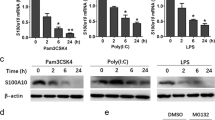
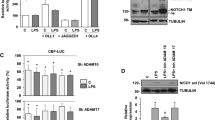
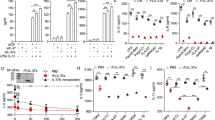
Abstract
The Toll-like receptor–interleukin 1 receptor signaling (TLR–IL-1R) receptor superfamily is important in differentially recognizing pathogen products and eliciting appropriate immune responses. These receptors alter gene expression, mainly through the activation of nuclear factor-κB and activating protein 1. SIGIRR (single immunoglobulin IL-1R-related molecule), a member of this family that does not activate these factors, instead negatively modulates immune responses. Inflammation is enhanced in SIGIRR-deficient mice, as shown by their enhanced chemokine induction after IL-1 injection and reduced threshold for lethal endotoxin challenge. Cells from SIGIRR-deficient mice showed enhanced activation in response to either IL-1 or certain Toll ligands. Finally, biochemical analysis indicated that SIGIRR binds to the TLR–IL-1R signaling components in a ligand-dependent way. Our data show that SIGIRR functions as a biologically important modulator of TLR–IL-1R signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Dinarello, C.A. Interleukin-1. Cytokine Growth Factor Rev. 8, 253–265 (1997).
Neumann, D., Kollewe, C., Martin, M.U. & Boraschi, D. The membrane form of the type II IL-1 receptor accounts for inhibitory function. J. Immunol. 165, 3350–3357 (2000).
Akira, S. The role of IL-18 in innate immunity. Curr. Opin. Immunol. 12, 59–63 (2000).
Townsend, M.J., Fallon, P.G., Matthews, D.J., Jolin, H.E. & McKenzie, A.N. T1/ST2-deficient mice demonstrate the importance of T1/ST2 in developing primary T helper cell type 2 responses. J. Exp. Med. 191, 1069–1076 (2000).
Alexopoulou, L., Holt, A.C., Medzhitov, R. & Flavell, R.A. Recognition of double-stranded RNA and activation of NF-κB by Toll-like receptor 3. Nature 413, 732–738 (2001).
Takeuchi, O. et al. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 11, 443–451 (1999).
Ozinsky, A. et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between toll-like receptors. Proc. Natl. Acad. Sci. USA 97, 13766–13771 (2000).
Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740–745 (2000).
Hayashi, F. et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 410, 1099–1103 (2001).
Hoshino, K. et al. Cutting edge: Toll-like receptor 4 (TLR4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR4 as the Lps gene product. J. Immunol. 162, 3749–3752 (1999).
Dunne, A. & O'Neill, L.A. The interleukin-1 receptor/Toll-like receptor superfamily: signal transduction during inflammation and host defense. Sci. STKE 2003, re3 (2003).
Muzio, M., Ni, J., Feng, P. & Dixit, V.M. IRAK (Pelle) family member IRAK-2 and MyD88 as proximal mediators of IL-1 signaling. Science 278, 1612–1615 (1997).
Wesche, H., Henzel, W.J., Shillinglaw, W., Li, S. & Cao, Z. MyD88: an adapter that recruits IRAK to the IL-1 receptor complex. Immunity 7, 837–847 (1997).
Burns, K. et al. MyD88, an adapter protein involved in interleukin-1 signaling. J. Biol. Chem. 273, 12203–12209 (1998).
Cao, Z., Henzel, W.J. & Gao, X. IRAK: a kinase associated with the interleukin-1 receptor. Science 271, 1128–1131 (1996).
Li, S., Strelow, A., Fontana, E.J. & Wesche, H. IRAK-4: a novel member of the IRAK family with the properties of an IRAK-kinase. Proc. Natl. Acad. Sci. USA 99, 5567–5572 (2002).
Jiang, Z. et al. Pellino 1 is required for Interleukin-1 (IL-1)-mediated signaling through its interaction with the IL-1 receptor-associated kinase 4 (IRAK4)-IRAK-tumor necrosis factor receptor-associated factor 6 (TRAF6) complex. J. Biol. Chem. 278, 10952–10956 (2003).
Jiang, Z., Ninomiya-Tsuji, J., Qian, Y., Matsumoto, K. & Li, X. Interleukin-1 (IL-1) receptor-associated kinase-dependent IL-1-induced signaling complexes phosphorylate TAK1 and TAB2 at the plasma membrane and activate TAK1 in the cytosol. Mol. Cell. Biol. 22, 7158–7167 (2002).
