Abstract
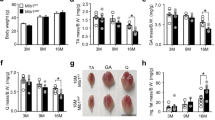
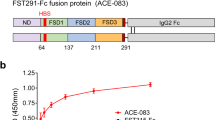
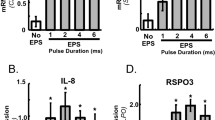
Limited neural input results in muscle weakness in neuromuscular disease because of a reduction in the density of muscle innervation, the rate of neuromuscular junction activation or the efficiency of synaptic transmission1. We developed a small-molecule fast-skeletal–troponin activator, CK-2017357, as a means to increase muscle strength by amplifying the response of muscle when neural input is otherwise diminished secondary to neuromuscular disease. Binding selectively to the fast-skeletal–troponin complex, CK-2017357 slows the rate of calcium release from troponin C and sensitizes muscle to calcium. As a consequence, the force-calcium relationship of muscle fibers shifts leftwards, as does the force-frequency relationship of a nerve-muscle pair, so that CK-2017357 increases the production of muscle force in situ at sub-maximal nerve stimulation rates. Notably, we show that sensitization of the fast-skeletal–troponin complex to calcium improves muscle force and grip strength immediately after administration of single doses of CK-2017357 in a model of the neuromuscular disease myasthenia gravis. Troponin activation may provide a new therapeutic approach to improve physical activity in diseases where neuromuscular function is compromised.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Research Group in Neuromuscular Diseases. Classification of the neuromuscular disorders. Lancet 291, 1020–1021 (1968).
Pollard, T. & Earnshaw, W. Cell Biology (Saunders/Elsevier, Philadelphia, 2008).
Freund, H.J. Motor unit and muscle activity in voluntary motor control. Physiol. Rev. 63, 387–436 (1983).
Jasmin, B.J. & Gardiner, P.F. Patterns of EMG activity of rat plantaris muscle during swimming and other locomotor activities. J. Appl. Physiol. 63, 713–718 (1987).
Sharma, K.R. & Miller, R.G. Electrical and mechanical properties of skeletal muscle underlying increased fatigue in patients with amyotrophic lateral sclerosis. Muscle Nerve 19, 1391–1400 (1996).
Kent-Braun, J.A. & Miller, R.G. Central fatigue during isometric exercise in amyotrophic lateral sclerosis. Muscle Nerve 23, 909–914 (2000).
Patzkó, A. & Shy, M.E. Update on Charcot-Marie-Tooth disease. Curr. Neurol. Neurosci. Rep. 11, 78–88 (2011).
Monani, U.R. Spinal muscular atrophy: a deficiency in a ubiquitous protein; a motor neuron-specific disease. Neuron 48, 885–896 (2005).
Drachman, D.B. Myasthenia gravis. N. Engl. J. Med. 330, 1797–1810 (1994).
Conti-Fine, B.M., Milani, M. & Kaminski, H.J. Myasthenia gravis: past, present, and future. J. Clin. Invest. 116, 2843–2854 (2006).
Solaro, R.J., Pang, D.C. & Briggs, F.N. The purification of cardiac myofibrils with Triton X-100. Biochim. Biophys. Acta 245, 259–262 (1971).
Young, O.A. & Davey, C.L. Electrophoretic analysis of proteins from single bovine muscle fibres. Biochem. J. 195, 317–327 (1981).
Herrmann, C., Sleep, J., Chaussepied, P., Travers, F. & Barman, T. A structural and kinetic study on myofibrils prevented from shortening by chemical cross-linking. Biochemistry 32, 7255–7263 (1993).
Bloemink, M.J., Adamek, N., Reggiani, C. & Geeves, M.A. Kinetic analysis of the slow skeletal myosin MHC-1 isoform from bovine masseter muscle. J. Mol. Biol. 373, 1184–1197 (2007).
Malik, F.I. et al. Cardiac myosin activation: a potential therapeutic approach for systolic heart failure. Science 331, 1439–1443 (2011).
Rosenfeld, S.S. & Taylor, E.W. Kinetic studies of calcium and magnesium binding to troponin C. J. Biol. Chem. 260, 242–251 (1985).
Poole, K.J. et al. A comparison of muscle thin filament models obtained from electron microscopy reconstructions and low-angle X-ray fibre diagrams from non-overlap muscle. J. Struct. Biol. 155, 273–284 (2006).
Brooks, S.V. & Faulkner, J.A. Contraction-induced injury: recovery of skeletal muscles in young and old mice. Am. J. Physiol. 258, C436–C442 (1990).
