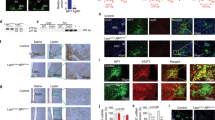
Abstract
Few effective measures exist to combat the worldwide obesity epidemic1, and the identification of potential therapeutic targets requires a deeper understanding of the mechanisms that control energy balance. Leptin, an adipocyte-derived hormone that signals the long-term status of bodily energy stores, acts through multiple types of leptin receptor long isoform (LepRb)-expressing neurons (called here LepRb neurons) in the brain to control feeding, energy expenditure and endocrine function2,3,4. The modest contributions to energy balance that are attributable to leptin action in many LepRb populations5,6,7,8,9 suggest that other previously unidentified hypothalamic LepRb neurons have key roles in energy balance. Here we examine the role of LepRb in neuronal nitric oxide synthase (NOS1)-expressing LebRb (LepRbNOS1) neurons that comprise approximately 20% of the total hypothalamic LepRb neurons. Nos1cre-mediated genetic ablation of LepRb (LeprNos1KO) in mice produces hyperphagic obesity, decreased energy expenditure and hyperglycemia approaching that seen in whole-body LepRb-null mice. In contrast, the endocrine functions in LeprNos1KO mice are only modestly affected by the genetic ablation of LepRb in these neurons. Thus, hypothalamic LepRbNOS1 neurons are a key site of action of the leptin-mediated control of systemic energy balance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
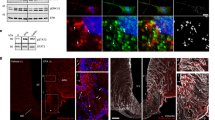
References
Bray, G.A. & Ryan, D.H. Drug treatment of obesity. Psychiatr. Clin. North Am. 34, 871–880 (2011).
Ahima, R.S., Saper, C.B., Flier, J.S. & Elmquist, J.K. Leptin regulation of neuroendocrine systems. Front. Neuroendocrinol. 21, 263–307 (2000).
Friedman, J.M. & Halaas, J.L. Leptin and the regulation of body weight in mammals. Nature 395, 763–770 (1998).
Farooqi, I.S. & O'Rahilly, S. Leptin: a pivotal regulator of human energy homeostasis. Am. J. Clin. Nutr. 89, 980S–984S (2009).
Balthasar, N. et al. Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron 42, 983–991 (2004).
Dhillon, H. et al. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 49, 191–203 (2006).
van de Wall, E. et al. Collective and individual functions of leptin receptor modulated neurons controlling metabolism and ingestion. Endocrinology 149, 1773–1785 (2008).
Hayes, M.R. et al. Endogenous leptin signaling in the caudal nucleus tractus solitarius and area postrema is required for energy balance regulation. Cell Metab. 11, 77–83 (2010).
Scott, M.M., Williams, K.W., Rossi, J., Lee, C.E. & Elmquist, J.K. Leptin receptor expression in hindbrain Glp-1 neurons regulates food intake and energy balance in mice. J. Clin. Invest. 121, 2413–2421 (2011).
Scott, M.M. et al. Leptin targets in the mouse brain. J. Comp. Neurol. 514, 518–532 (2009).
Myers, M.G. Jr., Münzberg, H., Leinninger, G.M. & Leshan, R.L. The geometry of leptin action in the brain: more complicated than a simple ARC. Cell Metab. 9, 117–123 (2009).
Patterson, C.M., Leshan, R.L., Jones, J.C. & Myers, M.G. Jr. Molecular mapping of mouse brain regions innervated by leptin receptor-expressing cells. Brain Res. 1378, 18–28 (2011).
Grill, H.J. Distributed neural control of energy balance: contributions from hindbrain and hypothalamus. Obesity (Silver Spring) 14 (suppl. 5), 216S–221S (2006).
Fulton, S. et al. Leptin regulation of the mesoaccumbens dopamine pathway. Neuron 51, 811–822 (2006).
Hommel, J.D. et al. Leptin receptor signaling in midbrain dopamine neurons regulates feeding. Neuron 51, 801–810 (2006).
Liu, J., Perez, S.M., Zhang, W., Lodge, D.J. & Lu, X.Y. Selective deletion of the leptin receptor in dopamine neurons produces anxiogenic-like behavior and increases dopaminergic activity in amygdala. Mol. Psychiatry 16, 1024–1038 (2011).
Leshan, R.L. et al. Ventral tegmental area leptin receptor neurons specifically project to and regulate cocaine- and amphetamine-regulated transcript neurons of the extended central amygdala. J. Neurosci. 30, 5713–5723 (2010).
Lam, D.D. et al. Leptin does not directly affect CNS serotonin neurons to influence appetite. Cell Metab. 13, 584–591 (2011).
Yadav, V.K. et al. A serotonin-dependent mechanism explains the leptin regulation of bone mass, appetite, and energy expenditure. Cell 138, 976–989 (2009).
Ring, L.E. & Zeltser, L.M. Disruption of hypothalamic leptin signaling in mice leads to early-onset obesity, but physiological adaptations in mature animals stabilize adiposity levels. J. Clin. Invest. 120, 2931–2941 (2010).
Leinninger, G.M. et al. Leptin action via neurotensin neurons controls orexin, the mesolimbic dopamine system and energy balance. Cell Metab. 14, 313–323 (2011).
Louis, G.W., Leinninger, G.M., Rhodes, C.J. & Myers, M.G. Jr. Direct innervation and modulation of orexin neurons by lateral hypothalamic LepRb neurons. J. Neurosci. 30, 11278–11287 (2010).
