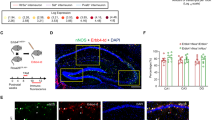
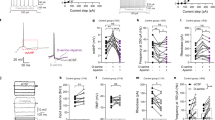
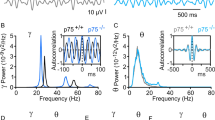
Abstract
Hypofunction of the N-methyl D-aspartate subtype of glutamate receptor (NMDAR) is hypothesized to be a mechanism underlying cognitive dysfunction in individuals with schizophrenia. For the schizophrenia-linked genes NRG1 and ERBB4, NMDAR hypofunction is thus considered a key detrimental consequence of the excessive NRG1-ErbB4 signaling found in people with schizophrenia. However, we show here that neuregulin 1β–ErbB4 (NRG1β-ErbB4) signaling does not cause general hypofunction of NMDARs. Rather, we find that, in the hippocampus and prefrontal cortex, NRG1β-ErbB4 signaling suppresses the enhancement of synaptic NMDAR currents by the nonreceptor tyrosine kinase Src. NRG1β-ErbB4 signaling prevented induction of long-term potentiation at hippocampal Schaffer collateral–CA1 synapses and suppressed Src-dependent enhancement of NMDAR responses during theta-burst stimulation. Moreover, NRG1β-ErbB4 signaling prevented theta burst–induced phosphorylation of GluN2B by inhibiting Src kinase activity. We propose that NRG1-ErbB4 signaling participates in cognitive dysfunction in schizophrenia by aberrantly suppressing Src-mediated enhancement of synaptic NMDAR function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Dingledine, R., Borges, K., Bowie, D. & Traynelis, S.F. The glutamate receptor ion channels. Pharmacol. Rev. 51, 7–61 (1999).
Paoletti, P. & Neyton, J. NMDA receptor subunits: function and pharmacology. Curr. Opin. Pharmacol. 7, 39–47 (2007).
Grant, S.G. & Blackstock, W.P. Proteomics in neuroscience: from protein to network. J. Neurosci. 21, 8315–8318 (2001).
Liu, X.J. et al. Treatment of inflammatory and neuropathic pain by uncoupling Src from the NMDA receptor complex. Nat. Med. 14, 1325–1332 (2008).
Lu, Y.M., Roder, J.C., Davidow, J. & Salter, M.W. Src activation in the induction of long-term potentiation in CA1 hippocampal neurons. Science 279, 1363–1367 (1998).
MacDonald, J.F., Kotecha, S.A., Lu, W.Y. & Jackson, M.F. Convergence of PKC-dependent kinase signal cascades on NMDA receptors. Curr. Drug Targets 2, 299–312 (2001).
Salter, M.W. & Kalia, L.V. Src kinases: a hub for NMDA receptor regulation. Nat. Rev. Neurosci. 5, 317–328 (2004).
Coyle, J.T., Tsai, G. & Goff, D. Converging evidence of NMDA receptor hypofunction in the pathophysiology of schizophrenia. Ann. NY Acad. Sci. 1003, 318–327 (2003).
Pilowsky, L.S. et al. First in vivo evidence of an NMDA receptor deficit in medication-free schizophrenic patients. Mol. Psychiatry 11, 118–119 (2006).
Balu, D.T. & Coyle, J.T. Neuroplasticity signaling pathways linked to the pathophysiology of schizophrenia. Neurosci. Biobehav. Rev. 35, 848–870 (2011).
Lindsley, C.W. et al. Progress towards validating the NMDA receptor hypofunction hypothesis of schizophrenia. Curr. Top. Med. Chem. 6, 771–785 (2006).
Hahn, C.G. et al. Altered neuregulin 1-erbB4 signaling contributes to NMDA receptor hypofunction in schizophrenia. Nat. Med. 12, 824–828 (2006).
Yu, X.M., Askalan, R., Keil, G.J. & Salter, M.W. NMDA channel regulation by channel-associated protein tyrosine kinase Src. Science 275, 674–678 (1997).
