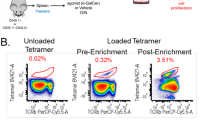
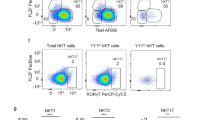
Abstract
Natural killer (NK) cell development is thought to occur in the bone marrow. Here we identify the transcription factor GATA-3 and CD127 (IL-7Rα) as molecular markers of a pathway of mouse NK cell development that originates in the thymus. Thymus-derived CD127+ NK cells repopulated peripheral lymphoid organs, and their homeostasis was strictly dependent on GATA-3 and interleukin 7. The CD127+ NK cells had a distinct phenotype (CD11bloCD16−CD69hiLy49lo) and unusual functional attributes, including reduced cytotoxicity but considerable cytokine production. Those characteristics are reminiscent of human CD56hiCD16− NK cells, which we found expressed CD127 and had more GATA-3 expression than human CD56+CD16+ NK cells. We propose that bone marrow and thymic NK cell pathways generate distinct mouse NK cells with properties similar to those of the two human CD56 NK cell subsets.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
03 November 2006
In the version of this article initially published, the symbols in the key of Figure 7a are incorrect. The filled ovals are Splenic; the filled diamonds are Thymic. The error has been corrected in the HTML and PDF versions of the article.
References
Herberman, R.B., Nunn, M.E., Holden, H.T. & Lavrin, D.H. Natural cytotoxic reactivity of mouse lymphoid cells against syngeneic and allogeneic tumors. II. Characterization of effector cells. Int. J. Cancer 16, 230–239 (1975).
Di Santo, J.P. Natural killer cell developmental pathways: a question of balance. Annu. Rev. Immunol. 24, 257–286 (2006).
Vosshenrich, C.A., Samson Villeger, S.I. & Di Santo, J.P. Distinguishing features of developing natural killer cells. Curr. Opin. Immunol. 17, 151–158 (2005).
Rosmaraki, E.E. et al. Identification of committed NK cell progenitors in adult murine bone marrow. Eur. J. Immunol. 31, 1900–1909 (2001).
Raulet, D.H., Vance, R.E. & McMahon, C.W. Regulation of the natural killer cell receptor repertoire. Annu. Rev. Immunol. 19, 291–330 (2001).
Kim, S. et al. In vivo developmental stages in murine natural killer cell maturation. Nat. Immunol. 3, 523–528 (2002).
Cooper, M.A. et al. In vivo evidence for a dependence on interleukin 15 for survival of natural killer cells. Blood 100, 3633–3638 (2002).
Kennedy, M.K. et al. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J. Exp. Med. 191, 771–780 (2000).
Mrozek, E., Anderson, P. & Caligiuri, M.A. Role of interleukin-15 in the development of human CD56+ natural killer cells from CD34+ hematopoietic progenitor cells. Blood 87, 2632–2640 (1996).
Ranson, T. et al. IL-15 is an essential mediator of peripheral NK cell homeostasis. Blood (2003).
Peschon, J.J. et al. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J. Exp. Med. 180, 1955–1960 (1994).
He, Y.W. & Malek, T.R. Interleukin-7 receptor α is essential for the development of γδ+ T cells, but not natural killer cells. J. Exp. Med. 184, 289–293 (1996).
Moore, T.A., von Freeden-Jeffry, U., Murray, R. & Zlotnik, A. Inhibition of γδ T cell development and early thymocyte maturation in IL-7−/− mice. J. Immunol. 157, 2366–2373 (1996).
Vosshenrich, C.A. et al. Roles for common cytokine receptor γ-chain-dependent cytokines in the generation, differentiation, and maturation of NK cell precursors and peripheral NK cells in vivo. J. Immunol. 174, 1213–1221 (2005).
Rothenberg, E.V. & Taghon, T. Molecular genetics of T cell development. Annu. Rev. Immunol. 23, 601–649 (2005).
Singh, H., Medina, K.L. & Pongubala, J.M. Contingent gene regulatory networks and B cell fate specification. Proc. Natl. Acad. Sci. USA 102, 4949–4953 (2005).
Samson, S.I. et al. GATA3 promotes maturation, IFN-γ production, and liver-specific homing of NK cells. Immunity 19, 701–711 (2003).
Taki, S., Nakajima, S., Ichikawa, E., Saito, T. & Hida, S. IFN regulatory factor-2 deficiency revealed a novel checkpoint critical for the generation of peripheral NK cells. J. Immunol. 174, 6005–6012 (2005).
Townsend, M.J. et al. T-bet regulates the terminal maturation and homeostasis of NK and Vα14i NKT cells. Immunity 20, 477–494 (2004).
Garni-Wagner, B.A. et al. Natural killer cells in the thymus. Studies in mice with severe combined immune deficiency. J. Immunol. 144, 796–803 (1990).
Takeda, K. et al. TRAIL identifies immature natural killer cells in newborn mice and adult mouse liver. Blood 105, 2082–2089 (2005).
Haller, O. & Wigzell, H. Suppression of natural killer cell activity with radioactive strontium: effector cells are marrow dependent. J. Immunol. 118, 1503–1506 (1977).
