 availability. Nat. Commun. 5, 3495 (2014).
Pandiyan, P., Zheng, L., Ishihara, S., Reed, J. & Lenardo, M.J. CD4+CD25+Foxp3+ regulatory T cells induce cytokine deprivation-mediated apoptosis of effector CD4+ T cells. Nat. Immunol. 8, 1353–1362 (2007).
Chen, Y. et al. Foxp3+ regulatory T cells promote T helper 17 cell development in vivo through regulation of interleukin-2. Immunity 34, 409–421 (2011).
Shevach, E.M. Mechanisms of Foxp3+ T regulatory cell-mediated suppression. Immunity 30, 636–645 (2009).
Johnston, R.J., Choi, Y.S., Diamond, J.A., Yang, J.A. & Crotty, S. STAT5 is a potent negative regulator of TFH cell differentiation. J. Exp. Med. 209, 243–250 (2012).
Nurieva, R.I. et al. STAT5 protein negatively regulates T follicular helper (Tfh) cell generation and function. J. Biol. Chem. 287, 11234–11239 (2012).
Ballesteros-Tato, A. et al. Interleukin-2 inhibits germinal center formation by limiting T follicular helper cell differentiation. Immunity 36, 847–856 (2012).
Smigiel, K.S. et al. CCR7 provides localized access to IL-2 and defines homeostatically distinct regulatory T cell subsets. J. Exp. Med. 211, 121–136 (2014).
Gratz, I.K. et al. Cutting Edge: memory regulatory t cells require IL-7 and not IL-2 for their maintenance in peripheral tissues. J. Immunol. 190, 4483–4487 (2013).
Raynor, J. et al. IL-15 Fosters Age-Driven Regulatory T Cell Accrual in the Face of Declining IL-2 Levels. Front. Immunol. 4, 161 (2013).
Chung, Y. et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nat. Med. 17, 983–988 (2011).
Linterman, M.A. et al. Foxp3+ follicular regulatory T cells control the germinal center response. Nat. Med. 17, 975–982 (2011).
Wollenberg, I. et al. Regulation of the germinal center reaction by Foxp3+ follicular regulatory T cells. J. Immunol. 187, 4553–4560 (2011).
Sage, P.T., Francisco, L.M., Carman, C.V. & Sharpe, A.H. The receptor PD-1 controls follicular regulatory T cells in the lymph nodes and blood. Nat. Immunol. 14, 152–161 (2013).
Sage, P.T., Paterson, A.M., Lovitch, S.B. & Sharpe, A.H. The coinhibitory receptor CTLA-4 controls B cell responses by modulating T follicular helper, T follicular regulatory, and T regulatory cells. Immunity 41, 1026–1039 (2014).
Wing, J.B., Ise, W., Kurosaki, T. & Sakaguchi, S. Regulatory T cells control antigen-specific expansion of Tfh cell number and humoral immune responses via the coreceptor CTLA-4. Immunity 41, 1013–1025 (2014).
Oestreich, K.J., Mohn, S.E. & Weinmann, A.S. Molecular mechanisms that control the expression and activity of Bcl-6 in TH1 cells to regulate flexibility with a TFH-like gene profile. Nat. Immunol. 13, 405–411 (2012).
Levin, A.M. et al. Exploiting a natural conformational switch to engineer an interleukin-2 'superkine'. Nature 484, 529–533 (2012).
Aloulou, M. et al. Follicular regulatory T cells can be specific for the immunizing antigen and derive from naive T cells. Nat. Commun. 7, 10579 (2016).
Wang, L. et al. Key role for IL-21 in experimental autoimmune uveitis. Proc. Natl. Acad. Sci. USA 108, 9542–9547 (2011).
Malek, T.R., Yu, A., Vincek, V., Scibelli, P. & Kong, L. CD4 regulatory T cells prevent lethal autoimmunity in IL-2Rbeta-deficient mice. Implications for the nonredundant function of IL-2. Immunity 17, 167–178 (2002).
Zheng, S.G., Wang, J., Wang, P., Gray, J.D. & Horwitz, D.A. IL-2 is essential for TGF-beta to convert naive CD4+CD25− cells to CD25+Foxp3+ regulatory T cells and for expansion of these cells. J. Immunol. 178, 2018–2027 (2007).
Davidson, T.S., DiPaolo, R.J., Andersson, J. & Shevach, E.M. Cutting edge: IL-2 is essential for TGF-beta-mediated induction of Foxp3+ T regulatory cells. J. Immunol. 178, 4022–4026 (2007).