Leinninger, G.M. et al. Leptin acts via leptin receptor-expressing lateral hypothalamic neurons to modulate the mesolimbic dopamine system and suppress feeding. Cell Metab. 10, 89–98 (2009).
Donato, J. Jr., Frazao, R., Fukuda, M., Vianna, C.R. & Elias, C.F. Leptin induces phosphorylation of neuronal nitric oxide synthase in defined hypothalamic neurons. Endocrinology 151, 5415–5427 (2010).
Leshan, R.L. et al. Direct innervation of GnRH neurons by metabolic- and sexual odorant–sensing leptin receptor neurons in the hypothalamic ventral premammillary nucleus. J. Neurosci. 29, 3138–3147 (2009).
Elmquist, J.K., Elias, C.F. & Saper, C.B. From lesions to leptin: hypothalamic control of food intake and body weight. Neuron 22, 221–232 (1999).
Münzberg, H., Huo, L., Nillni, E.A., Hollenberg, A.N. & Bjørbaek, C. Role of signal transducer and activator of transcription 3 in regulation of hypothalamic proopiomelanocortin gene expression by leptin. Endocrinology 144, 2121–2131 (2003).
McMinn, J.E. et al. An allelic series for the leptin receptor gene generated by CRE and FLP recombinase. Mamm. Genome 15, 677–685 (2004).
Bjørbaek, C., Elmquist, J.K., Frantz, J.D., Shoelson, S.E. & Flier, J.S. Identification of SOCS-3 as a potential mediator of central leptin resistance. Mol. Cell 1, 619–625 (1998).
Vong, L. et al. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 71, 142–154 (2011).
Feil, R. & Kleppisch, T. NO/cGMP-dependent modulation of synaptic transmission. Handb. Exp. Pharmacol. 184, 529–560 (2008).
Huang, P.L. Mouse models of nitric oxide synthase deficiency. J. Am. Soc. Nephrol. 11, (suppl. 16), S120–S123 (2000).
Williams, K.W. et al. The acute effects of leptin require PI3K signaling in the hypothalamic ventral premammillary nucleus. J. Neurosci. 31, 13147–13156 (2011).
Donato, J. Jr. et al. Leptin's effect on puberty in mice is relayed by the ventral premammillary nucleus and does not require signaling in Kiss1 neurons. J. Clin. Invest. 121, 355–368 (2011).
Yokosuka, M., Prins, G.S. & Hayashi, S. Co-localization of androgen receptor and nitric oxide synthase in the ventral premammillary nucleus of the newborn rat: an immunohistochemical study. Brain Res. Dev. Brain Res. 99, 226–233 (1997).
Soliman, G.A. et al. A simple qPCR-based method to detect correct insertion of homologous targeting vectors in murine ES cells. Transgenic Res. 16, 665–670 (2007).
Münzberg, H. et al. Appropriate inhibition of orexigenic hypothalamic arcuate nucleus neurons independently of leptin receptor/STAT3 signaling. J. Neurosci. 27, 69–74 (2007).
Bates, S.H. et al. STAT3 signaling is required for leptin regulation of energy balance but not reproduction. Nature 421, 856–859 (2003).
Acknowledgements
We thank Amylin Pharmaceuticals for the generous gift of leptin; S. Chua (Albert Einstein College of Medicine), B. Lowell (Beth Israel-Deaconess Medical Center) and G. Barsh (Stanford University) for the gift of Leprflox/flox, Agrpcre and Pomccre mice, respectively; and members of the Myers lab for helpful discussions and technical support. Core support (animal phenotyping, Cell and Molecular Biology (CMB); clinical, Microscopy and Image Analysis Core (MIAC)) was provided by the Michigan Diabetes Research and Training Center and Nutrition and Obesity Research Center. This work was supported by the Marilyn H. Vincent Foundation and grants from the American Diabetes Association (M.G.M.), the American Heart Association (M.G.M. and R.L.L.) and the US National Institutes of Health (NIH) (DK057768 to M.G.M.). M.G.-Y. is supported by NIH grant T32GM008322; C.M.P. was supported by NIH grant T32HL007853.
Author information
Authors and Affiliations
Contributions
R.L.L., M.G.-Y., C.M.P. and I.E.G. carried out the experiments (with staff of the Core facilities and other technical assistance). R.L.L., M.G.-Y. and C.M.P. analyzed and prepared data for publication. M.G.M. guided the overall approach, in collaboration with R.L.L. and M.G.-Y. M.G.M., R.L.L. and M.G.-Y. cowrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Methods (PDF 14977 kb)
Rights and permissions
About this article
Cite this article
Leshan, R., Greenwald-Yarnell, M., Patterson, C. et al. Leptin action through hypothalamic nitric oxide synthase-1–expressing neurons controls energy balance. Nat Med 18, 820–823 (2012). https://doi.org/10.1038/nm.2724
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2724
This article is cited by
-
Metabolic control of puberty: 60 years in the footsteps of Kennedy and Mitra’s seminal work
Nature Reviews Endocrinology (2024)
-
Xiaoyaosan ameliorates depressive-like behavior and susceptibility to glucose intolerance in rat: involvement of LepR-STAT3/PI3K pathway in hypothalamic arcuate nucleus
BMC Complementary Medicine and Therapies (2023)
-
Somatostatin neurons in prefrontal cortex initiate sleep-preparatory behavior and sleep via the preoptic and lateral hypothalamus
Nature Neuroscience (2023)
-
Profound and redundant functions of arcuate neurons in obesity development
Nature Metabolism (2020)
-
A gut-to-brain signal of fluid osmolarity controls thirst satiation
Nature (2019)