Abstract
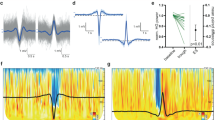
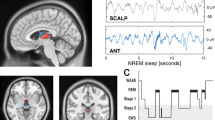
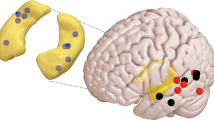
Interactions between the hippocampus and the cortex are critical for memory. Interictal epileptiform discharges (IEDs) identify epileptic brain regions and can impair memory, but the mechanisms by which they interact with physiological patterns of network activity are mostly undefined. We show in a rat model of temporal lobe epilepsy that spontaneous hippocampal IEDs correlate with impaired memory consolidation, and that they are precisely coordinated with spindle oscillations in the prefrontal cortex during nonrapid-eye-movement (NREM) sleep. This coordination surpasses the normal physiological ripple–spindle coupling and is accompanied by decreased ripple occurrence. IEDs also induce spindles during rapid-eye movement (REM) sleep and wakefulness—behavioral states that do not naturally express these oscillations—by generating a cortical 'down' state. In a pilot clinical examination of four subjects with focal epilepsy, we confirm a similar correlation of temporofrontal IEDs with spindles over anatomically restricted cortical regions. These findings imply that IEDs may impair memory via the misappropriation of physiological mechanisms for hippocampal–cortical coupling, which suggests a target for the treatment of memory impairment in epilepsy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hermann, B.P. et al. Cognitive prognosis in chronic temporal lobe epilepsy. Ann. Neurol. 60, 80–87 (2006).
Ebus, S. et al. Cognitive effects of interictal epileptiform discharges in children. Eur. J. Paediatr. Neurol. 16, 697–706 (2012).
Lv, Y., Wang, Z., Cui, L., Ma, D. & Meng, H. Cognitive correlates of interictal epileptiform discharges in adult patients with epilepsy in China. Epilepsy Behav. 29, 205–210 (2013).
Binnie, C.D. Cognitive impairment during epileptiform discharges: is it ever justifiable to treat the EEG? Lancet Neurol. 2, 725–730 (2003).
Brinciotti, M., Matricardi, M., Paolella, A., Porro, G. & Benedetti, P. Neuropsychological correlates of subclinical paroxysmal EEG activity in children with epilepsy. 1: Qualitative features (generalized and focal abnormalities). Funct. Neurol. 4, 235–239 (1989).
Holmes, G.L. & Lenck-Santini, P.P. Role of interictal epileptiform abnormalities in cognitive impairment. Epilepsy Behav. 8, 504–515 (2006).
Khan, O.I., Zhao, Q., Miller, F. & Holmes, G.L. Interictal spikes in developing rats cause long-standing cognitive deficits. Neurobiol. Dis. 39, 362–371 (2010).
Krauss, G.L., Summerfield, M., Brandt, J., Breiter, S. & Ruchkin, D. Mesial temporal spikes interfere with working memory. Neurology 49, 975–980 (1997).
Kleen, J.K., Scott, R.C., Holmes, G.L. & Lenck-Santini, P.P. Hippocampal interictal spikes disrupt cognition in rats. Ann. Neurol. 67, 250–257 (2010).
Kleen, J.K. et al. Hippocampal interictal epileptiform activity disrupts cognition in humans. Neurology 81, 18–24 (2013).
Diekelmann, S. & Born, J. The memory function of sleep. Nat. Rev. Neurosci. 11, 114–126 (2010).
Maviel, T., Durkin, T.P., Menzaghi, F. & Bontempi, B. Sites of neocortical reorganization critical for remote spatial memory. Science 305, 96–99 (2004).
Remondes, M. & Schuman, E.M. Role for a cortical input to hippocampal area CA1 in the consolidation of a long-term memory. Nature 431, 699–703 (2004).
Buzsáki, G. Two-stage model of memory trace formation: a role for “noisy” brain states. Neuroscience 31, 551–570 (1989).
Wilson, M.A. & McNaughton, B.L. Reactivation of hippocampal ensemble memories during sleep. Science 265, 676–679 (1994).
Girardeau, G., Benchenane, K., Wiener, S.I., Buzsáki, G. & Zugaro, M.B. Selective suppression of hippocampal ripples impairs spatial memory. Nat. Neurosci. 12, 1222–1223 (2009).
