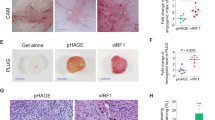
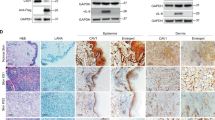
Abstract
Kaposi's sarcoma–associated herpesvirus (KSHV) is the causative agent of Kaposi's sarcoma1, a highly vascularized tumor originating from lymphatic endothelial cells, and of at least two different B cell malignancies2,3. A dimeric complex formed by the envelope glycoproteins H and L (gH-gL) is required for entry of herpesviruses into host cells4. We show that the ephrin receptor tyrosine kinase A2 (EphA2) is a cellular receptor for KSHV gH-gL. EphA2 co-precipitated with both gH-gL and KSHV virions. Infection of human epithelial cells with a GFP-expressing recombinant KSHV strain, as measured by FACS analysis, was increased upon overexpression of EphA2. Antibodies against EphA2 and siRNAs directed against EphA2 inhibited infection of endothelial cells. Pretreatment of KSHV with soluble EphA2 resulted in inhibition of KSHV infection by up to 90%. This marked reduction of KSHV infection was seen with all the different epithelial and endothelial cells used in this study. Similarly, pretreating epithelial or endothelial cells with the soluble EphA2 ligand ephrinA4 impaired KSHV infection. Deletion of the gene encoding EphA2 essentially abolished KSHV infection of mouse endothelial cells. Binding of gH-gL to EphA2 triggered EphA2 phosphorylation and endocytosis, a major pathway of KSHV entry5,6. Quantitative RT-PCR and in situ histochemistry revealed a close correlation between KSHV infection and EphA2 expression both in cultured cells derived from human Kaposi's sarcoma lesions or unaffected human lymphatic endothelium, and in situ in Kaposi's sarcoma specimens, respectively. Taken together, our results identify EphA2, a tyrosine kinase with known functions in neovascularization and oncogenesis, as an entry receptor for KSHV.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chang, Y. et al. Identification of herpesvirus-like DNA sequences in AIDS- associated Kaposi's sarcoma. Science 266, 1865–1869 (1994).
Cesarman, E., Chang, Y., Moore, P.S., Said, J.W. & Knowles, D.M. Kaposi's sarcoma–associated herpesvirus-like DNA sequences in AIDS-related body-cavity–based lymphomas. N. Engl. J. Med. 332, 1186–1191 (1995).
Soulier, J. et al. Kaposi's sarcoma–associated herpesvirus-like DNA sequences in multicentric Castleman's disease. Blood 86, 1276–1280 (1995).
Campadelli-Fiume, G. et al. The multipartite system that mediates entry of herpes simplex virus into the cell. Rev. Med. Virol. 17, 313–326 (2007).
Akula, S.M. et al. Kaposi's sarcoma–associated herpesvirus (human herpesvirus 8) infection of human fibroblast cells occurs through endocytosis. J. Virol. 77, 7978–7990 (2003).
Raghu, H., Sharma-Walia, N., Veettil, M.V., Sadagopan, S. & Chandran, B. Kaposi's sarcoma-associated herpesvirus utilizes an actin polymerization–dependent macropinocytic pathway to enter human dermal microvascular endothelial and human umbilical vein endothelial cells. J. Virol. 83, 4895–4911 (2009).
Chandran, B. Early events in Kaposi's sarcoma-associated herpesvirus infection of target cells. J. Virol. 84, 2188–2199 (2010).
Birkmann, A. et al. Cell surface heparan sulfate is a receptor for human herpesvirus 8 and interacts with envelope glycoprotein K8.1. J. Virol. 75, 11583–11593 (2001).
Wang, F.Z., Akula, S.M., Pramod, N.P., Zeng, L. & Chandran, B. Human herpesvirus 8 envelope glycoprotein K8.1A interaction with the target cells involves heparan sulfate. J. Virol. 75, 7517–7527 (2001).
Akula, S.M., Pramod, N.P., Wang, F.Z. & Chandran, B. Human herpesvirus 8 envelope-associated glycoprotein B interacts with heparan sulfate–like moieties. Virology 284, 235–249 (2001).
Hahn, A. et al. Kaposi's sarcoma-associated herpesvirus gH/gL: Glycoprotein export and interaction with cellular receptors. J. Virol. 83, 396–407 (2009).
