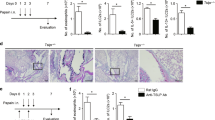
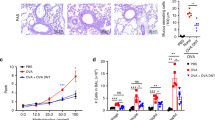
Abstract
It is widely accepted that T helper type 1 (TH1) cytokines such as interferon-γ (IFN-γ) antagonize allergic diseases mediated by TH2 cytokines. The 'hygiene hypothesis' has also proposed that decreased childhood exposure to pathogen-derived TH1 cytokines may underlie the recent increased prevalence of asthma, a TH2-mediated disease. We show here that influenza A viral infection, which induces large amounts of intrapulmonary IFN-γ production, unexpectedly enhanced later allergen-specific asthma and promoted dual allergen–specific TH1 and TH2 responses. Pulmonary dendritic cells obtained from the lung after viral clearance and resolution of acute inflammation conferred enhanced allergic disease and concurrent TH1 and TH2 immune responses, and these effects were dependent on IFN-γ secreted during the acute viral infection. Thus, respiratory viral infection and the acute TH1 response can positively regulate TH2-dependent allergic pulmonary disease in vivo, at least in part, by altering pulmonary dendritic cell function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Holgate, S.T. The epidemic of allergy and asthma. Nature 402, B2–4 (1999).
Gern, J.E. & Busse, W.W. Relationship of viral infections to wheezing illnesses and asthma. Nat. Rev. Immunol. 2, 132–138 (2002).
Redd, S.C. Asthma in the United States: burden and current theories. Environ. Health Perspect. 110, 557–560 (2002).
Wills-Karp, M., Santeliz, J. & Karp, C.L. The germless theory of allergic disease: revisiting the hygiene hypothesis. Nat. Rev. Immunol. 1, 69–75 (2001).
Morel, P.A. & Oriss, T.B. Crossregulation between Th1 and Th2 cells. Crit. Rev. Immunol. 18, 275–303 (1998).
Erb, K.J., Holloway, J.W., Sobeck, A., Moll, H. & Le Gros, G. Infection of mice with Mycobacterium bovis-Bacillus Calmette-Guerin (BCG) suppresses allergen-induced airway eosinophilia. J. Exp. Med. 187, 561–569 (1998).
Lewis, D.B. Allergy immunotherapy and inhibition of Th2 immune responses: a sufficient strategy? Curr. Opin. Immunol. 14, 644–651 (2002).
Schwarze, J. & Gelfand, E.W. Respiratory viral infections as promoters of allergic sensitization and asthma in animal models. Eur. Respir. J. 19, 341–349 (2002).
Martinez, F.D. What have we learned from the Tucson Children's Respiratory Study? Paediatr. Respir. Rev. 3, 193–197 (2002).
Schwarze, J., Hamelmann, E., Bradley, K.L., Takeda, K. & Gelfand, E.W. Respiratory syncytial virus infection results in airway hyperresponsiveness and enhanced airway sensitization to allergen. J. Clin. Invest. 100, 226–233 (1997).
Doherty, P.C. et al. Effector CD4+ and CD8+ T-cell mechanisms in the control of respiratory virus infections. Immunol. Rev. 159, 105–117 (1997).
Roman, E. et al. CD4 effector T cell subsets in the response to influenza: heterogeneity, migration, and function. J. Exp. Med. 196, 957–968 (2002).
Doyle, A.G., Buttigieg, K., Groves, P., Johnson, B.J. & Kelso, A. The activated type 1-polarized CD8+ T cell population isolated from an effector site contains cells with flexible cytokine profiles. J. Exp. Med. 190, 1081–1092 (1999).
Sarawar, S.R., Sangster, M., Coffman, R.L. & Doherty, P.C. Administration of anti-IFN-γ antibody to β2-microglobulin-deficient mice delays influenza virus clearance but does not switch the response to a T helper cell 2 phenotype. J. Immunol. 153, 1246–1253 (1994).
Graham, M.B. et al. Response to influenza infection in mice with a targeted disruption in the interferon γ gene. J. Exp. Med. 178, 1725–1732 (1993).
Eichelberger, M.C., Wang, M.L., Allan, W., Webster, R.G. & Doherty, P.C. Influenza virus RNA in the lung and lymphoid tissue of immunologically intact and CD4-depleted mice. J. Gen. Virol. 72, 1695–1698 (1991).