Koch, M.A. et al. The transcription factor T-bet controls regulatory T cell homeostasis and function during type 1 inflammation. Nat. Immunol. 10, 595–602 (2009).
Bedoya, F. et al. Viral antigen induces differentiation of Foxp3+ natural regulatory T cells in influenza virus-infected mice. J. Immunol. 190, 6115–6125 (2013).
Cretney, E. et al. The transcription factors Blimp-1 and IRF4 jointly control the differentiation and function of effector regulatory T cells. Nat. Immunol. 12, 304–311 (2011).
Hollister, K. et al. Insights into the role of Bcl6 in follicular Th cells using a new conditional mutant mouse model. J. Immunol. 191, 3705–3711 (2013).
Rubtsov, Y.P. et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity 28, 546–558 (2008).
Victora, G.D. & Nussenzweig, M.C. Germinal centers. Annu. Rev. Immunol. 30, 429–457 (2012).
Maceiras, A.R. et al. T follicular helper and T follicular regulatory cells have different TCR specificity. Nat. Commun. 8, 15067 (2017).
Hsieh, C.S., Lee, H.M. & Lio, C.W. Selection of regulatory T cells in the thymus. Nat. Rev. Immunol. 12, 157–167 (2012).
Hsieh, C.S., Zheng, Y., Liang, Y., Fontenot, J.D. & Rudensky, A.Y. An intersection between the self-reactive regulatory and nonregulatory T cell receptor repertoires. Nat. Immunol. 7, 401–410 (2006).
Wu, W.W., Sun, Y.H. & Panté, N. Nuclear import of influenza A viral ribonucleoprotein complexes is mediated by two nuclear localization sequences on viral nucleoprotein. Virol. J. 4, 49 (2007).
Avalos, A.M., Busconi, L. & Marshak-Rothstein, A. Regulation of autoreactive B cell responses to endogenous TLR ligands. Autoimmunity 43, 76–83 (2010).
Mandal, M. et al. Epigenetic repression of the Igk locus by STAT5-mediated recruitment of the histone methyltransferase Ezh2. Nat. Immunol. 12, 1212–1220 (2011).
Walker, S.R., Nelson, E.A. & Frank, D.A. STAT5 represses BCL6 expression by binding to a regulatory region frequently mutated in lymphomas. Oncogene 26, 224–233 (2007).
McDonald, P.W. et al. IL-7 signalling represses Bcl-6 and the TFH gene program. Nat. Commun. 7, 10285 (2016).
Vang, K.B. et al. IL-2, -7, and -15, but not thymic stromal lymphopoeitin, redundantly govern CD4+Foxp3+ regulatory T cell development. J. Immunol. 181, 3285–3290 (2008).
Bayer, A.L., Lee, J.Y., de la Barrera, A., Surh, C.D. & Malek, T.R. A function for IL-7R for CD4+CD25+Foxp3+ T regulatory cells. J. Immunol. 181, 225–234 (2008).
Rangel-Moreno, J. et al. The development of inducible bronchus-associated lymphoid tissue depends on IL-17. Nat. Immunol. 12, 639–646 (2011).
Lee, B.O. et al. CD4 T cell-independent antibody response promotes resolution of primary influenza infection and helps to prevent reinfection. J. Immunol. 175, 5827–5838 (2005).
Lim, W.K., Lyashenko, E. & Califano, A. Master regulators used as breast cancer metastasis classifier. Pac. Symp. Biocomput. 2009, 504–515 (2009).
Carro, M.S. et al. The transcriptional network for mesenchymal transformation of brain tumours. Nature 463, 318–325 (2010).
Bar-Joseph, Z., Gifford, D.K. & Jaakkola, T.S. Fast optimal leaf ordering for hierarchical clustering. Bioinformatics 17 (Suppl. 1), S22–S29 (2001).
Eisen, M.B., Spellman, P.T., Brown, P.O. & Botstein, D. Cluster analysis and display of genome-wide expression patterns. Proc. Natl. Acad. Sci. USA 95, 14863–14868 (1998).