Rappocciolo, G. et al. DC-SIGN is a receptor for human herpesvirus 8 on dendritic cells and macrophages. J. Immunol. 176, 1741–1749 (2006).
Sharma-Walia, N., Naranatt, P.P., Krishnan, H.H., Zeng, L. & Chandran, B. Kaposi's sarcoma-associated herpesvirus/human herpesvirus 8 envelope glycoprotein gB induces the integrin-dependent focal adhesion kinase-Src-phosphatidylinositol 3-kinase-Rho GTPase signal pathways and cytoskeletal rearrangements. J. Virol. 78, 4207–4223 (2004).
Naranatt, P.P., Akula, S.M., Zien, C.A., Krishnan, H.H. & Chandran, B. Kaposi's sarcoma–associated herpesvirus induces the phosphatidylinositol 3-kinase–PKC-ζ–MEK-ERK signaling pathway in target cells early during infection: implications for infectivity. J. Virol. 77, 1524–1539 (2003).
Kaleeba, J.A.R. & Berger, E.A. Kaposi's sarcoma-associated herpesvirus fusion-entry receptor: cystine transporter xCT. Science 311, 1921–1924 (2006).
Pertel, P.E. Human herpesvirus 8 glycoprotein B (gB), gH, and gL can mediate cell fusion. J. Virol. 76, 4390–4400 (2002).
Banfield, B.W., Leduc, Y., Esford, L., Schubert, K. & Tufaro, F. Sequential isolation of proteoglycan synthesis mutants by using herpes simplex virus as a selective agent: evidence for a proteoglycan-independent virus entry pathway. J. Virol. 69, 3290–3298 (1995).
Zhu, L., Puri, V. & Chandran, B. Characterization of human herpesvirus-8 K8.1A/B glycoproteins by monoclonal antibodies. Virology 262, 237–249 (1999).
Brantley-Sieders, D.M. et al. EphA2 receptor tyrosine kinase regulates endothelial cell migration and vascular assembly through phosphoinositide 3-kinase-mediated Rac1 GTPase activation. J. Cell Sci. 117, 2037–2049 (2004).
Vieira, J. & O'Hearn, P.M. Use of the red fluorescent protein as a marker of Kaposi's sarcoma–associated herpesvirus lytic gene expression. Virology 325, 225–240 (2004).
Stürzl, M. et al. Identification of interleukin-1 and platelet-derived growth factor-B as major mitogens for the spindle cells of Kaposi's sarcoma: a combined in vitro and in vivo analysis. Oncogene 10, 2007–2016 (1995).
Albini, A. et al. The beta-core fragment of human chorionic gonadotrophin inhibits growth of Kaposi's sarcoma–derived cells and a new immortalized Kaposi's sarcoma cell line. AIDS 11, 713–721 (1997).
Herndier, B.G. et al. Characterization of a human Kaposi's sarcoma cell line that induces angiogenic tumors in animals. AIDS 8, 575–581 (1994).
Kaleeba, J.A.R. & Berger, E.A. Broad target cell selectivity of Kaposi's sarcoma–associated herpesvirus glycoprotein-mediated cell fusion and virion entry. Virology 354, 7–14 (2006).
Lupberger, J. et al. EGFR and EphA2 are host factors for hepatitis C virus entry and possible targets for antiviral therapy. Nat. Med. 17, 589–595 (2011).
Vleck, S.E. et al. Structure−function analysis of varicella-zoster virus glycoprotein H identifies domain-specific roles for fusion and skin tropism. Proc. Natl. Acad. Sci. USA 108, 18412–18417 (2011).
Chesnokova, L.S., Nishimura, S.L. & Hutt-Fletcher, L.M. Fusion of epithelial cells by Epstein-Barr virus proteins is triggered by binding of viral glycoproteins gHgL to integrins αvβ6 or αvβ8 . Proc. Natl. Acad. Sci. USA 106, 20464–20469 (2009).
Chesnokova, L.S. & Hutt-Fletcher, L.M. Fusion of EBV with epithelial cells can be triggered by αvβ5 in addition to αvβ6 and αvβ8 and integrin binding triggers a conformational change in gHgL. J. Virol. 85, 13214–13223 (2011).