Stefansson, H. et al. Neuregulin 1 and susceptibility to schizophrenia. Am. J. Hum. Genet. 71, 877–892 (2002).
Yang, J.Z. et al. Association study of neuregulin 1 gene with schizophrenia. Mol. Psychiatry 8, 706–709 (2003).
Hashimoto, R. et al. Expression analysis of neuregulin-1 in the dorsolateral prefrontal cortex in schizophrenia. Mol. Psychiatry 9, 299–307 (2004).
Harrison, P.J. & Law, A.J. Neuregulin 1 and schizophrenia: genetics, gene expression, and neurobiology. Biol. Psychiatry 60, 132–140 (2006).
Nicodemus, K.K. et al. Further evidence for association between ErbB4 and schizophrenia and influence on cognitive intermediate phenotypes in healthy controls. Mol. Psychiatry 11, 1062–1065 (2006).
Norton, N. et al. Evidence that interaction between neuregulin 1 and its receptor erbB4 increases susceptibility to schizophrenia. Am. J. Med. Genet. B. Neuropsychiatr. Genet. 141B, 96–101 (2006).
Silberberg, G., Darvasi, A., Pinkas-Kramarski, R. & Navon, R. The involvement of ErbB4 with schizophrenia: association and expression studies. Am. J. Med. Genet. B. Neuropsychiatr. Genet. 141B, 142–148 (2006).
Law, A.J., Kleinman, J.E., Weinberger, D.R. & Weickert, C.S. Disease-associated intronic variants in the ErbB4 gene are related to altered ErbB4 splice-variant expression in the brain in schizophrenia. Hum. Mol. Genet. 16, 129–141 (2007).
Tan, W. et al. Molecular cloning of a brain-specific, developmentally regulated neuregulin 1 (NRG1) isoform and identification of a functional promoter variant associated with schizophrenia. J. Biol. Chem. 282, 24343–24351 (2007).
Colantuoni, C. et al. Age-related changes in the expression of schizophrenia susceptibility genes in the human prefrontal cortex. Brain Struct. Funct. 213, 255–271 (2008).
Nicodemus, K.K. et al. A 5′ promoter region SNP in NRG1 is associated with schizophrenia risk and type III isoform expression. Mol. Psychiatry 14, 741–743 (2009).
Chen, Y.J. et al. Type III neuregulin-1 is required for normal sensorimotor gating, memory-related behaviors, and corticostriatal circuit components. J. Neurosci. 28, 6872–6883 (2008).
O'Tuathaigh, C.M. et al. Disruption to social dyadic interactions but not emotional/anxiety-related behaviour in mice with heterozygous 'knockout' of the schizophrenia risk gene neuregulin-1. Prog. Neuropsychopharmacol. Biol. Psychiatry 32, 462–466 (2008).
Duffy, L., Cappas, E., Lai, D., Boucher, A.A. & Karl, T. Cognition in transmembrane domain neuregulin 1 mutant mice. Neuroscience 170, 800–807 (2010).
Deakin, I.H. et al. Behavioural characterization of neuregulin 1 type I overexpressing transgenic mice. Neuroreport 20, 1523–1528 (2009).
Kato, T. et al. Phenotypic characterization of transgenic mice overexpressing neuregulin-1. PLoS ONE 5, e14185 (2010).
Chong, V.Z. et al. Elevated neuregulin-1 and ErbB4 protein in the prefrontal cortex of schizophrenic patients. Schizophr. Res. 100, 270–280 (2008).
Law, A.J. et al. Neuregulin 1 transcripts are differentially expressed in schizophrenia and regulated by 5′ SNPs associated with the disease. Proc. Natl. Acad. Sci. USA 103, 6747–6752 (2006).
Huang, Y.Z. et al. Regulation of neuregulin signaling by PSD-95 interacting with ErbB4 at CNS synapses. Neuron 26, 443–455 (2000).
