Abstract
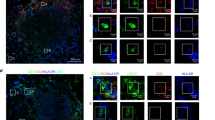
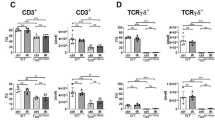
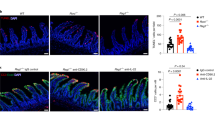
Localized abdominal surgery can lead to disruption of motility in the entire gastrointestinal tract (postoperative ileus). Intestinal macrophages produce mediators that paralyze myocytes, but it is unclear how the macrophages are activated, especially those in unmanipulated intestinal areas. Here we show that intestinal surgery activates intestinal CD103+CD11b+ dendritic cells (DCs) to produce interleukin-12 (IL-12). This promotes interferon-γ (IFN-γ) secretion by CCR9+ memory T helper type 1 (TH1) cells which activates the macrophages. IL-12 also caused some TH1 cells to migrate from surgically manipulated sites through the bloodstream to unmanipulated intestinal areas where they induced ileus. Preventing T cell migration with the drug FTY720 or inhibition of IL-12, T-bet (TH1-specific T box transcription factor) or IFN-γ prevented postoperative ileus. CCR9+ TH1 memory cells were detected in the venous blood of subjects 1 h after abdominal surgery. These findings indicate that postoperative ileus is a TH1 immune-mediated disease and identify potential targets for disease monitoring and therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kalff, J.C., Schraut, W.H., Billiar, T.R., Simmons, R.L. & Bauer, A.J. Role of inducible nitric oxide synthase in postoperative intestinal smooth muscle dysfunction in rodents. Gastroenterology 118, 316–327 (2000).
Bauer, A.J., Schwarz, N.T., Moore, B.A., Turler, A. & Kalff, J.C. Ileus in critical illness: mechanisms and management. Curr. Opin. Crit. Care 8, 152–157 (2002).
Schwarz, N.T. et al. Selective jejunal manipulation causes postoperative pan-enteric inflammation and dysmotility. Gastroenterology 126, 159–169 (2004).
Wehner, S. et al. Inhibition of macrophage function prevents intestinal inflammation and postoperative ileus in rodents. Gut 56, 176–185 (2007).
Bauer, A.J. Mentation on the immunological modulation of gastrointestinal motility. Neurogastroenterol. Motil. 20 (Suppl 1), 81–90 (2008).
Goldstein, J.L. et al. Inpatient economic burden of postoperative ileus associated with abdominal surgery in the United States. P&T Journal 32, 82–90 (2007).
Iyer, S., Saunders, W.B. & Stemkowski, S. Economic burden of postoperative ileus associated with colectomy in the United States. J. Manag. Care Pharm. 15, 485–494 (2009).
Barquist, E. et al. Neuronal pathways involved in abdominal surgery–induced gastric ileus in rats. Am. J. Physiol. 270, R888–R894 (1996).
Barada, K.A. et al. Localized colonic inflammation increases cytokine levels in distant small intestinal segments in the rat. Life Sci. 79, 2032–2042 (2006).
de Jonge, W.J. et al. Postoperative ileus is maintained by intestinal immune infiltrates that activate inhibitory neural pathways in mice. Gastroenterology 125, 1137–1147 (2003).
Türler, A. et al. Leukocyte-derived inducible nitric oxide synthase mediates murine postoperative ileus. Ann. Surg. 244, 220–229 (2006).
Wehner, S. et al. Inhibition of p38 mitogen–activated protein kinase pathway as prophylaxis of postoperative ileus in mice. Gastroenterology 136, 619–629 (2009).
Dardalhon, V., Korn, T., Kuchroo, V.K. & Anderson, A.C. Role of TH1 and TH17 cells in organ-specific autoimmunity. J. Autoimmun. 31, 252–256 (2008).
Szabo, S.J. et al. Distinct effects of T-bet in TH1 lineage commitment and IFN-γ production in CD4 and CD8 T cells. Science 295, 338–342 (2002).
Neurath, M.F., Finotto, S. & Glimcher, L.H. The role of TH1/TH2 polarization in mucosal immunity. Nat. Med. 8, 567–573 (2002).
Bouma, G. & Strober, W. The immunological and genetic basis of inflammatory bowel disease. Nat. Rev. Immunol. 3, 521–533 (2003).
Strober, W., Fuss, I. & Mannon, P. The fundamental basis of inflammatory bowel disease. J. Clin. Invest. 117, 514–521 (2007).
Gutcher, I. & Becher, B. APC-derived cytokines and T cell polarization in autoimmune inflammation. J. Clin. Invest. 117, 1119–1127 (2007).
Cua, D.J. et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421, 744–748 (2003).