Acknowledgements
The authors would like to thank W.J. Leonard (US National Institutes of Health) for providing the Il21-mCherry-Il2-emGFP dual reporter transgenic mice, E. Meffre (Yale University) for providing the Blimp-1 reporter mice, A. Rudensky (Memorial Sloan-Kettering Cancer Center) for providing the Foxp3-DTR-GFP mice, and T.S. Simpler and U. Mudunuru for animal husbandry. This work was supported by University of Alabama at Birmingham (UAB) and National Institutes of Health grants 1R01 AI110480 to A.B.-T., R01 AI116584 to B.L., AI097357 and AI109962 to T.D.R., AIAI109962 to F.E.L., AI061061 to A.S.W., and AI049360 to A.J.Z. The X-RAD 320 unit was purchased using a Research Facility Improvement Grant, 1 G20RR022807-01, from the National Center for Research Resources, National Institutes of Health. Support for the UAB flow cytometry core was provided by grants P30 AR048311 and P30 AI027767.
Author information
Authors and Affiliations
Contributions
A.B.-T. designed and performed the experiments with help from B.L., H.B., M.J.F., J.E.B. and D.B. A.B.-T., D.B. and B.L. analyzed the data. T.T.M.-L. and A.S.W. analyzed the RNAseq data. A.B.-T. wrote the manuscript. D.B. and B.L. contributed to data interpretation and manuscript editing. A.J.Z. performed LCMV infections. T.D.R. and F.E.L. contributed to manuscript editing and discussion, and provided reagents that were critical to this work. All of the authors reviewed the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 TFR cell responses.
(A) B6 mice were infected with PR8 and serial cryosections from the mLN on day 30 were stained with anti-B220 (blue), anti-FoxP3 (green) and anti-CD35 (red) or with anti-IgD (green), anti-FoxP3 (purple) and anti-CD35 (red). Stained cryosections were analyzed by fluorescence microscopy. White lines define the border of the B-cell follicle. Images are representative of three independent experiments. (B) B6 mice were infected with PR8 and serial cryosections from the mLN on day 30 were stained with anti-B220 (blue), anti-FoxP3 (red) and anti-CD4 (green). Stained cryosections were analyzed by fluorescence microscopy. Images are representative of two independent experiments. (C) B6 mice were infected with PR8 and cryosections of the mLN were stained with anti-B220 (blue), anti-FoxP3 (green) and anti-CD35 (red) on days 10 and 30. White lines define the border of the B cell follicle. Images are representative of three independent experiments. (D-E) B6 mice were immunized with 50 μg of hemaglutinin (HA) adsorbed to alum, and the frequency (D) and number (E) of Bcl-6hiCXCR5hi TFR cells were calculated in the mLN at the indicated time points. Representative plots were gated on FoxP3+CD69hiCD19- CD4+ T cells. Data are representative of two independent experiments (mean ± SD of 4-5 mice per group). *P < 0.05, **P < 0.01, ***P < 0.001. P values were determined using a two-tailed Student´s t-test. (F-G) B6 mice were infected with LCMV-Armstrong and the frequency (F) and number (G) of Bcl-6hiCXCR5hi TFR cells were calculated in the mLN at the indicated time-points. Representative plots were gated on FoxP3+CD69hiCD19- CD4+ T cells. Data are representative of two independent experiments (mean ± SD of 5 mice per group). *P < 0.05, **P < 0.01, ***P < 0.001. P values were determined using a two-tailed Student´s t-test.
Supplementary Figure 2 CD25loFoxp3+ cells fail to up-regulate TFR cell markers at the peak of the infection.
(A-C) B6 mice were infected with PR8 and CD69hiFoxP3+CD4+ T cells from the mLN were analyzed for expression of CD25, Bcl-6, CXCR5 and PD-1 on day 10 (A) and day 30 (B) after infection by flow cytometry. Data are representative of five independent experiments. Data are shown as the mean ± SD (n=4-5 mice). (C) B6 mice were infected with PR8 and the frequency of Bcl-6hiCXCR5hi TFR cells within the CD25loCD69hiFoxP3+CD4+ T cell population were calculated by flow cytometry at the indicated time-points. Data are shown as the mean ± SD (n=4-5 mice/time point). Data are representative of three independent experiments. (D-E). B6 mice were infected with LCMV (D) or immunized with HA adsorbed to alum (E) and the frequency of CD25hi and CD25lo FoxP3+CD69hi CD4+ T cells from the mLNs with a Bcl-6hiCXCR5hi phenotype is shown. Data are representative of two independent experiments (mean ± SD of 5 mice per group). (F) GSEA showing enrichment in the IL-2–STAT5 Hallmark-signaling pathway in Treg cells vs TFR cells (adjusted p-value <0.05, Log2fold change greater than or equal to 1). Three replicates for each cell type were obtained from three independent experiments.