Lindstrom, J.M., Engel, A.G., Seybold, M.E., Lennon, V.A. & Lambert, E.H. Pathological mechanisms in experimental autoimmune myasthenia gravis. II. Passive transfer of experimental autoimmune myasthenia gravis in rats with anti-acetylcholine receptor antibodies. J. Exp. Med. 144, 739–753 (1976).
Jiménez-Moreno, R., Wang, Z.M., Gerring, R.C. & Delbono, O. Sarcoplasmic reticulum Ca2+ release declines in muscle fibers from aging mice. Biophys. J. 94, 3178–3188 (2008).
Erim, Z., Beg, M.F., Burke, D.T. & de Luca, C.J. Effects of aging on motor-unit control properties. J. Neurophysiol. 82, 2081–2091 (1999).
Ling, S.M., Conwit, R.A., Ferrucci, L. & Metter, E.J. Age-associated changes in motor unit physiology: observations from the Baltimore Longitudinal Study of Aging. Arch. Phys. Med. Rehabil. 90, 1237–1240 (2009).
Talmadge, R.J. & Roy, R.R. Electrophoretic separation of rat skeletal muscle myosin heavy-chain isoforms. J. Appl. Physiol. 75, 2337–2340 (1993).
Margossian, S.S. & Lowey, S. Preparation of myosin and its subfragments from rabbit skeletal muscle. Methods Enzymol. 85 Pt B, 55–71 (1982).
Pardee, J.D. & Spudich, J.A. Purification of muscle actin. Methods Enzymol. 85 Pt B, 164–181 (1982).
Potter, J.D. Preparation of troponin and its subunits. Methods Enzymol. 85 Pt B, 241–263 (1982).
Smillie, L.B. Preparation and identification of α- and β-tropomyosins. Methods Enzymol. 85 Pt B, 234–241 (1982).
Ebashi, S., Ebashi, F. & Kodama, A. Troponin as the Ca++-receptive protein in the contractile system. J. Biochem. 62, 137–138 (1967).
Spudich, J.A. & Watt, S. The regulation of rabbit skeletal muscle contraction. I. Biochemical studies of the interaction of the tropomyosin-troponin complex with actin and the proteolytic fragments of myosin. J. Biol. Chem. 246, 4866–4871 (1971).
Claflin, D.R. et al. Effects of high- and low-velocity resistance training on the contractile properties of skeletal muscle fibers from young and older humans. J. Appl. Physiol. 111, 1021–1030 (2011).
De La Cruz, E.M. & Ostap, E.M. Kinetic and equilibrium analysis of the myosin ATPase. Methods Enzymol. 455, 157–192 (2009).
Rosenfeld, S.S. & Taylor, E.W. Kinetic studies of calcium binding to regulatory complexes from skeletal muscle. J. Biol. Chem. 260, 252–261 (1985).
Acknowledgements
The project described was supported by Cytokinetics, Inc. and by award number 1RC3NS070670-01 (F.I.M.) from the National Institute of Neurological Disorders and Stroke. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Neurological Disorders and Stroke or the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
All authors contributed extensively to the work presented in this paper.
Corresponding author
Ethics declarations
Competing interests
A.J.R., J.J.H., A.C.H., A.R.M., R.K., L.D., G.G., K.H.L., D.M., W.F.B., M.M.C., D.C., S.E.C., M.G., R.H., Z.J., P.-P.L., H.R., K.G.S., J.S., V.V., D.J.M., B.P.M. and F.I.M. are past or present employees of Cytokinetics, Inc. and own stock or stock options in Cytokinetics, Inc. D.L.A. and D.R.C. received research support from Cytokinetics, Inc.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2 (PDF 92 kb)
Rights and permissions
About this article
Cite this article
Russell, A., Hartman, J., Hinken, A. et al. Activation of fast skeletal muscle troponin as a potential therapeutic approach for treating neuromuscular diseases. Nat Med 18, 452–455 (2012). https://doi.org/10.1038/nm.2618
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2618
This article is cited by
-
Intermolecular And Dynamic Investigation of The Mechanism of Action of Reldesemtiv on Fast Skeletal Muscle Troponin Complex Toward the Treatment of Impaired Muscle Function
The Protein Journal (2023)
-
Matrigel 3D bioprinting of contractile human skeletal muscle models recapitulating exercise and pharmacological responses
Communications Biology (2021)
-
Nebulin: big protein with big responsibilities
Journal of Muscle Research and Cell Motility (2020)
-
Effects of levosimendan on respiratory muscle function in patients weaning from mechanical ventilation
Intensive Care Medicine (2019)
-
Diaphragm abnormalities in heart failure and aging: mechanisms and integration of cardiovascular and respiratory pathophysiology
Heart Failure Reviews (2017)