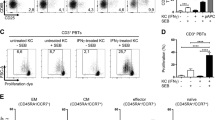
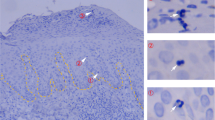
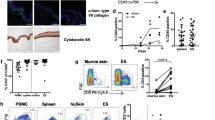
Abstract
The skin harbors a variety of resident leukocyte subsets that must be tightly regulated to maintain immune homeostasis. Hair follicles are unique structures in the skin that contribute to skin dendritic cell homeostasis through chemokine production. We demonstrate that CD4+ and CD8+ skin-resident memory T cells (TRM cells), which are responsible for long-term skin immunity, reside predominantly within the hair follicle epithelium of the unperturbed epidermis. TRM cell tropism for the epidermis and follicles is herein termed epidermotropism. Hair follicle expression of IL-15 was required for CD8+ TRM cells, and IL-7 for CD8+ and CD4+ TRM cells, to exert epidermotropism. A lack of either cytokine in the skin led to impaired hapten-induced contact hypersensitivity responses. In a model of cutaneous T cell lymphoma, epidermotropic CD4+ TRM lymphoma cell localization depended on the presence of hair follicle–derived IL-7. These findings implicate hair follicle–derived cytokines as regulators of malignant and non-malignant TRM cell tissue residence, and they suggest that the cytokines may be targeted therapeutically in inflammatory skin diseases and lymphoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cotsarelis, G., Sun, T.T. & Lavker, R.M. Label-retaining cells reside in the bulge area of pilosebaceous unit: implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell 61, 1329–1337 (1990).
Nishimura, E.K. et al. Dominant role of the niche in melanocyte stem-cell fate determination. Nature 416, 854–860 (2002).
Christoph, T. et al. The human hair follicle immune system: cellular composition and immune privilege. Br. J. Dermatol. 142, 862–873 (2000).
Nagao, K. et al. Stress-induced production of chemokines by hair follicles regulates the trafficking of dendritic cells in skin. Nat. Immunol. 13, 744–752 (2012).
Clark, R.A. et al. The vast majority of CLA+ T cells are resident in normal skin. J. Immunol. 176, 4431–4439 (2006).
Gebhardt, T. et al. Memory T cells in nonlymphoid tissue that provide enhanced local immunity during infection with herpes simplex virus. Nat. Immunol. 10, 524–530 (2009).
Gebhardt, T. et al. Different patterns of peripheral migration by memory CD4+ and CD8+ T cells. Nature 477, 216–219 (2011).
Jiang, X. et al. Skin infection generates non-migratory memory CD8+ T(RM) cells providing global skin immunity. Nature 483, 227–231 (2012).
Wakim, L.M., Waithman, J., van Rooijen, N., Heath, W.R. & Carbone, F.R. Dendritic cell-induced memory T cell activation in nonlymphoid tissues. Science 319, 198–202 (2008).
Mackay, L.K. et al. The developmental pathway for CD103+CD8+ tissue-resident memory T cells of skin. Nat. Immunol. 14, 1294–1301 (2013).
Shiow, L.R. et al. CD69 acts downstream of interferon-α/β to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature 440, 540–544 (2006).
Bromley, S.K., Yan, S., Tomura, M., Kanagawa, O. & Luster, A.D. Recirculating memory T cells are a unique subset of CD4+ T cells with a distinct phenotype and migratory pattern. J. Immunol. 190, 970–976 (2013).
Suffia, I., Reckling, S.K., Salay, G. & Belkaid, Y. A role for CD103 in the retention of CD4+CD25+ Treg and control of Leishmania major infection. J. Immunol. 174, 5444–5455 (2005).
Sanchez Rodriguez, R. et al. Memory regulatory T cells reside in human skin. J. Clin. Invest. 124, 1027–1036 (2014).
Shiohara, T. & Moriya, N. Epidermal T cells: their functional role and disease relevance for dermatologists. J. Invest. Dermatol. 109, 271–275 (1997).