Ma, L. et al. Ligand-dependent recruitment of the ErbB4 signaling complex into neuronal lipid rafts. J. Neurosci. 23, 3164–3175 (2003).
Bjarnadottir, M. et al. Neuregulin1 (NRG1) signaling through Fyn modulates NMDA receptor phosphorylation: differential synaptic function in Nrg1+/− knock-outs compared with wild-type mice. J. Neurosci. 27, 4519–4529 (2007).
Kwon, O.B., Longart, M., Vullhorst, D., Hoffman, D.A. & Buonanno, A. Neuregulin-1 reverses long-term potentiation at CA1 hippocampal synapses. J. Neurosci. 25, 9378–9383 (2005).
Pitcher, G.M., Beggs, S., Woo, R.S., Mei, L. & Salter, M.W. ErbB4 is a suppressor of long-term potentiation in the adult hippocampus. Neuroreport 19, 139–143 (2008).
Iyengar, S.S. & Mott, D.D. Neuregulin blocks synaptic strengthening after epileptiform activity in the rat hippocampus. Brain Res. 1208, 67–73 (2008).
Huang, Y. et al. CAKβ/Pyk2 kinase is a signaling link for induction of long-term potentiation in CA1 hippocampus. Neuron 29, 485–496 (2001).
Pelkey, K.A. et al. Tyrosine phosphatase STEP is a tonic brake on induction of long-term potentiation. Neuron 34, 127–138 (2002).
Gao, X.M. et al. Ionotropic glutamate receptors and expression of N-methyl-D-aspartate receptor subunits in subregions of human hippocampus: effects of schizophrenia. Am. J. Psychiatry 157, 1141–1149 (2000).
Siekmeier, P.J., Hasselmo, M.E., Howard, M.W. & Coyle, J. Modeling of context-dependent retrieval in hippocampal region CA1: implications for cognitive function in schizophrenia. Schizophr. Res. 89, 177–190 (2007).
Zierhut, K. et al. The role of hippocampus dysfunction in deficient memory encoding and positive symptoms in schizophrenia. Psychiatry Res. 183, 187–194 (2010).
Benetti, S. et al. Functional integration between the posterior hippocampus and prefrontal cortex is impaired in both first episode schizophrenia and the at risk mental state. Brain 132, 2426–2436 (2009).
Tidcombe, H. et al. Neural and mammary gland defects in ErbB4 knockout mice genetically rescued from embryonic lethality. Proc. Natl. Acad. Sci. USA 100, 8281–8286 (2003).
Fukazawa, R. et al. Neuregulin-1 protects ventricular myocytes from anthracycline-induced apoptosis via erbB4-dependent activation of PI3-kinase/Akt. J. Mol. Cell. Cardiol. 35, 1473–1479 (2003).
Woo, R.S. et al. Neuregulin-1 enhances depolarization-induced GABA release. Neuron 54, 599–610 (2007).
Macdonald, D.S. et al. Modulation of NMDA receptors by pituitary adenylate cyclase activating peptide in CA1 neurons requires Gαq, protein kinase C and activation of Src. J. Neurosci. 25, 11374–11384 (2005).
Xu, J. et al. Control of excitatory synaptic transmission by C-terminal Src kinase. J. Biol. Chem. 283, 17503–17514 (2008).
Gu, Z., Jiang, Q., Fu, A.K., Ip, N.Y. & Yan, Z. Regulation of NMDA receptors by neuregulin signaling in prefrontal cortex. J. Neurosci. 25, 4974–4984 (2005).
Eisenberg, D.P. & Berman, K.F. Executive function, neural circuitry and genetic mechanisms in schizophrenia. Neuropsychopharmacology 35, 258–277 (2010).
Merrill, M.A., Chen, Y., Strack, S. & Hell, J.W. Activity-driven postsynaptic translocation of CaMKII. Trends Pharmacol. Sci. 26, 645–653 (2005).
Rostas, J.A. et al. Enhanced tyrosine phosphorylation of the 2B subunit of the N-methyl-D-aspartate receptor in long-term potentiation. Proc. Natl. Acad. Sci. USA 93, 10452–10456 (1996).