Supplementary Figure 3 IL-2 production in PR8-infected and HA-alum immunized mice.
I l21-mCherry-Il2-emGFP mice were infected with PR8 (left panel) or immunized with HA adsorbed to alum (right panel) and cells from the mLN were analyzed on day 10. The frequency of GFP/IL-2+ cells within the CD4+ T-cell population is shown. Data are shown as the mean ± SD (n=3-5 mice per group). Data are representative of two independent experiments.
Supplementary Figure 4 NP-specific TFH cell response in TFR cell-deficient mice
(A-B) B6 and Bcl-6fl/flFoxp3YFP/Cre mice were infected with PR8 and cells from the mLN were analyzed by flow cytometry on day 30. The frequency (A) and number (B) of NP-specific CD4+ T cells with a CXCR5hiPD-1hi TFH cell phenotype were determined. Data are shown as the mean ± SD (n=3-5 mice). Data are representative of three independent experiments.
Supplementary Figure 5 TFH cell and B cell responses in TFR cell-depleted mice
(A-E) Tcr−/− mice were irradiated and reconstituted with a 50:50 mix of BM from CD45.1+ FoxP3-DTR and CD45.2+ B6 donors (FoxP3-B6) or from CD45.1+ FoxP3-DTR and CD45.2+ Cxcr5−/− donors (FoxP3- Cxcr5−/−). Reconstituted FoxP3-B6 and FoxP3-Cxcr5−/− chimeras were infected with PR8, treated with PBS or DT every four days starting 20 days after infection, and cells from the mLN were analyzed on day 50. Frequency (A) and number (B) of Bcl6hiCXCR5hi TFH cells. Representative plots were gated on FoxP3-CD19- CD4+ T cells. Frequency (c) and number (D) of CD19+CD138-GL-7+CD38loCD95hi GC B cells. Number (E) of CD138+ ASCs. Data are shown as the mean ± SD (n=3-5 mice). Data are representative of three independent experiments. P values were determined using a two-tailed Student´s t-test. (F-L) B6 mice were infected with PR8 and treated daily with 15,000 U of rIL-2 or PBS starting 20 days after infection. Cells from the mLN were analyzed by flow cytometry on day 30. Number (F) of TFR cells in PBS and rIL-2 treated mice. Frequency (G) and number (H) of Bcl-6hiCXCR5hi TFH cells. Representative plots were gated on FoxP3-CD19- CD4+ T cells. Frequency (I) and number (J) of NP-specific CD4+ T cells with a Bcl-6hiCXCR5hi TFH cell phenotype. Representative plots were gated on FoxP3-CD19- NP+CD4+ T cells. The frequency (K) and number (L) of CD19+CD138-GL-7+CD95hi GC B cells. Data are shown as the mean ± SD (n=4-5 mice). Data are representative of four independent experiments. P values were determined using a two-tailed Student´s t-test.
Supplementary Figure 6 Influenza-infected Bcl-6fl/flFoxp3YFP/Cre mice develop self-reactive ASC responses.
The presence of ANAs in the serum from naïve and age-matched day 30 influenza-infected B6 (top panels) and Bcl6fl/flFoxp3YFP/Cre mice (bottom panels) was determined by fluorescence microscopy using HEp-2 slides. Images are representative of two independent experiments (n=4-5 mice/group).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1–3 (PDF 2690 kb)
Rights and permissions
About this article
Cite this article
Botta, D., Fuller, M., Marquez-Lago, T. et al. Dynamic regulation of T follicular regulatory cell responses by interleukin 2 during influenza infection. Nat Immunol 18, 1249–1260 (2017). https://doi.org/10.1038/ni.3837
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3837
This article is cited by
-
B cell-reactive triad of B cells, follicular helper and regulatory T cells at homeostasis
Cell Research (2024)
-
Same yet different — how lymph node heterogeneity affects immune responses
Nature Reviews Immunology (2023)
-
Regulatory T cells (Tregs) in liver fibrosis
Cell Death Discovery (2023)
-
Proximity-enabled covalent binding of IL-2 to IL-2Rα selectively activates regulatory T cells and suppresses autoimmunity
Signal Transduction and Targeted Therapy (2023)
-
Aiolos represses CD4+ T cell cytotoxic programming via reciprocal regulation of TFH transcription factors and IL-2 sensitivity
Nature Communications (2023)