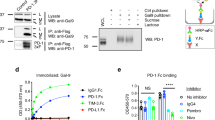
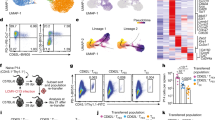
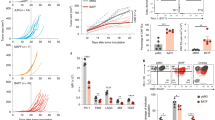
Abstract
T cell immunoglobulin and mucin domain–containing 3 (Tim-3) is an inhibitory receptor that is expressed on exhausted T cells during infection with HIV-1 and hepatitis C virus. By contrast, Tim-3 expression and function are defective in multiple human autoimmune diseases. However, the molecular mechanisms modulating Tim-3 function are not well understood. Here we show that human leukocyte antigen B (HLA-B)-associated transcript 3 (Bat3) binds to, and represses the function of, Tim-3. Bat3 protects T helper type 1 (TH1) cells from galectin-9–mediated cell death and promotes both proliferation and proinflammatory cytokine production. Bat3-deficient T cells have elevated expression of exhaustion-associated molecules such as Tim-3, Lag3, Prdm1 and Pbx3, and Bat3 knockdown in myelin-antigen–specific CD4+ T cells markedly inhibits the development of experimental autoimmune encephalomyelitis while promoting the expansion of a dysfunctional Tim-3hi, interferon-γ (IFN-γ)loCD4+ cell population. Furthermore, expression of Bat3 is reduced in exhausted Tim-3+ T cells from mouse tumors and HIV-1–infected individuals. These data indicate that Bat3 acts as an inhibitor of Tim-3–dependent exhaustion and cell death. Bat3 may thus represent a viable therapeutic target in autoimmune disorders, chronic infections and cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
21 August 2012
In the version of this article initially published online, the x axis in Figure 6c was incorrectly labeled. The error has been corrected for the print, PDF and HTML versions of this article.
References
Dunn, G.P. et al. Interferon-γ and cancer immunoediting. Immunol. Res. 32, 231–245 (2005).
Myrmel, H., Ulvestad, E. & Asjo, B. The hepatitis C virus enigma. APMIS 117, 427–439 (2009).
Romagnani, S. Th1/Th2 cells. Inflamm. Bowel Dis. 5, 285–294 (1999).
Moss, R.B. et al. Th1/Th2 cells in inflammatory disease states: therapeutic implications. Expert Opin. Biol. Ther. 4, 1887–1896 (2004).
Monney, L. et al. Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature 415, 536–541 (2002).
Zhu, C. et al. The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat. Immunol. 6, 1245–1252 (2005).
Sabatos, C.A. et al. Interaction of Tim-3 and Tim-3 ligand regulates T helper type 1 responses and induction of peripheral tolerance. Nat. Immunol. 4, 1102–1110 (2003).
Sánchez-Fueyo, A. et al. Tim-3 inhibits T helper type 1-mediated auto- and alloimmune responses and promotes immunological tolerance. Nat. Immunol. 4, 1093–1101 (2003).
Koguchi, K. et al. Dysregulated T cell expression of TIM3 in multiple sclerosis. J. Exp. Med. 203, 1413–1418 (2006).
Yang, L., Anderson, D.E., Kuchroo, J. & Hafler, D.A. Lack of TIM-3 immunoregulation in multiple sclerosis. J. Immunol. 180, 4409–4414 (2008).
Morimoto, K. et al. Dysregulated upregulation of T-cell immunoglobulin and mucin domain-3 on mucosal T helper 1 cells in patients with Crohn's disease. Scand. J. Gastroenterol. 46, 701–709 (2011).
Jones, R.B. et al. Tim-3 expression defines a novel population of dysfunctional T cells with highly elevated frequencies in progressive HIV-1 infection. J. Exp. Med. 205, 2763–2779 (2008).
Golden-Mason, L. et al. Negative immune regulator Tim-3 is overexpressed on T cells in hepatitis C virus infection and its blockade rescues dysfunctional CD4+ and CD8+ T cells. J. Virol. 83, 9122–9130 (2009).
Jin, H.T. et al. Cooperation of Tim-3 and PD-1 in CD8 T-cell exhaustion during chronic viral infection. Proc. Natl. Acad. Sci. USA 107, 14733–14738 (2010).
Takamura, S. et al. Premature terminal exhaustion of Friend virus-specific effector CD8+ T cells by rapid induction of multiple inhibitory receptors. J. Immunol. 184, 4696–4707 (2010).
Sakuishi, K. et al. Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J. Exp. Med. 207, 2187–2194 (2010).
