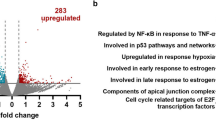
Abstract
Activating transcription factor-5 (ATF5) is highly expressed in malignant glioma and has a key role in promoting cell survival. Here we perform a genome-wide RNAi screen to identify transcriptional regulators of ATF5. Our results reveal an essential survival pathway in malignant glioma, whereby activation of a RAS–mitogen-activated protein kinase or phosphoinositide-3-kinase signaling cascade leads to induction of the transcription factor cAMP response element–binding protein-3–like-2 (CREB3L2), which directly activates ATF5 expression. ATF5, in turn, promotes survival by stimulating transcription of myeloid cell leukemia sequence-1 (MCL1), an antiapoptotic B cell leukemia-2 family member. Analysis of human malignant glioma samples indicates that ATF5 expression inversely correlates with disease prognosis. The RAF kinase inhibitor sorafenib suppresses ATF5 expression in glioma stem cells and inhibits malignant glioma growth in cell culture and mouse models. Our results demonstrate that ATF5 is essential in malignant glioma genesis and reveal that the ATF5-mediated survival pathway described here provides potential therapeutic targets for treatment of malignant glioma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hanahan, D. & Weinberg, R.A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Luo, J., Solimini, N.L. & Elledge, S.J. Principles of cancer therapy: oncogene and non-oncogene addiction. Cell 136, 823–837 (2009).
Persengiev, S.P. & Green, M.R. The role of ATF/CREB family members in cell growth, survival and apoptosis. Apoptosis 8, 225–228 (2003).
Greene, L.A., Lee, H.Y. & Angelastro, J.M. The transcription factor ATF5: role in neurodevelopment and neural tumors. J. Neurochem. 108, 11–22 (2009).
Monaco, S.E., Angelastro, J.M., Szabolcs, M. & Greene, L.A. The transcription factor ATF5 is widely expressed in carcinomas, and interference with its function selectively kills neoplastic, but not nontransformed, breast cell lines. Int. J. Cancer 120, 1883–1890 (2007).
Angelastro, J.M. et al. Selective destruction of glioblastoma cells by interference with the activity or expression of ATF5. Oncogene 25, 907–916 (2006).
Persengiev, S.P., Devireddy, L.R. & Green, M.R. Inhibition of apoptosis by ATFx: a novel role for a member of the ATF/CREB family of mammalian bZIP transcription factors. Genes Dev. 16, 1806–1814 (2002).
Collier, R.J. Diphtheria toxin: mode of action and structure. Bacteriol. Rev. 39, 54–85 (1975).
Silva, J.M. et al. Second-generation shRNA libraries covering the mouse and human genomes. Nat. Genet. 37, 1281–1288 (2005).
Sato, T. & Gotoh, N. The FRS2 family of docking/scaffolding adaptor proteins as therapeutic targets of cancer treatment. Expert Opin. Ther. Targets 13, 689–700 (2009).
da Fonseca, C.O. et al. Recent advances in the molecular genetics of malignant gliomas disclose targets for antitumor agent perillyl alcohol. Surg. Neurol. 65 Suppl. 1, S1:2–1:8; discussion S1:8–1:9 (2006).
Kumar, R., Gururaj, A.E. & Barnes, C.J. p21-activated kinases in cancer. Nat. Rev. Cancer 6, 459–471 (2006).
Kondo, S. et al. BBF2H7, a novel transmembrane bZIP transcription factor, is a new type of endoplasmic reticulum stress transducer. Mol. Cell. Biol. 27, 1716–1729 (2007).
Lonze, B.E. & Ginty, D.D. Function and regulation of CREB family transcription factors in the nervous system. Neuron 35, 605–623 (2002).
Mohammadi, M. et al. Crystal structure of an angiogenesis inhibitor bound to the FGF receptor tyrosine kinase domain. EMBO J. 17, 5896–5904 (1998).
Discafani, C.M. et al. Irreversible inhibition of epidermal growth factor receptor tyrosine kinase with in vivo activity by N-[4-[(3-bromophenyl)amino]-6-quinazolinyl]-2-butynamide (CL-387,785). Biochem. Pharmacol. 57, 917–925 (1999).
Hara, M. et al. Identification of Ras farnesyltransferase inhibitors by microbial screening. Proc. Natl. Acad. Sci. USA 90, 2281–2285 (1993).