Lloyd, C.M., Gonzalo, J.A., Coyle, A.J. & Gutierrez-Ramos, J.C. Mouse models of allergic airway disease. Adv. Immunol. 77, 263–295 (2001).
Finkelman, F.D. et al. Lymphokine control of in vivo immunoglobulin isotype selection. Annu. Rev. Immunol. 8, 303–333 (1990).
Coffman, R.L., Lebman, D.A. & Rothman, P. Mechanism and regulation of immunoglobulin isotype switching. Adv. Immunol. 54, 229–270 (1993).
Lambrecht, B.N. et al. Myeloid dendritic cells induce Th2 responses to inhaled antigen, leading to eosinophilic airway inflammation. J. Clin. Invest. 106, 551–559 (2000).
Julia, V. et al. A restricted subset of dendritic cells captures airborne antigens and remains able to activate specific T cells long after antigen exposure. Immunity 16, 271–283 (2002).
Cella, M., Facchetti, F., Lanzavecchia, A. & Colonna, M. Plasmacytoid dendritic cells activated by influenza virus and CD40L drive a potent TH1 polarization. Nat. Immunol. 1, 305–310 (2000).
Brimnes, M.K., Bonifaz, L., Steinman, R.M. & Moran, T.M. Influenza virus-induced dendritic cell maturation is associated with the induction of strong T cell immunity to a coadministered, normally nonimmunogenic protein. J. Exp. Med. 198, 133–144 (2003).
Kolopp-Sarda, M.N., Bene, M.C., Massin, N., Moulin, J.J. & Faure, G.C. Immunohistological analysis of macrophages, B-cells, and T-cells in the mouse lung. Anat. Rec. 239, 150–157 (1994).
Huang, S. et al. Immune response in mice that lack the interferon-γ receptor. Science 259, 1742–1745 (1993).
Murphy, K.M., Heimberger, A.B. & Loh, D.Y. Induction by antigen of intrathymic apoptosis of CD4+CD8+TCRlo thymocytes in vivo. Science 250, 1720–1723 (1990).
Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat. Rev. Immunol. 3, 133–146 (2003).
Hansen, G., Berry, G., DeKruyff, R.H. & Umetsu, D.T. Allergen-specific Th1 cells fail to counterbalance Th2 cell-induced hyperreactivity but cause severe airway inflammation. J. Clin. Invest. 103, 175–183 (1999).
Randolph, D.A., Stephens, R., Carruthers, C.J. & Chaplin, D.D. Cooperation between Th1 and Th2 cells in a murine model of eosinophilic airway inflammation. J. Clin. Invest. 104, 1021–1029 (1999).
Mo, X.Y., Sarawar, S.R. & Doherty, P.C. Induction of cytokines in mice with parainfluenza pneumonia. J. Virol. 69, 1288–1291 (1995).
Hussell, T., Spender, L.C., Georgiou, A., O'Garra, A. & Openshaw, P.J. Th1 and Th2 cytokine induction in pulmonary T cells during infection with respiratory syncytial virus. J. Gen. Virol. 77, 2447–2455 (1996).
Schwarze, J. et al. IL-5 and eosinophils are essential for the development of airway hyperresponsiveness following acute respiratory syncytial virus infection. J. Immunol. 162, 2997–3004 (1999).
Schwarze, J. et al. CD8 T cells are essential in the development of respiratory syncytial virus-induced lung eosinophilia and airway hyperresponsiveness. J. Immunol. 162, 4207–4211 (1999).
Schwarze, J. et al. Transfer of the enhancing effect of respiratory syncytial virus infection on subsequent allergic airway sensitization by T lymphocytes. J. Immunol. 163, 5729–5734 (1999).
Wohlleben, G. et al. Influenza A virus infection inhibits the efficient recruitment of Th2 cells into the airways and the development of airway eosinophilia. J. Immunol. 170, 4601–4611 (2003).
Kuchroo, V.K., Umetsu, D.T., DeKruyff, R.H. & Freeman, G.J. The TIM gene family: emerging roles in immunity and disease. Nat. Rev. Immunol. 3, 454–462 (2003).