Fourcade, J. et al. Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen-specific CD8+ T cell dysfunction in melanoma patients. J. Exp. Med. 207, 2175–2186 (2010).
Zhou, Q. et al. Co-expression of Tim-3 and PD-1 identifies a CD8+ T-cell exhaustion phenotype in mice with disseminated acute myelogenous leukemia. Blood 117, 4501–4510 (2011).
Elahi, S. et al. Protective HIV-specific CD8+ T cells evade Treg cell suppression. Nat. Med. 17, 989–995 (2011).
Desmots, F., Russell, H.R., Lee, Y., Boyd, K. & McKinnon, P.J. The reaper-binding protein scythe modulates apoptosis and proliferation during mammalian development. Mol. Cell. Biol. 25, 10329–10337 (2005).
Sasaki, T. et al. HLA-B–associated transcript 3 (Bat3)/Scythe is essential for p300-mediated acetylation of p53. Genes Dev. 21, 848–861 (2007).
Kuchroo, V.K. et al. Experimental allergic encephalomyelitis mediated by cloned T cells specific for a synthetic peptide of myelin proteolipid protein. Fine specificity and T cell receptor V β usage. J. Immunol. 148, 3776–3782 (1992).
van de Weyer, P.S. et al. A highly conserved tyrosine of Tim-3 is phosphorylated upon stimulation by its ligand galectin-9. Biochem. Biophys. Res. Commun. 351, 571–576 (2006).
Bettelli, E. et al. Myelin oligodendrocyte glycoprotein-specific T cell receptor transgenic mice develop spontaneous autoimmune optic neuritis. J. Exp. Med. 197, 1073–1081 (2003).
Tsunoda, I. & Fujinami, R.S. Two models for multiple sclerosis: experimental allergic encephalomyelitis and Theiler's murine encephalomyelitis virus. J. Neuropathol. Exp. Neurol. 55, 673–686 (1996).
Jäger, A., Dardalhon, V., Sobel, R.A., Bettelli, E. & Kuchroo, V.K. Th1, Th17, and Th9 effector cells induce experimental autoimmune encephalomyelitis with different pathological phenotypes. J. Immunol. 183, 7169–7177 (2009).
Wherry, E.J. T cell exhaustion. Nat. Immunol. 12, 492–499 (2011).
Blackburn, S.D. & Wherry, E.J. IL-10, T cell exhaustion and viral persistence. Trends Microbiol. 15, 143–146 (2007).
Priatel, J.J. et al. Chronic immunodeficiency in mice lacking RasGRP1 results in CD4 T cell immune activation and exhaustion. J. Immunol. 179, 2143–2152 (2007).
Shin, H. et al. A role for the transcriptional repressor Blimp-1 in CD8+ T cell exhaustion during chronic viral infection. Immunity 31, 309–320 (2009).
Wherry, E.J. et al. Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 27, 670–684 (2007).
Blackburn, S.D. et al. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat. Immunol. 10, 29–37 (2009).
Lee, J. et al. Phosphotyrosine-dependent coupling of Tim-3 to T-cell receptor signaling pathways. Mol. Cell. Biol. 31, 3963–3974 (2011).
Mustelin, T. & Tasken, K. Positive and negative regulation of T-cell activation through kinases and phosphatases. Biochem. J. 371, 15–27 (2003).
Nika, K. et al. Constitutively active Lck kinase in T cells drives antigen receptor signal transduction. Immunity 32, 766–777 (2010).
Oosterwegel, M.A., Greenwald, R.J., Mandelbrot, D.A., Lorsbach, R.B. & Sharpe, A.H. CTLA-4 and T cell activation. Curr. Opin. Immunol. 11, 294–300 (1999).
Okazaki, T., Maeda, A., Nishimura, H., Kurosaki, T. & Honjo, T. PD-1 immunoreceptor inhibits B cell receptor–mediated signaling by recruiting src homology 2-domain-containing tyrosine phosphatase 2 to phosphotyrosine. Proc. Natl. Acad. Sci. USA 98, 13866–13871 (2001).
Chemnitz, J.M., Parry, R.V., Nichols, K.E., June, C.H. & Riley, J.L. SHP-1 and SHP-2 associate with immunoreceptor tyrosine-based switch motif of programmed death 1 upon primary human T cell stimulation, but only receptor ligation prevents T cell activation. J. Immunol. 173, 945–954 (2004).