Jadhav, S.P., Kemere, C., German, P.W. & Frank, L.M. Awake hippocampal sharp-wave ripples support spatial memory. Science 336, 1454–1458 (2012).
Steriade, M., McCormick, D.A. & Sejnowski, T.J. Thalamocortical oscillations in the sleeping and aroused brain. Science 262, 679–685 (1993).
Johnson, L.A., Euston, D.R., Tatsuno, M. & McNaughton, B.L. Stored-trace reactivation in rat prefrontal cortex is correlated with down-to-up state fluctuation density. J. Neurosci. 30, 2650–2661 (2010).
Battaglia, F.P., Benchenane, K., Sirota, A., Pennartz, C.M. & Wiener, S.I. The hippocampus: hub of brain network communication for memory. Trends Cogn. Sci. 15, 310–318 (2011).
Mölle, M., Yeshenko, O., Marshall, L., Sara, S.J. & Born, J. Hippocampal sharp wave-ripples linked to slow oscillations in rat slow-wave sleep. J. Neurophysiol. 96, 62–70 (2006).
Peyrache, A., Battaglia, F.P. & Destexhe, A. Inhibition recruitment in prefrontal cortex during sleep spindles and gating of hippocampal inputs. Proc. Natl. Acad. Sci. USA 108, 17207–17212 (2011).
Siapas, A.G. & Wilson, M.A. Coordinated interactions between hippocampal ripples and cortical spindles during slow-wave sleep. Neuron 21, 1123–1128 (1998).
Churchwell, J.C., Morris, A.M., Musso, N.D. & Kesner, R.P. Prefrontal and hippocampal contributions to encoding and retrieval of spatial memory. Neurobiol. Learn. Mem. 93, 415–421 (2010).
Gais, S. et al. Sleep transforms the cerebral trace of declarative memories. Proc. Natl. Acad. Sci. USA 104, 18778–18783 (2007).
Jones, M.W. & Wilson, M.A. Theta rhythms coordinate hippocampal-prefrontal interactions in a spatial memory task. PLoS Biol. 3, e402 (2005).
Jay, T.M., Burette, F. & Laroche, S. NMDA receptor-dependent long-term potentiation in the hippocampal afferent fibre system to the prefrontal cortex in the rat. Eur. J. Neurosci. 7, 247–250 (1995).
Jay, T.M. & Witter, M.P. Distribution of hippocampal CA1 and subicular efferents in the prefrontal cortex of the rat studied by means of anterograde transport of Phaseolus vulgaris-leucoagglutinin. J. Comp. Neurol. 313, 574–586 (1991).
Takita, M., Izaki, Y., Jay, T.M., Kaneko, H. & Suzuki, S.S. Induction of stable long-term depression in vivo in the hippocampal-prefrontal cortex pathway. Eur. J. Neurosci. 11, 4145–4148 (1999).
Varela, C., Kumar, S., Yang, J.Y. & Wilson, M.A. Anatomical substrates for direct interactions between hippocampus, medial prefrontal cortex, and the thalamic nucleus reuniens. Brain Struct. Funct. 219, 911–929 (2014).
Colgin, L.L. Oscillations and hippocampal-prefrontal synchrony. Curr. Opin. Neurobiol. 21, 467–474 (2011).
Malow, B.A., Lin, X., Kushwaha, R. & Aldrich, M.S. Interictal spiking increases with sleep depth in temporal lobe epilepsy. Epilepsia 39, 1309–1316 (1998).
de Guzman, P.H., Nazer, F. & Dickson, C.T. Short-duration epileptic discharges show a distinct phase preference during ongoing hippocampal slow oscillations. J. Neurophysiol. 104, 2194–2202 (2010).
Ferrillo, F. et al. Sleep-EEG modulation of interictal epileptiform discharges in adult partial epilepsy: a spectral analysis study. Clin. Neurophysiol. 111, 916–923 (2000).
Nobili, L. et al. Relationship of sleep interictal epileptiform discharges to sigma activity (12-16 Hz) in benign epilepsy of childhood with rolandic spikes. Clin. Neurophysiol. 110, 39–46 (1999).
Morimoto, K., Fahnestock, M. & Racine, R.J. Kindling and status epilepticus models of epilepsy: rewiring the brain. Prog. Neurobiol. 73, 1–60 (2004).