Rosenblum, K., Dudai, Y. & Richter-Levin, G. Long-term potentiation increases tyrosine phosphorylation of the N-methyl-D-aspartate receptor subunit 2B in rat dentate gyrus in vivo. Proc. Natl. Acad. Sci. USA 93, 10457–10460 (1996).
Kim, H. et al. The c-Src tyrosine kinase associates with the catalytic domain of ErbB-2: implications for ErbB-2 mediated signaling and transformation. Oncogene 24, 7599–7607 (2005).
Olayioye, M.A., Beuvink, I., Horsch, K., Daly, J.M. & Hynes, N.E. ErbB receptor–induced activation of stat transcription factors is mediated by Src tyrosine kinases. J. Biol. Chem. 274, 17209–17218 (1999).
Lisman, J. & Buzsaki, G. A neural coding scheme formed by the combined function of gamma and theta oscillations. Schizophr. Bull. 34, 974–980 (2008).
Uhlhaas, P.J., Haenschel, C., Nikolic, D. & Singer, W. The role of oscillations and synchrony in cortical networks and their putative relevance for the pathophysiology of schizophrenia. Schizophr. Bull. 34, 927–943 (2008).
Lei, G. et al. Gain control of N-methyl-D-aspartate receptor activity by receptor-like protein tyrosine phosphatase alpha. EMBO J. 21, 2977–2989 (2002).
Kalia, L.V., Pitcher, G.M., Pelkey, K.A. & Salter, M.W. PSD-95 is a negative regulator of the tyrosine kinase Src in the NMDA receptor complex. EMBO J. 25, 4971–4982 (2006).
Eilam, R., Pinkas-Kramarski, R., Ratzkin, B.J., Segal, M. & Yarden, Y. Activity-dependent regulation of Neu differentiation factor/neuregulin expression in rat brain. Proc. Natl. Acad. Sci. USA 95, 1888–1893 (1998).
Acknowledgements
We thank J.F. MacDonald, P.A. Frankland, L.Y. Wang, M.F. Jackson and M.H. Pitcher for critical reading of the manuscript, and Genentech and L. Mei for providing NRG1β. Src knockout mice were obtained as a gift from B.F. Boyce, University of Rochester Medical Center. This study was supported by grants from the Canadian Institutes of Health Research (CIHR) to M.W.S. (MT-12682) and to E.K.L. (MOP-89825), and from the Deafness Research Foundation to K.T.Y. M.W.S. holds a Canada research chair (tier I) in neuroplasticity and pain, and is an International Research Scholar of the Howard Hughes Medical Institute. E.K.L. holds a Canada research chair (tier II) in developmental cortical physiology. G.M.P. was a CIHR postdoctoral fellow and was supported by a Merck Frosst Fellowship award. D.N. held a CIHR doctoral research award, L.V.K. was a CIHR MD-PhD student, and N.M.G. holds a CIHR Canada Graduate Scholarship doctoral award. We thank D. Wong, J. Hicks and S. Singhroy for technical support.
Author information
Authors and Affiliations
Contributions
G.M.P. designed the project, conducted the hippocampal experiments, analyzed the data and wrote the manuscript. L.V.K. and D.N. carried out and analyzed biochemical experiments. E.K.L. oversaw and analyzed the prefrontal cortex experiments. N.M.G. carried out and analyzed the prefrontal cortex experiments. K.T.Y. maintained, housed and provided ErbB4 knockout mice. All authors participated in revising the manuscript and agreed to the final version. M.W.S. conceived the study, analyzed data, supervised the overall project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Methods (PDF 7051 kb)
Rights and permissions
About this article
Cite this article
Pitcher, G., Kalia, L., Ng, D. et al. Schizophrenia susceptibility pathway neuregulin 1–ErbB4 suppresses Src upregulation of NMDA receptors. Nat Med 17, 470–478 (2011). https://doi.org/10.1038/nm.2315
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2315