Yokosuka, T. et al. Programmed cell death 1 forms negative costimulatory microclusters that directly inhibit T cell receptor signaling by recruiting phosphatase SHP2. J. Exp. Med. 209, 1201–1217 (2012).
Thress, K., Song, J., Morimoto, R.I. & Kornbluth, S. Reversible inhibition of Hsp70 chaperone function by Scythe and Reaper. EMBO J. 20, 1033–1041 (2001).
Winnefeld, M. et al. Human SGT interacts with Bag-6/Bat-3/Scythe and cells with reduced levels of either protein display persistence of few misaligned chromosomes and mitotic arrest. Exp. Cell Res. 312, 2500–2514 (2006).
Desmots, F., Russell, H.R., Michel, D. & McKinnon, P.J. Scythe regulates apoptosis-inducing factor stability during endoplasmic reticulum stress-induced apoptosis. J. Biol. Chem. 283, 3264–3271 (2008).
Tsukahara, T. et al. Scythe/BAT3 regulates apoptotic cell death induced by papillomavirus binding factor in human osteosarcoma. Cancer Sci. 100, 47–53 (2009).
Kwak, J.H., Kim, S.I., Kim, J.K. & Choi, M.E. BAT3 interacts with transforming growth factor-β (TGF-β) receptors and enhances TGF-β1–induced type I collagen expression in mesangial cells. J. Biol. Chem. 283, 19816–19825 (2008).
Degli-Esposti, M.A. et al. Ancestral haplotypes reveal the role of the central MHC in the immunogenetics of IDDM. Immunogenetics 36, 345–356 (1992).
Hara, H. et al. The apoptotic protease-activating factor 1–mediated pathway of apoptosis is dispensable for negative selection of thymocytes. J. Immunol. 168, 2288–2295 (2002).
Acknowledgements
We thank D. Kozoriz, D. Lee, J. Sullivan and M. King for technical assistance; J. Zhang (Brigham and Women's Hospital, Boston) for reagents; B.H. Waksman for valuable guidance and discussion; and B. Walker for critical reading of the manuscript. M.R. was previously supported by Fellowships from the Multiple Sclerosis Society of Canada (MSSOC) and the Canadian Institutes of Health Research (CIHR) and is currently supported by the EMD Serono, Canada and an MS Research and Training Network Transitional Career Development Award from the MSSOC and the Multiple Sclerosis Scientific Research Foundation. K.S. holds a Fellowship from the Sankyo Foundation of Life Science. These studies were funded by grants from the US National Institutes of Health to V.K.K. (NS045937, AI073748 and NS038037) and S.X. (K01DK090105), the Ragon Institute of MGH, MIT and Harvard (V.K.K., C.Z. and M.M.A.), the American Cancer Society (A.C.A.) and the Harvard University Center for AIDS Research (M.M.A.; P30 AI060354).
Author information
Authors and Affiliations
Contributions
M.R. and C.Z. designed and performed experiments, and collected data. M.R. wrote the manuscript. K.S., S.X., J.K., A.C. and M.A. provided help in performing experiments. A.W. helped generate fetal liver chimeric mice. E.A.G. helped generate Bat3 antiserum. R.A.S. analyzed histopathological data. H.O. generated the Bag6−/− strain. P.J.M. and T.W.M. provided Bag6+/− mice and fetal liver chimeras, respectively. M.M.A. and A.C.A. designed and supervised experiments involving HIV samples and tumor mice, respectively. V.K.K. supervised the project and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14, Supplementary Table 1 and Supplementary Methods (PDF 1138 kb)
Rights and permissions
About this article
Cite this article
Rangachari, M., Zhu, C., Sakuishi, K. et al. Bat3 promotes T cell responses and autoimmunity by repressing Tim-3–mediated cell death and exhaustion. Nat Med 18, 1394–1400 (2012). https://doi.org/10.1038/nm.2871
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2871
This article is cited by
-
Incorporating the Molecular Mimicry of Environmental Antigens into the Causality of Autoimmune Hepatitis
Digestive Diseases and Sciences (2023)
-
Immune Checkpoints and targeted agents in relapse and graft-versus-host disease after hematopoietic stem cell transplantation
Molecular Biology Reports (2023)
-
Application and Effectiveness of Chinese Medicine in Regulating Immune Checkpoint Pathways
Chinese Journal of Integrative Medicine (2023)
-
TIM-3 as a promising target for cancer immunotherapy in a wide range of tumors
Cancer Immunology, Immunotherapy (2023)
-
Regulation of activated T cell survival in rheumatic autoimmune diseases
Nature Reviews Rheumatology (2022)