Takaesu, G. et al. Interleukin-1 (IL-1) receptor-associated kinase leads to activation of TAK1 by inducing TAB2 translocation in the IL-1 signaling pathway. Mol. Cell. Biol. 21, 2475–2484 (2001).
Regnier, C.H. et al. Identification and characterization of an IκB kinase. Cell 90, 373–383 (1997).
Mercurio, F. et al. IKK-1 and IKK-2: cytokine-activated IκB kinases essential for NF-κB activation. Science 278, 860–866 (1997).
Jiang, Z. et al. Poly I:C-induced TLR3-mediated activation of NF-κB and MAP kinases is through an IRAK-independent pathway employing signaling components TLR3-TRAF6-TAK1-TAB2-PKR. J. Biol. Chem. 278, 16713–16719 (2003).
Yamamoto, M. et al. Cutting edge: a novel Toll/IL-1 receptor domain-containing adapter that preferentially activates the IFN-β promoter in the Toll-like receptor signaling. J. Immunol. 169, 6668–6672 (2002).
Imler, J.L. & Hoffmann, J.A. Toll signaling: the TIReless quest for specificity. Nat. Immunol. 4, 105–106 (2003).
Glauser, M.P. Pathophysiologic basis of sepsis: considerations for future strategies of intervention. Crit. Care. Med. 28, S4–8 (2000).
Ardizzone, S. & Porro, G.B. Inflammatory bowel disease: new insights into pathogenesis and treatment. J. Intern. Med. 252, 475–496 (2002).
Bingham, C.O., III. The pathogenesis of rheumatoid arthritis: pivotal cytokines involved in bone degradation and inflammation. J. Rheumatol. Suppl. 65, 3–9 (2002).
Kobayashi, K. et al. IRAK-M is a negative regulator of Toll-like receptor signaling. Cell 110, 191–202 (2002).
Burns, K. et al. Inhibition of interleukin 1 receptor/Toll-like receptor signaling through the alternatively spliced, short form of MyD88 is due to its failure to recruit IRAK-4. J. Exp. Med. 197, 263–268 (2003).
Janssens, S., Burns, K., Tschopp, J. & Beyaert, R. Regulation of interleukin-1- and lipopolysaccharide-induced NF-κB activation by alternative splicing of MyD88. Curr. Biol. 12, 467–471 (2002).
Thomassen, E., Renshaw, B.R. & Sims, J.E. Identification and characterization of SIGIRR, a molecule representing a novel subtype of the IL-1R superfamily. Cytokine 11, 389–399 (1999).
Cao, Z., Xiong, J., Takeuchi, M., Kurama, T. & Goeddel, D.V. TRAF6 is a signal transducer for interleukin-1. Nature 383, 443–446 (1996).
Saccani, S., Polentarutti, N., Penton-Rol, G., Sims, J.E. & Mantovani, A. Divergent effects of LPS on expression of IL-1 receptor family members in mononuclear phagocytes in vitro and in vivo. Cytokine 10, 773–780 (1998).
Penton-Rol, G. et al. Bacterial lipopolysaccharide causes rapid shedding, followed by inhibition of mRNA expression, of the IL-1 type II receptor, with concomitant up-regulation of the type I receptor and induction of incompletely spliced transcripts. J. Immunol. 162, 2931–2938 (1999).
Mosley, B. et al. The murine interleukin-4 receptor: molecular cloning and characterization of secreted and membrane bound forms. Cell 59, 335–348 (1989).
Mitchell, T. & Sugden, B. Stimulation of NF-κB-mediated transcription by mutant derivatives of the latent membrane protein of Epstein-Barr virus. J. Virol. 69, 2968–2976 (1995).
Acknowledgements
This work was supported by the National Institutes of Health (grant GM 600020 to X.L.) and by Amgen.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Wald, D., Qin, J., Zhao, Z. et al. SIGIRR, a negative regulator of Toll-like receptor–interleukin 1 receptor signaling. Nat Immunol 4, 920–927 (2003). https://doi.org/10.1038/ni968
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni968
This article is cited by
-
IL-33 promotes double negative T cell survival via the NF-κB pathway
Cell Death & Disease (2023)
-
Identification of biomarkers for glycaemic deterioration in type 2 diabetes
Nature Communications (2023)
-
SIGIRR deficiency contributes to CD4 T cell abnormalities by facilitating the IL1/C/EBPβ/TNF-α signaling axis in rheumatoid arthritis
Molecular Medicine (2022)
-
Amphiphile-CpG vaccination induces potent lymph node activation and COVID-19 immunity in mice and non-human primates
npj Vaccines (2022)
-
The unique role of innate lymphoid cells in cancer and the hepatic microenvironment
Cellular & Molecular Immunology (2022)