Vanarsdall, A.L., Chase, M.C. & Johnson, D.C. Human cytomegalovirus glycoprotein gO complexes with gH-gL, promoting interference with viral entry into human fibroblasts but not entry into epithelial cells. J. Virol. 85, 11638–11645 (2011).
Ryckman, B.J., Chase, M.C. & Johnson, D.C. HCMV gH/gL/UL128–131 interferes with virus entry into epithelial cells: evidence for cell type-specific receptors. Proc. Natl. Acad. Sci. USA 105, 14118–14123 (2008).
Chowdary, T.K. et al. Crystal structure of the conserved herpesvirus fusion regulator complex gH-gL. Nat. Struct. Mol. Biol. 17, 882–888 (2010).
Matsuura, H., Kirschner, A.N., Longnecker, R. & Jardetzky, T.S. Crystal structure of the Epstein-Barr virus (EBV) glycoprotein H/glycoprotein L (gH/gL) complex. Proc. Natl. Acad. Sci. USA 107, 22641–22646 (2010).
Walker-Daniels, J., Riese, D.J. & Kinch, M.S. c-Cbl–dependent EphA2 protein degradation is induced by ligand binding. Mol. Cancer Res. 1, 79–87 (2002).
Zhuang, G., Hunter, S., Hwang, Y. & Chen, J. Regulation of EphA2 receptor endocytosis by SHIP2 lipid phosphatase via phosphatidylinositol 3-kinase-dependent Rac1 activation. J. Biol. Chem. 282, 2683–2694 (2007).
Wang, H.W. et al. Kaposi sarcoma herpesvirus–induced cellular reprogramming contributes to the lymphatic endothelial gene expression in Kaposi sarcoma. Nat. Genet. 36, 687–693 (2004).
Ogawa, K. et al. The ephrin-A1 ligand and its receptor, EphA2, are expressed during tumor neovascularization. Oncogene 19, 6043–6052 (2000).
Scehnet, J.S. et al. The role of Ephs, ephrins and growth factors in Kaposi sarcoma and implications of ephrinB2 blockade. Blood 113, 254–263 (2009).
Cheng, N. et al. Blockade of EphA receptor tyrosine kinase activation inhibits vascular endothelial cell growth factor–induced angiogenesis. Mol. Cancer Res. 1, 2–11 (2002).
Brantley-Sieders, D.M. et al. EphA2 receptor tyrosine kinase regulates endothelial cell migration and vascular assembly through phosphoinositide 3-kinase-mediated Rac1 GTPase activation. J. Cell Sci. 117, 2037–2049 (2004).
Herndier, B.G. et al. Characterization of a human Kaposi's sarcoma cell line that induces angiogenic tumors in animals. AIDS 8, 575–581 (1994).
McAllister, R.M., Melnyk, J., Finkelstein, J.Z., Adams, E.C. Jr. & Gardner, M.B. Cultivation in vitro of cells derived from a human rhabdomyosarcoma. Cancer 24, 520–526 (1969).
Knowles, B.B., Howe, C.C. & Aden, D.P. Human hepatocellular carcinoma cell lines secrete the major plasma proteins and hepatitis B surface antigen. Science 209, 497–499 (1980).
Stürzl, M. et al. Identification of interleukin-1 and platelet-derived growth factor-B as major mitogens for the spindle cells of Kaposi's sarcoma: a combined in vitro and in vivo analysis. Oncogene 10, 2007–2016 (1995).
Vieira, J. & O'Hearn, P.M. Use of the red fluorescent protein as a marker of Kaposi's sarcoma–associated herpesvirus lytic gene expression. Virology 325, 225–240 (2004).
Myoung, J. & Ganem, D. Generation of a doxycycline-inducible KSHV producer cell line of endothelial origin: Maintenance of tight latency with efficient reactivation upon induction. J. Virol. Methods 174, 12–21 (2011).
Wickramasinghe, S.R., Kalbfuss, B., Zimmermann, A., Thom, V. & Reichl, U. Tangential flow microfiltration and ultrafiltration for human influenza A virus concentration and purification. Biotechnol. Bioeng. 92, 199–208 (2005).