Bettelli, E., Oukka, M. & Kuchroo, V.K. TH-17 cells in the circle of immunity and autoimmunity. Nat. Immunol. 8, 345–350 (2007).
Paust, H.J. et al. The IL-23/TH17 axis contributes to renal injury in experimental glomerulonephritis. J. Am. Soc. Nephrol. 20, 969–979 (2009).
Yen, D. et al. IL-23 is essential for T cell–mediated colitis and promotes inflammation via IL-17 and IL-6. J. Clin. Invest. 116, 1310–1316 (2006).
Mucida, D. et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260 (2007).
Leppkes, M. et al. RORγ-expressing TH17 cells induce murine chronic intestinal inflammation via redundant effects of IL-17A and IL-17F. Gastroenterology 136, 257–267 (2009).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Gebhardt, T. et al. Memory T cells in nonlymphoid tissue that provide enhanced local immunity during infection with herpes simplex virus. Nat. Immunol. 10, 524–530 (2009).
Wakim, L.M., Waithman, J., van Rooijen, N., Heath, W.R. & Carbone, F.R. Dendritic cell-induced memory T cell activation in nonlymphoid tissues. Science 319, 198–202 (2008).
Heymann, F. et al. Kidney dendritic cell activation is required for progression of renal disease in a mouse model of glomerular injury. J. Clin. Invest. 119, 1286–1297 (2009).
Coombes, J.L. & Powrie, F. Dendritic cells in intestinal immune regulation. Nat. Rev. Immunol. 8, 435–446 (2008).
Rescigno, M. & Di Sabatino, A. Dendritic cells in intestinal homeostasis and disease. J. Clin. Invest. 119, 2441–2450 (2009).
Varol, C., Zigmond, E. & Jung, S. Securing the immune tightrope: mononuclear phagocytes in the intestinal lamina propria. Nat. Rev. Immunol. 10, 415–426 (2010).
Kalff, J.C., Schraut, W.H., Simmons, R.L. & Bauer, A.J. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann. Surg. 228, 652–663 (1998).
Lahl, K. et al. Selective depletion of Foxp3+ regulatory T cells induces a scurfy-like disease. J. Exp. Med. 204, 57–63 (2007).
Schulz, E.G., Mariani, L., Radbruch, A. & Hofer, T. Sequential polarization and imprinting of type 1 T helper lymphocytes by interferon-γ and interleukin-12. Immunity 30, 673–683 (2009).
Mora, J.R. et al. Selective imprinting of gut-homing T cells by Peyer's patch dendritic cells. Nature 424, 88–93 (2003).
Cyster, J.G. Chemokines, sphingosine-1-phosphate and cell migration in secondary lymphoid organs. Annu. Rev. Immunol. 23, 127–159 (2005).
Tomita, T. et al. Colitogenic CD4+ effector-memory T cells actively recirculate in chronic colitic mice. Inflamm. Bowel Dis. 14, 1630–1640 (2008).
Kunisawa, J. et al. Sphingosine 1-phosphate dependence in the regulation of lymphocyte trafficking to the gut epithelium. J. Exp. Med. 204, 2335–2348 (2007).
Jung, S. et al. In vivo depletion of CD11c+ dendritic cells abrogates priming of CD8+ T cells by exogenous cell-associated antigens. Immunity 17, 211–220 (2002).
de Jonge, W.J. et al. Stimulation of the vagus nerve attenuates macrophage activation by activating the Jak2-STAT3 signaling pathway. Nat. Immunol. 6, 844–851 (2005).
Schaefer, N. et al. Role of resident macrophages in the immunologic response and smooth muscle dysfunction during acute allograft rejection after intestinal transplantation. Transpl. Int. 21, 778–791 (2008).
Boeckxstaens, G.E. & de Jonge, W.J. Neuroimmune mechanisms in postoperative ileus. Gut 58, 1300–1311 (2009).
Ahern, P.P., Izcue, A., Maloy, K.J. & Powrie, F. The interleukin-23 axis in intestinal inflammation. Immunol. Rev. 226, 147–159 (2008).
Weigmann, B. et al. The transcription factor NFATc2 controls IL-6–dependent T cell activation in experimental colitis. J. Exp. Med. 205, 2099–2110 (2008).
Ogawa, A., Andoh, A., Araki, Y., Bamba, T. & Fujiyama, Y. Neutralization of interleukin-17 aggravates dextran sulfate sodium-induced colitis in mice. Clin. Immunol. 110, 55–62 (2004).
Haak, S. et al. IL-17A and IL-17F do not contribute vitally to autoimmune neuro-inflammation in mice. J. Clin. Invest. 119, 61–69 (2009).
McGeachy, M.J. et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17–producing effector T helper cells in vivo. Nat. Immunol. 10, 314–324 (2009).