Wilhelm, S. & Chien, D.S. BAY 43–9006: preclinical data. Curr. Pharm. Des. 8, 2255–2257 (2002).
Favata, M.F. et al. Identification of a novel inhibitor of mitogen-activated protein kinase kinase. J. Biol. Chem. 273, 18623–18632 (1998).
Ohori, M. et al. Identification of a selective ERK inhibitor and structural determination of the inhibitor-ERK2 complex. Biochem. Biophys. Res. Commun. 336, 357–363 (2005).
Vlahos, C.J., Matter, W.F., Hui, K.Y. & Brown, R.F. A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4-morpholinyl)-8-phenyl-4H–1-benzopyran-4-one (LY294002). J. Biol. Chem. 269, 5241–5248 (1994).
Cuenda, A. et al. SB 203580 is a specific inhibitor of a MAP kinase homologue which is stimulated by cellular stresses and interleukin-1. FEBS Lett. 364, 229–233 (1995).
Bonny, C., Oberson, A., Negri, S., Sauser, C. & Schorderet, D.F. Cell-permeable peptide inhibitors of JNK: novel blockers of beta-cell death. Diabetes 50, 77–82 (2001).
Yu, C. et al. The role of Mcl-1 downregulation in the proapoptotic activity of the multikinase inhibitor BAY 43–9006. Oncogene 24, 6861–6869 (2005).
Rhodes, D.R. et al. Oncomine 3.0: genes, pathways and networks in a collection of 18,000 cancer gene expression profiles. Neoplasia 9, 166–180 (2007).
Sun, L. et al. Neuronal and glioma-derived stem cell factor induces angiogenesis within the brain. Cancer Cell 9, 287–300 (2006).
Fischer, U. et al. A different view on DNA amplifications indicates frequent, highly complex, and stable amplicons on 12q13–21 in glioma. Mol. Cancer Res. 6, 576–584 (2008).
Aoki, H. et al. Phosphorylated Pak1 level in the cytoplasm correlates with shorter survival time in patients with glioblastoma. Clin. Cancer Res. 13, 6603–6609 (2007).
Pelloski, C.E. et al. Prognostic associations of activated mitogen-activated protein kinase and Akt pathways in glioblastoma. Clin. Cancer Res. 12, 3935–3941 (2006).
Parsons, D.W. et al. An integrated genomic analysis of human glioblastoma multiforme. Science 321, 1807–1812 (2008).
Rahmani, M. et al. The kinase inhibitor sorafenib induces cell death through a process involving induction of endoplasmic reticulum stress. Mol. Cell. Biol. 27, 5499–5513 (2007).
Rahmani, M., Davis, E.M., Bauer, C., Dent, P. & Grant, S. Apoptosis induced by the kinase inhibitor BAY 43–9006 in human leukemia cells involves down-regulation of Mcl-1 through inhibition of translation. J. Biol. Chem. 280, 35217–35227 (2005).
Alcantara Llaguno, S. et al. Malignant astrocytomas originate from neural stem/progenitor cells in a somatic tumor suppressor mouse model. Cancer Cell 15, 45–56 (2009).
Bao, S. et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444, 756–760 (2006).
Li, L. et al. EGFRvIII expression and PTEN loss synergistically induce chromosomal instability and glial tumors. Neuro-oncol. 11, 9–21 (2009).
Villano, J.L., Seery, T.E. & Bressler, L.R. Temozolomide in malignant gliomas: current use and future targets. Cancer Chemother. Pharmacol. 64, 647–655 (2009).
Weiss, W.A., Taylor, S.S. & Shokat, K.M. Recognizing and exploiting differences between RNAi and small-molecule inhibitors. Nat. Chem. Biol. 3, 739–744 (2007).
Sparidans, R.W. et al. Liquid chromatography–tandem mass spectrometric assay for sorafenib and sorafenib-glucuronide in mouse plasma and liver homogenate and identification of the glucuronide metabolite. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 877, 269–276 (2009).
Lagas, J.S. et al. Breast cancer resistance protein and P-glycoprotein limit sorafenib brain accumulation. Mol. Cancer Ther. 9, 319–326 (2010).
Hu, S. et al. Interaction of the multikinase inhibitors sorafenib and sunitinib with solute carriers and ATP-binding cassette transporters. Clin. Cancer Res. 15, 6062–6069 (2009).