Mizukawa, Y. et al. Direct evidence for interferon-γ production by effector-memory-type intraepidermal T cells residing at an effector site of immunopathology in fixed drug eruption. Am. J. Pathol. 161, 1337–1347 (2002).
Hwang, S.T., Janik, J.E., Jaffe, E.S. & Wilson, W.H. Mycosis fungoides and Sezary syndrome. Lancet 371, 945–957 (2008).
Campbell, J.J., Clark, R.A., Watanabe, R. & Kupper, T.S. Sezary syndrome and mycosis fungoides arise from distinct T-cell subsets: a biologic rationale for their distinct clinical behaviors. Blood 116, 767–771 (2010).
Clark, R.A. et al. Skin effector memory T cells do not recirculate and provide immune protection in alemtuzumab-treated CTCL patients. Sci. Transl. Med. 4, 117ra7 (2012).
Edelson, R.L. Cutaneous T cell lymphoma: mycosis fungoides, Sezary syndrome, and other variants. J. Am. Acad. Dermatol. 2, 89–106 (1980).
Seneschal, J., Clark, R.A., Gehad, A., Baecher-Allan, C.M. & Kupper, T.S. Human epidermal Langerhans cells maintain immune homeostasis in skin by activating skin resident regulatory T cells. Immunity 36, 873–884 (2012).
Sanchez Rodriguez, R. et al. Memory regulatory T cells reside in human skin. J. Clin. Invest. 124, 1027–1036 (2014).
Ariotti, S. et al. T cell memory. Skin-resident memory CD8+ T cells trigger a state of tissue-wide pathogen alert. Science 346, 101–105 (2014).
Lanzavecchia, A. & Sallusto, F. Understanding the generation and function of memory T cell subsets. Curr. Opin. Immunol. 17, 326–332 (2005).
van Leeuwen, E.M., Sprent, J. & Surh, C.D. Generation and maintenance of memory CD4+ T cells. Curr. Opin. Immunol. 21, 167–172 (2009).
Vasioukhin, V., Degenstein, L., Wise, B. & Fuchs, E. The magical touch: genome targeting in epidermal stem cells induced by tamoxifen application to mouse skin. Proc. Natl. Acad. Sci. USA 96, 8551–8556 (1999).
Liang, B. et al. Role of hepatocyte-derived IL-7 in maintenance of intrahepatic NKT cells and T cells and development of B cells in fetal liver. J. Immunol. 189, 4444–4450 (2012).
Dorfman, D.M. & Shahsafaei, A. CD69 expression correlates with expression of other markers of Th1 T cell differentiation in peripheral T cell lymphomas. Hum. Pathol. 33, 330–334 (2002).
Yamanaka, K. et al. Skin-derived interleukin-7 contributes to the proliferation of lymphocytes in cutaneous T-cell lymphoma. Blood 107, 2440–2445 (2006).
Espinet, B. & Salgado, R. Mycosis fungoides and Sezary syndrome. Methods Mol. Biol. 973, 175–188 (2013).
Kanavaros, P. et al. Mycosis fungoides: expression of C-myc p62 p53, bcl-2 and PCNA proteins and absence of association with Epstein-Barr virus. Pathol. Res. Pract. 190, 767–774 (1994).
Kamijo, T., Bodner, S., van de Kamp, E., Randle, D.H. & Sherr, C.J. Tumor spectrum in ARF-deficient mice. Cancer Res. 59, 2217–2222 (1999).
Serrano, M. et al. Role of the INK4a locus in tumor suppression and cell mortality. Cell 85, 27–37 (1996).
Sugihara, E. et al. Ink4a and Arf are crucial factors in the determination of the cell of origin and the therapeutic sensitivity of Myc-induced mouse lymphoid tumor. Oncogene 31, 2849–2861 (2012).
Tarutani, M. et al. Tissue-specific knockout of the mouse Pig-a gene reveals important roles for GPI-anchored proteins in skin development. Proc. Natl. Acad. Sci. USA 94, 7400–7405 (1997).