Buzsáki, G., Horváth, Z., Urioste, R., Hetke, J. & Wise, K. High-frequency network oscillation in the hippocampus. Science 256, 1025–1027 (1992).
Chersi, F. & Burgess, N. The Cognitive Architecture of Spatial Navigation: Hippocampal and Striatal Contributions. Neuron 88, 64–77 (2015).
Dupret, D., O'Neill, J., Pleydell-Bouverie, B. & Csicsvari, J. The reorganization and reactivation of hippocampal maps predict spatial memory performance. Nat. Neurosci. 13, 995–1002 (2010).
Shatskikh, T.N., Raghavendra, M., Zhao, Q., Cui, Z. & Holmes, G.L. Electrical induction of spikes in the hippocampus impairs recognition capacity and spatial memory in rats. Epilepsy Behav. 9, 549–556 (2006).
Buzsaki, G. et al. Nucleus basalis and thalamic control of neocortical activity in the freely moving rat. J. Neurosci. 8, 4007–4026 (1988).
Steriade, M., Nuñez, A. & Amzica, F. Intracellular analysis of relations between the slow (< 1 Hz) neocortical oscillation and other sleep rhythms of the electroencephalogram. J. Neurosci. 13, 3266–3283 (1993).
Shewmon, D.A. & Erwin, R.J. The effect of focal interictal spikes on perception and reaction time. I. General considerations. Electroencephalogr. Clin. Neurophysiol. 69, 319–337 (1988).
Holmes, G.L. EEG abnormalities as a biomarker for cognitive comorbidities in pharmacoresistant epilepsy. Epilepsia 54 (suppl. 2), 60–62 (2013).
Titiz, A.S., Mahoney, J.M., Testorf, M.E., Holmes, G.L. & Scott, R.C. Cognitive impairment in temporal lobe epilepsy: role of online and offline processing of single cell information. Hippocampus 24, 1129–1145 (2014).
Lenck-Santini, P.P. & Holmes, G.L. Altered phase precession and compression of temporal sequences by place cells in epileptic rats. J. Neurosci. 28, 5053–5062 (2008).
Marcelin, B. et al. h channel-dependent deficit of theta oscillation resonance and phase shift in temporal lobe epilepsy. Neurobiol. Dis. 33, 436–447 (2009).
Nádasdy, Z., Hirase, H., Czurkó, A., Csicsvari, J. & Buzsáki, G. Replay and time compression of recurring spike sequences in the hippocampus. J. Neurosci. 19, 9497–9507 (1999).
Bragin, A., Engel, J. Jr., Wilson, C.L., Fried, I. & Mathern, G.W. Hippocampal and entorhinal cortex high-frequency oscillations (100–500 Hz) in human epileptic brain and in kainic acid–treated rats with chronic seizures. Epilepsia 40, 127–137 (1999).
Bragin, A., Wilson, C.L. & Engel, J. Jr. Chronic epileptogenesis requires development of a network of pathologically interconnected neuron clusters: a hypothesis. Epilepsia 41 (suppl. 6), S144–S152 (2000).
Buzsáki, G., Hsu, M., Slamka, C., Gage, F.H. & Horváth, Z. Emergence and propagation of interictal spikes in the subcortically denervated hippocampus. Hippocampus 1, 163–180 (1991).
Wadman, W.J., Da Silva, F.H. & Leung, L.W. Two types of interictal transients of reversed polarity in rat hippocampus during kindling. Electroencephalogr. Clin. Neurophysiol. 55, 314–319 (1983).
Bernhardt, B.C. et al. Longitudinal and cross-sectional analysis of atrophy in pharmacoresistant temporal lobe epilepsy. Neurology 72, 1747–1754 (2009).
Kleen, J.K., Wu, E.X., Holmes, G.L., Scott, R.C. & Lenck-Santini, P.P. Enhanced oscillatory activity in the hippocampal-prefrontal network is related to short-term memory function after early-life seizures. J. Neurosci. 31, 15397–15406 (2011).
Frauscher, B. et al. Facilitation of epileptic activity during sleep is mediated by high amplitude slow waves. Brain 138, 1629–1641 (2015).
Massimini, M. et al. Triggering sleep slow waves by transcranial magnetic stimulation. Proc. Natl. Acad. Sci. USA 104, 8496–8501 (2007).