Krug, N. et al. T-cell cytokine profile evaluated at the single cell level in BAL and blood in allergic asthma. Am. J. Respir. Cell. Mol. Biol. 14, 319–326 (1996).
ten Hacken, N.H. et al. Elevated serum interferon-γ in atopic asthma correlates airways responsiveness and circadian peak expiratory flow variation. Eur. Respir. J. 11, 312–316 (1998).
Sampath, D., Castro, M., Look, D.C. & Holtzman, M.J. Constitutive activation of an epithelial signal transducer and activator of transcription (STAT) pathway in asthma. J. Clin. Invest. 103, 1353–1361 (1999).
Magnan, A.O. et al. Assessment of the Th1/Th2 paradigm in whole blood in atopy and asthma. Increased IFN-γ-producing CD8+ T cells in asthma. Am. J. Respir. Crit. Care Med. 161, 1790–1796 (2000).
Yamamoto, N. et al. Dendritic cells are associated with augmentation of antigen sensitization by influenza A virus infection in mice. Eur. J. Immunol. 30, 316–326 (2000).
Yamamoto, N. et al. Immune response induced by airway sensitization after influenza A virus infection depends on timing of antigen exposure in mice. J. Virol. 75, 499–505 (2001).
Kamath, A.T., Henri, S., Battye, F., Tough, D.F. & Shortman, K. Developmental kinetics and lifespan of dendritic cells in mouse lymphoid organs. Blood 100, 1734–1741 (2002).
Cherwinski, H.M., Schumacher, J.H., Brown, K.D. & Mosmann, T.R. Two types of mouse helper T cell clone. III. Further differences in lymphokine synthesis between Th1 and Th2 clones revealed by RNA hybridization, functionally monospecific bioassays, and monoclonal antibodies. J. Exp. Med. 166 1229–1244 (1987).
Tsitoura, D.C. et al. Respiratory infection with influenza A virus interferes with of tolerance to aeroallergens. J. Immunol. 165, 3484–3491 (2000).
Dabbagh, K. et al. Local blockade of allergic airway hyperreactivity and inflammation by the poxvirus-derived pan-CC-chemokine inhibitor vCCI. J. Immunol. 165, 3418–3422 (2000).
Dabbagh, K., Dahl, M.E., Stepick-Biek, P. & Lewis, D.B. Toll-like receptor 4 is required for optimal development of Th2 immune responses: role of dendritic cells. J. Immunol. 168, 4524–4530 (2002).
Lyons, A.B., Hasbold, J. & Hodgkin, P.D. Flow cytometric analysis of cell division history using dilution of carboxyfluorescein diacetate succinimidyl ester, a stably integrated fluorescent probe. Methods Cell Biol. 63, 375–398 (2001).
Marrack, P., Shimonkevitz, R., Hannum, C., Haskins, K. & Kappler, J. The major histocompatibility complex-restricted antigen receptor on T cells. IV. An antiidiotypic antibody predicts both antigen and I-specificity. J. Exp. Med. 158, 1635–1646 (1983).
Acknowledgements
We thank P. Doherty (University of Melbourne, Victoria, Australia) for providing the stocks of influenza A virus and control allantoic fluid; and M. Khan and E. Adams for technical assistance. Supported by the National Institutes of Health grant RO1 AI-44699, the American Lung Association grant RT-017-N, and the Allergy Foundation of America.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Dahl, M., Dabbagh, K., Liggitt, D. et al. Viral-induced T helper type 1 responses enhance allergic disease by effects on lung dendritic cells. Nat Immunol 5, 337–343 (2004). https://doi.org/10.1038/ni1041
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1041
This article is cited by
-
Interferons as negative regulators of ILC2s in allergic lung inflammation and respiratory viral infections
Journal of Molecular Medicine (2023)
-
Influenza-induced monocyte-derived alveolar macrophages confer prolonged antibacterial protection
Nature Immunology (2020)
-
Hygiene Hypothesis: What Is the Current Thinking?
Current Otorhinolaryngology Reports (2017)
-
Cross-roads in the lung: immune cells and tissue interactions as determinants of allergic asthma
Immunologic Research (2012)
-
Role of dendritic cells: a step forward for the hygiene hypothesis
Cellular & Molecular Immunology (2011)