Puzanov, I.J., Bennett, M. & Kumar, V. IL-15 can substitute for the marrow microenvironment in the differentiation of natural killer cells. J. Immunol. 157, 4282–4285 (1996).
Sanchez, M.J., Muench, M.O., Roncarolo, M.G., Lanier, L.L. & Phillips, J.H. Identification of a common T/natural killer cell progenitor in human fetal thymus. J. Exp. Med. 180, 569–576 (1994).
Spits, H. et al. Early stages in the development of human T, natural killer and thymic dendritic cells. Immunol. Rev. 165, 75–86 (1998).
Michie, A.M. et al. Clonal characterization of a bipotent T cell and NK cell progenitor in the mouse fetal thymus. J. Immunol. 164, 1730–1733 (2000).
Cooper, M.A. et al. Human natural killer cells: a unique innate immunoregulatory role for the CD56bright subset. Blood 97, 3146–3151 (2001).
Hendriks, R.W. et al. Expression of the transcription factor GATA3 is required for the development of the earliest T cell progenitors and correlates with stages of cellular proliferation in the thymus. Eur. J. Immunol. 29, 1912–1918 (1999).
Dong, C. & Flavell, R.A. Control of T helper cell differentiation–in search of master genes. Sci. STKE 2000 PE1 (2000).
Penix, L., Weaver, W.M., Pang, Y., Young, H.A. & Wilson, C.B. Two essential regulatory elements in the human interferon γ promoter confer activation specific expression in T cells. J. Exp. Med. 178, 1483–1496 (1993).
Leiden, J.M. Transcriptional regulation of T cell receptor genes. Annu. Rev. Immunol. 11, 539–570 (1993).
Ma, A., Koka, R. & Burkett, P. Diverse functions of IL-2, IL-15, and IL-7 in lymphoid homeostasis. Annu. Rev. Immunol. 24, 657–679 (2006).
Degli-Esposti, M.A. & Smyth, M.J. Close encounters of different kinds: dendritic cells and NK cells take centre stage. Nat. Rev. Immunol. 5, 112–124 (2005).
Chan, C.W. et al. Interferon-producing killer dendritic cells provide a link between innate and adaptive immunity. Nat. Med. 12, 207–213 (2006).
Taieb, J. et al. A novel dendritic cell subset involved in tumor immunosurveillance. Nat. Med. 12, 214–219 (2006).
Martin-Fontecha, A. et al. Induced recruitment of NK cells to lymph nodes provides IFN-γ for TH1 priming. Nat. Immunol. 5, 1260–1265 (2004).
Ferlazzo, G. & Munz, C. NK cell compartments and their activation by dendritic cells. J. Immunol. 172, 1333–1339 (2004).
Freud, A.G. et al. A human CD34+ subset resides in lymph nodes and differentiates into CD56bright natural killer cells. Immunity 22, 295–304 (2005).
Hansson, M., Kiessling, R., Andersson, B., Karre, K. & Roder, J. NK cell-sensitive T-cell subpopulation in thymus: inverse correlation to host NK activity. Nature 278, 174–176 (1979).
Schott, E., Bonasio, R. & Ploegh, H.L. Elimination in vivo of developing T cells by natural killer cells. J. Exp. Med. 198, 1213–1224 (2003).
Mowen, K.A. & Glimcher, L.H. Signaling pathways in Th2 development. Immunol. Rev. 202, 203–222 (2004).
Haddad, R. et al. Dynamics of thymus-colonizing cells during human development. Immunity 24, 217–230 (2006).
Acknowledgements
We thank B. Canque, G. de Saint Basile, E. Six and M. Albert for thymocytes, peripheral blood mononuclear cells and antibodies for human studies, and R. Hendriks for Gata3+/nlslacZ mice. Supported by the Institut Pasteur, the Institut National de la Santé et Recherche Medicale, the Ligue Nationale Contre le Cancer and the Fondation de la Recherche Médicale, as well as the Pasteur Foundation and Institut National de la Santé et Recherche Medicale (M.G.O.).
Author information
Authors and Affiliations
Contributions
C.A.J.V., M.E.G.-O., S.I.S.-V., L.E., D.G.-G., L.R., S.E. and J.P.D. did the experiments; V.P., O.R.-L. and E.C. provided technical assistance; B.R., A.C. and S.E. contributed to the experimental design and analysis; and C.A.J.V. and J.P.D. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Vosshenrich, C., García-Ojeda, M., Samson-Villéger, S. et al. A thymic pathway of mouse natural killer cell development characterized by expression of GATA-3 and CD127. Nat Immunol 7, 1217–1224 (2006). https://doi.org/10.1038/ni1395
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1395
This article is cited by
-
Reprogramming the tumor immune microenvironment via nanomaterial-mediated dynamic therapy
Nano Research (2023)
-
Innate lymphoid cells and cancer
Nature Immunology (2022)
-
Stem cells-derived natural killer cells for cancer immunotherapy: current protocols, feasibility, and benefits of ex vivo generated natural killer cells in treatment of advanced solid tumors
Cancer Immunology, Immunotherapy (2021)
-
Natural killer cells in cancer biology and therapy
Molecular Cancer (2020)
-
Innate lymphoid cells control signaling circuits to regulate tissue-specific immunity
Cell Research (2020)