Vyazovskiy, V.V., Faraguna, U., Cirelli, C. & Tononi, G. Triggering slow waves during NREM sleep in the rat by intracortical electrical stimulation: effects of sleep/wake history and background activity. J. Neurophysiol. 101, 1921–1931 (2009).
Fahoum, F., Zelmann, R., Tyvaert, L., Dubeau, F. & Gotman, J. Epileptic discharges affect the default mode network--FMRI and intracerebral EEG evidence. PLoS One 8, e68038 (2013).
Inostroza, M. & Born, J. Sleep for preserving and transforming episodic memory. Annu. Rev. Neurosci. 36, 79–102 (2013).
Krook-Magnuson, E., Gelinas, J.N., Soltesz, I. & Buzsáki, G. Neuroelectronics and biooptics: closed-loop technologies in neurological disorders. JAMA Neurol. 72, 823–829 (2015).
Peele, D.B. & Gilbert, M.E. Functional dissociation of acute and persistent cognitive deficits accompanying amygdala-kindled seizures. Behav. Brain Res. 48, 65–76 (1992).
Liu, X. et al. Seizure-induced changes in place cell physiology: relationship to spatial memory. J. Neurosci. 23, 11505–11515 (2003).
Boukhezra, O. et al. Effect of the postictal state on visual-spatial memory in immature rats. Epilepsy Res. 55, 165–175 (2003).
Stark, E. et al. Inhibition-induced theta resonance in cortical circuits. Neuron 80, 1263–1276 (2013).
Yang, A.I. et al. Localization of dense intracranial electrode arrays using magnetic resonance imaging. Neuroimage 63, 157–165 (2012).
Desikan, R.S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31, 968–980 (2006).
Stark, E. & Abeles, M. Unbiased estimation of precise temporal correlations between spike trains. J. Neurosci. Methods 179, 90–100 (2009).
Acknowledgements
This work was supported by US National Institute of Health grants (NS90583, MH54671, MH107396, MH102840; G.B.), the US National Science Foundation PIRE (G.B.), and the Mathers Foundation (G.B.). J.N.G. is a fellow of the Pediatric Scientist Development Program, and this project was supported through the March of Dimes Foundation. D.K. is supported through the Simons Foundation (junior fellow). We thank A. Peyrache for fruitful discussion; J. Long for use of the cheeseboard maze and advice on behavioral protocol; and Z. Zhao for technical support. We thank K. Berry, A. Boomhaur and P. del Prato for providing access to the sleep electrocorticography epilepsy data. Thanks also to H.X. Wang for providing the MRI-based electrode localizations for this data.
Author information
Authors and Affiliations
Contributions
J.N.G. and G.B. conceived the project. J.N.G. and D.K. did the in vivo rat experiments. J.N.G. and D.K. analyzed rat and human neural data. T.T. and O.D. supervised the human epilepsy recordings and processes related to the institutional review board. J.N.G., D.K. and G.B. wrote the paper, with input from the other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Tables 1 and 2 (PDF 3617 kb)
41591_2016_BFnm4084_MOESM16_ESM.mp4
Rat performs cheeseboard-maze trial Caption: Left, rat navigates to and consumes three hidden water rewards during the last trial of a training session. Right, offline analysis of rat's location during same trial, with navigation periods in red and consummatory periods in blue. (MP4 11283 kb)
Rights and permissions
About this article
Cite this article
Gelinas, J., Khodagholy, D., Thesen, T. et al. Interictal epileptiform discharges induce hippocampal–cortical coupling in temporal lobe epilepsy. Nat Med 22, 641–648 (2016). https://doi.org/10.1038/nm.4084
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4084
This article is cited by
-
Spike-spindle coupling during sleep and its mechanism explanation in childhood focal epilepsy
Cognitive Neurodynamics (2024)
-
Mobile cognition: imaging the human brain in the ‘real world’
Nature Reviews Neuroscience (2023)
-
Wake slow waves in focal human epilepsy impact network activity and cognition
Nature Communications (2023)
-
Thermal neuromodulation using pulsed and continuous infrared illumination in a penicillin-induced acute epilepsy model
Scientific Reports (2023)
-
Dynamic neural representations of memory and space during human ambulatory navigation
Nature Communications (2023)