Hu, J. & August, A. Naive and innate memory phenotype CD4+ T cells have different requirements for active Itk for their development. J. Immunol. 180, 6544–6552 (2008).
Johansson-Lindbom, B. et al. Functional specialization of gut CD103+ dendritic cells in the regulation of tissue-selective T cell homing. J. Exp. Med. 202, 1063–1073 (2005).
Sun, C.M. et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 Treg cells via retinoic acid. J. Exp. Med. 204, 1775–1785 (2007).
Coombes, J.L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β and retinoic acid–dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Denning, T.L., Wang, Y.C., Patel, S.R., Williams, I.R. & Pulendran, B. Lamina propria macrophages and dendritic cells differentially induce regulatory and interleukin 17–producing T cell responses. Nat. Immunol. 8, 1086–1094 (2007).
Uematsu, S. et al. Regulation of humoral and cellular gut immunity by lamina propria dendritic cells expressing Toll-like receptor 5. Nat. Immunol. 9, 769–776 (2008).
Mannon, P.J. et al. Anti–interleukin-12 antibody for active Crohn's disease. N. Engl. J. Med. 351, 2069–2079 (2004).
Ludwig-Portugall, I., Hamilton-Williams, E.E., Gottschalk, C. & Kurts, C. Cutting edge: CD25+ regulatory T cells prevent expansion and induce apoptosis of B cells specific for tissue autoantigens. J. Immunol. 181, 4447–4451 (2008).
Kalff, J.C., Schwarz, N.T., Walgenbach, K.J., Schraut, W.H. & Bauer, A.J. Leukocytes of the intestinal muscularis: their phenotype and isolation. J. Leukoc. Biol. 63, 683–691 (1998).
Schaefer, N. et al. Resident macrophages are involved in intestinal transplantation-associated inflammation and motoric dysfunction of the graft muscularis. Am. J. Transplant. 7, 1062–1070 (2007).
Wehner, S. et al. Measurement of gastrointestinal and colonic transit in mice. Nat. Protoc. published online, doi:10.1038/protex.2010.1158 (28 November 2010).
Chirdo, F.G., Millington, O.R., Beacock-Sharp, H. & Mowat, A.M. Immunomodulatory dendritic cells in intestinal lamina propria. Eur. J. Immunol. 35, 1831–1840 (2005).
Semmling, V. et al. Alternative cross-priming through CCL17-CCR4–mediated attraction of CTLs toward NKT cell-licensed DCs. Nat. Immunol. 11, 313–320 (2010).
Acknowledgements
We thank M. Lysson for technical assistance, S. Hegenbarth for producing monoclonal antibodies and M. Forst for animal maintenance, and we acknowledge the support of the Central Animal Facilities and the Flow Cytometry Core Facility at the Institutes for Molecular Medicine and Experimental Immunology. D.R.E., S.W., J.C.K., A.K. and C.K. were supported by the Cooperative Clinical Research Center KFO115 of the Deutsche Forschungsgemeinschaft (Grants Ka1270/9-1/2 and Ku1063/4-1).
Author information
Authors and Affiliations
Contributions
D.R.E., A.K., M.S., L.F. and J.M. performed the experiments. D.R.E., A.K., J.C.K. and C.K. designed the study and wrote the manuscript. J.C.K. supervised the surgical part and C.K. supervised the immunological part of the study. S.W., A.H. and P.A.K. provided crucial ideas. T.S., B.S. and A.L. provided crucial reagents. All authors discussed and interpreted results.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–7, Supplementary Table 1 and Supplementary Methods (PDF 1899 kb)
Rights and permissions
About this article
Cite this article
Engel, D., Koscielny, A., Wehner, S. et al. T helper type 1 memory cells disseminate postoperative ileus over the entire intestinal tract. Nat Med 16, 1407–1413 (2010). https://doi.org/10.1038/nm.2255
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2255
This article is cited by
-
Effect of the gut microbiota and their metabolites on postoperative intestinal motility and its underlying mechanisms
Journal of Translational Medicine (2023)
-
Impact of intravenous dexmedetomidine on gastrointestinal function recovery after laparoscopic hysteromyomectomy: a randomized clinical trial
Scientific Reports (2022)
-
Steroid Receptor Coactivator-3 Is Required for Inhibition of the Intestinal Muscularis Inflammatory Response of Postoperative Ileus in Mice
Inflammation (2021)
-
Intestinal T lymphocyte homing is associated with gastric emptying and epithelial barrier function in critically ill: a prospective observational study
Critical Care (2017)
-
Electroacupuncture treatment partly promotes the recovery time of postoperative ileus by activating the vagus nerve but not regulating local inflammation
Scientific Reports (2017)