Awada, A. et al. Phase I safety and pharmacokinetics of BAY 43–9006 administered for 21 days on/7 days off in patients with advanced, refractory solid tumours. Br. J. Cancer 92, 1855–1861 (2005).
Strumberg, D. et al. Phase I clinical and pharmacokinetic study of the Novel Raf kinase and vascular endothelial growth factor receptor inhibitor BAY 43–9006 in patients with advanced refractory solid tumors. J. Clin. Oncol. 23, 965–972 (2005).
Clark, J.W., Eder, J.P., Ryan, D., Lathia, C. & Lenz, H.J. Safety and pharmacokinetics of the dual action Raf kinase and vascular endothelial growth factor receptor inhibitor, BAY 43–9006, in patients with advanced, refractory solid tumors. Clin. Cancer Res. 11, 5472–5480 (2005).
Richly, H. et al. Results of a phase I trial of sorafenib (BAY 43–9006) in combination with doxorubicin in patients with refractory solid tumors. Ann. Oncol. 17, 866–873 (2006).
McCubrey, J.A. et al. Emerging Raf inhibitors. Expert Opin. Emerg. Drugs 14, 633–648 (2009).
Jung, S. et al. In vivo depletion of CD11c+ dendritic cells abrogates priming of CD8+ T cells by exogenous cell-associated antigens. Immunity 17, 211–220 (2002).
Gazin, C., Wajapeyee, N., Gobeil, S., Virbasius, C.M. & Green, M.R. An elaborate pathway required for Ras-mediated epigenetic silencing. Nature 449, 1073–1077 (2007).
Sheng, Z., Wang, S.Z. & Green, M.R. Transcription and signalling pathways involved in BCR-ABL–mediated misregulation of 24p3 and 24p3R. EMBO J. 28, 866–876 (2009).
Sen, A., Kallos, M.S. & Behie, L.A. New tissue dissociation protocol for scaled-up production of neural stem cells in suspension bioreactors. Tissue Eng. 10, 904–913 (2004).
Ihaka, R. & Gentleman, R. A language for data analysis and graphics. J. Comput. Graph. Statist. 5, 299–314 (1996).
Ruxton, G.D. The unequal variance t-test is an underused alternative to Student's t-test and the Mann-Whitney U test. Behav. Ecol. 17, 688–690 (2006).
Acknowledgements
We thank Y. Gillespie (University of Alabama at Birmingham) for GL261 cells, D. Bigner (Duke University) for D456MG cells and R. Lang (University of Cincinnati) for mCD11C-DTR-EFGP; the University of Massachusetts Medical School (UMMS) RNAi Core Facility for providing shRNAs; the UMMS Department of Pathology for providing human tissue sections and the UMMS Diabetes and Endocrinology Research Center core for performing the immunohistochemistry; K. Rock for suggesting the diphtheria toxin strategy; Y. Sun for critical advice on MRI; C. Gilbert for maintaining GS9-6 cells; S. Griggs for technical support; and S. Evans for editorial assistance. This work is supported by US National Institutes of Health grant RO1CA115817 to M.R.G. M.R.G. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
Z.S. and M.R.G. designed all experiments. Z.S. performed all experiments. Z.S. and M.R.G. prepared the manuscript. L.L. and A.H.R. assisted with intracranial injections. T.W.S., A.D. and R.P.M. helped analyze human malignant gliomas. L.J.Z. performed all statistical analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1–3 (PDF 2567 kb)
Rights and permissions
About this article
Cite this article
Sheng, Z., Li, L., Zhu, L. et al. A genome-wide RNA interference screen reveals an essential CREB3L2-ATF5-MCL1 survival pathway in malignant glioma with therapeutic implications. Nat Med 16, 671–677 (2010). https://doi.org/10.1038/nm.2158
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2158
This article is cited by
-
Insight into the mitochondrial unfolded protein response and cancer: opportunities and challenges
Cell & Bioscience (2022)
-
Connexin 43 confers chemoresistance through activating PI3K
Oncogenesis (2022)
-
Unveiling OASIS family as a key player in hypoxia–ischemia cases induced by cocaine using generative adversarial networks
Scientific Reports (2022)
-
The role of aquaporin 4 in brain tumors: implications for pathophysiology, diagnosis and therapy
Molecular Biology Reports (2022)
-
Mitochondria: intracellular sentinels of infections
Medical Microbiology and Immunology (2022)