Kaplan, D.H., Jenison, M.C., Saeland, S., Shlomchik, W.D. & Shlomchik, M.J. Epidermal Langerhans cell-deficient mice develop enhanced contact hypersensitivity. Immunity 23, 611–620 (2005).
Bennett, C.L. et al. Inducible ablation of mouse Langerhans cells diminishes but fails to abrogate contact hypersensitivity. J. Cell Biol. 169, 569–576 (2005).
Livak, K.J. & Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-delta delta C(T)) method. Methods 25, 402–408 (2001).
Li, M. et al. Skin abnormalities generated by temporally controlled RXRα mutations in mouse epidermis. Nature 407, 633–636 (2000).
Ouchi, T. et al. Langerhans cell antigen capture through tight junctions confers pre-emptive immunity in experimental staphylococcal scalded skin syndrome. J. Exp. Med. 208, 2607–2613 (2011).
Fyhrquist, N., Wolff, H., Lauerma, A. & Alenius, H. CD8+ T cell migration to the skin requires CD4+ help in a murine model of contact hypersensitivity. PLoS ONE 7, e41038 (2012).
Takahashi, H. et al. Desmoglein 3-specific CD4+ T cells induce pemphigus vulgaris and interface dermatitis in mice. J. Clin. Invest. 121, 3677–3688 (2011).
Morita, S., Kojima, T. & Kitamura, T. Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther. 7, 1063–1066 (2000).
Sperling, L., Cowper, S.E. & Knopp, E.A. Evaluating and describing transverse (horizontal) sections. In An Atlas of Hair Pathology with Clinical Correlations 17–25 (CRC Press, 2012).
Acknowledgements
We thank M. Ohyama for helpful discussion on human hair follicles; N. Sakai, K. Eguchi and S. Sato (Keio University School of Medicine) for their technical assistance; H. Kong (National Institutes of Health) for their discussions on human CTCL; Y. Madokoro (Keio University School of Medicine) for human CTCL immunohistochemical staining; T. Kitamura (University of Tokyo) for providing the retroviral vector pMXs-IG and Plat-E cells; J. Takeda (Osaka University) for providing K5-Cre mice; D.H. Kaplan (University of Minnesota) for providing Langerin-DTA mice; and B.E. Clausen (Johannes Gutenberg University of Mainz) for providing Langerin-DTR mice. This work was supported by the Japan Society for the Promotion of Science, The Kanae Foundation for the Promotion of Medical Science, the Japanese Society for Investigative Dermatology's (JSID's) Fellowship Shiseido Award and the NIH NCI Intramural Research Programs.
Author information
Authors and Affiliations
Contributions
T.A. and K.N. conceived of and designed all experiments. Experiments were performed by T.A. with the assistance of T.K.; E.S. and H.S. provided Cdkn2a−/− mice and assisted with retroviral transduction; T.Y. assisted with immunohistochemical staining; K.I. provided Il7-floxed mice; S.P. provided human CTCL samples; M.A. discussed the data and provided administrative support; K.N. guided the project; and T.A. and K.N. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 & Supplementary Table 1 (PDF 4996 kb)
Rights and permissions
About this article
Cite this article
Adachi, T., Kobayashi, T., Sugihara, E. et al. Hair follicle–derived IL-7 and IL-15 mediate skin-resident memory T cell homeostasis and lymphoma. Nat Med 21, 1272–1279 (2015). https://doi.org/10.1038/nm.3962
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3962
This article is cited by
-
3D sheep rumen epithelial structures driven from single cells in vitro
Veterinary Research (2023)
-
IKK2/NFkB signaling controls lung resident CD8+ T cell memory during influenza infection
Nature Communications (2023)
-
Tissue-resident memory T cells in the urogenital tract
Nature Reviews Nephrology (2022)
-
Regulation of tissue-resident memory T cells by the Microbiota
Mucosal Immunology (2022)
-
Selective dependence on IL-7 for antigen-specific CD8 T cell responses during airway influenza infection
Scientific Reports (2022)