Garrigues, H.J., Rubinchikova, Y.E., DiPersio, C.M. & Rose, T.M. Integrin αV3 Binds to the RGD motif of glycoprotein B of Kaposi's sarcoma–associated herpesvirus and functions as an RGD-dependent entry receptor. J. Virol. 82, 1570–1580 (2008).
Hahn, A. et al. Kaposi's sarcoma-associated herpesvirus gH-gL: glycoprotein export and interaction with cellular receptors. J. Virol. 83, 396–407 (2009).
Ensser, A., Pfinder, A., Muller-Fleckenstein, I. & Fleckenstein, B. The URNA genes of herpesvirus saimiri (strain C488) are dispensable for transformation of human T cells in vitro. J. Virol. 73, 10551–10555 (1999).
Glykofrydes, D. et al. Herpesvirus saimiri vFLIP provides an antiapoptotic function but is not essential for viral replication, transformation, or pathogenicity. J. Virol. 74, 11919–11927 (2000).
Birkmann, A. et al. Cell surface heparan sulfate is a receptor for human herpesvirus 8 and interacts with envelope glycoprotein K8.1. J. Virol. 75, 11583–11593 (2001).
König, S. et al. Sodium dodecyl sulfate versus acid-labile surfactant gel electrophoresis: comparative proteomic studies on rat retina and mouse brain. Electrophoresis 24, 751–756 (2003).
Schmidt, K., Wies, E. & Neipel, F. Kaposi's sarcoma-associated herpesvirus viral interferon regulatory factor 3 inhibits gamma interferon and major histocompatibility complex class II expression. J. Virol. 85, 4530–4537 (2011).
Naschberger, E. et al. Angiostatic immune reaction in colorectal carcinoma: impact on survival and perspectives for antiangiogenic therapy. Int. J. Cancer 123, 2120–2129 (2008).
Acknowledgements
This work was supported by the Akademie der Wissenschaften und der Literatur (Mainz), the European Community research project TargetHerpes, Deutsche Forschungsgemeinschaft center grant 643 and graduate school grant 1071 “viruses of the immune system.” M. Stürzl, E.N. and F.N. were supported by the interdisciplinary center for clinical research at the University Hospital of the University of Erlangen. M. Stürzl and E.N. were supported by the German Cancer Aid. We thank L. Wall for experimental assistance and R.C. Desrosiers for helpful discussions and critical reading of the manuscript. KSImm cells were kindly provided by A. Albini (Science and Technology Pole, IRCCS MultiMedica). 4G10 mouse hybridoma supernatant was kindly provided by B. Biesinger (Virologisches Institut, Universitätsklinikum Erlangen).
Author information
Authors and Affiliations
Contributions
A.S.H. and F.N. designed the study. A.S.H., J.K.K. and E.W. performed the key experiments. J.P.-I., K.S., M. Schmidt and A.H. performed real-time PCR experiments, cell culture and infection assays. S.K. performed mass spectrometry and analyzed the data. J.C., A.E., J.M. and N.H.B. contributed key reagents. E.N. and M. Stürzl helped with endothelial cell cultures and performed immunohistochemistry experiments. A.S.H. and F.N. wrote the manuscript. B.F. contributed expertise and helped write the paper.
Corresponding author
Ethics declarations
Competing interests
A. Hahn filed a patent to the United States Patent and Trademark Office involving the inhibition of EphA2 and KSHV infection with a small molecule.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1 (PDF 559 kb)
Rights and permissions
About this article
Cite this article
Hahn, A., Kaufmann, J., Wies, E. et al. The ephrin receptor tyrosine kinase A2 is a cellular receptor for Kaposi's sarcoma–associated herpesvirus. Nat Med 18, 961–966 (2012). https://doi.org/10.1038/nm.2805
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2805
This article is cited by
-
KSHV (HHV8) vaccine: promises and potential pitfalls for a new anti-cancer vaccine
npj Vaccines (2022)
-
The structural basis of herpesvirus entry
Nature Reviews Microbiology (2021)
-
Molecular basis of EphA2 recognition by gHgL from gammaherpesviruses
Nature Communications (2020)
-
Eph receptors: the bridge linking host and virus
Cellular and Molecular Life Sciences (2020)
-
Towards Better Understanding of KSHV Life Cycle: from Transcription and Posttranscriptional Regulations to Pathogenesis
Virologica Sinica (2019)