Abstract
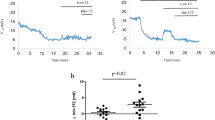
We report the results of a double-blind, placebo-controlled trial in nine cystic fibrosis (CF) subjects receiving cationic liposome complexed with a complementary DNA encoding the CF transmembrane conductance regulator (CFTR), and six CF subjects receiving only liposome to the nasal epithelium. No adverse clinical effects were seen and nasal biopsies showed no histological or immuno-histological changes. A partial restoration of the deficit between CF and non-CF subjects of 20% was seen for the response to low Cl− perfusion following CFTR cDNA administration. This was maximal around day three and had reverted to pretreatment values by day seven. In some cases the response to low Cl− was within the range for non-CF subjects. Plasmid DNA and transgene-derived RNA were detected in the majority of treated subjects. Although these data are encouraging, it is likely that transfection efficiency and the duration of expression will need to be increased for therapeutic benefit.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Change history
01 March 1995
In the January issue of Nature Medicine, an error in typography resulted in the incorrect printing of the Greek characters and μ. This error affected several papers. Nature Medicine regrets the errors, and all reprints of the articles have been corrected. Liposome-mediated CFTR gene transfer to the nasal epithelium of patients with cystic fibrosis Natasha J.
References
Riordan, J.R. et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 245, 1066–1073 (1989).
Welsh, M.J. Abnormal regulation of ion channel) in cystic fibrosis epithelia. FASEB J. 4, 2718–2725 (1990).
Knowles, M.R., Gatzy, J.T. & Boucher, R.C. Increased bioelectric potential difference across respiratory epithelia in cystic fibrosis. N. Engl. J. Med. 305, 1489–1495 (1981).
Hardcastle, J., Hardcastle, P.T., Taylor, C.J. & Goldhill, J. Failure of cholinergic stimulation to induce a secretory response from the rectal mucosa in cystic fibrosis. Gut 32, 1035–1039 (1991).
Drumm, M.L. et al. Correction of the cystic fibrosis defect in vitro by retrovirus-mediated gene transfer. Cell 62, 1227–1233 (1990).
Rich, D.P. et al. Expression of cystic fibrosis transmembrane conductance regulator corrects defective chloride channel regulation in cystic fibrosis airway epithelial cells. Nature 347, 358–363 (1990).
Alton, E.W.F.W. et al. Non-invasive liposome-mediated gene delivery can correct the ion transport defect in cystic fibrosis mutant mice. Nature Genet. 5, 135–142 (1993).
Hyde, S. et al. Correction of the ion transport defect in cystic fibrosis transgenic mice by gene therapy. Nature 362, 250–255 (1993).
Collins, F.S., Molecular biology and therapeutic implications. Science 256, 774–779 (1992).
Rosenfeld, M.A. et al. In vivo transfer of the human cystic fibrosis transmembrane conductance regulator gene to the airway epithelium. Cell 68, 143–155 (1992).
Crystal, R.C. et al. Protocol for gene therapy of the respiratory manifestations of cystic fibrosis using a replication deficient, recombinant adenovirus to transfer the normal human cystic fibrosis transmembrane conductance regulator cDNA to the airway epithelium. Federal Register 57, 49584. (1992).
Welsh, M.J. et al. Clinical Protocol. Cystic fibrosis gene therapy using adenovirus vector: In vivo safety and efficacy in nasal epithelium. Hum. gene Ther. 5, 209–219 (1994).
Wilson, J.M., Engelhardt, J.F., Grossman, M., Simon, R.H. & Yang, Y., Clinical Protocol: Gene therapy of cystic fibrosis lung disease using El deleted adenovirus: a phase 1 trial. Hum. gene Ther. (In the Press 1994).
Boucher, R.C. et al. Gene therapy for cystic fibrosis using E1-deleted adenovirus: a Phase I trial in the nasal cavity. Hum. gene Ther. 5, 501–519 (1994).
Zabner, J. et al. Adenovirus-mediated gene transfer transiently corrects the chloride transport defect in nasal epithelia of patients with cystic fibrosis. Cell 75, 1–20 (1993).
Crystal, R.G. et al. Administration of an adenovirus containing the human CFTR cDNA to the respiratory tract of individuals with cystic fibrosis. Nature Genet. 8, 42–51 (1994).
Smith, T.A.G. et al. Adenovirus mediated express levels of human factor IX in mice. Nature Genet. 5, 397–402 (1993).
Yei, S., Mittereder, N., Tang, K., O'Sullivan, C. & Trapnell, B.C. Adenovirus-mediated gene transfer for cystic fibrosis. Quantitative evaluation of repeated in vivo vector administration to the lung. Gene Ther. 1, 192–200 (1994).
Gao, X. & Haung, L. A novel cationic liposome reagent for efficient transfection of mammalian cells. Biochem. Biophys. Res. Comm. 179, 280–285 (1991).
Varney, V.A. et al. Immunohistology of the nasal mucosa following allergen-induced rhinitis. Identification of activated T-lymphocytes, eosinophils and neutrophils. Am. Rev. respir. Dis. 145, 170–176 (1992).
Alton, E.W.F.W. et al. Nasal potential difference: A clinical diagnostic test for cystic fibrosis. Eur. Resp. J. 3, 922–926 (1990).
Middleton, P.G. et al. Nasal application of the cationic liposome DC-Chol:DOPE does not alter ion transport, lung function or bacterial growth. Eur. Resp. J. 7, 442–445 (1994).
Middleton, P.G., Geddes, D.M. & Alton, E.W.F.W. Protocols for in vivo meaurement for ion transport defects in cystic fibrosis nasal epithelium. Eur. Resp. J.
Nabel, E.G. et al. Gene transfer in vivo with DNA-Liposome complexes: Lack of autoimmunity and gonadal localization. Hum. gene Ther. 3, 649–656 (1992).
Wilson, R. et al. Upper respiratory tract viral infection and mucociliary clearance Eur. J. Resp. Dis., 70,, 272–279 (1987).
Fokkens, W.J., Vroom, T.M., Gerritsma, V. & Rijntes, E. A biopsy method to obtain high quality specimens of nasal mucosa. Rhinology 26, 293–295 (1988).
Mason, D.Y. et al. Detection of T cells in paraffin wax embedded tissue using antibodies against a peptide sequence from CD3 antigen. J. Clin. Pathol. 42, 1194–1120 (1989).
Caplen, N.J. et al. Gene therapy for cystic fibrosis in humans by liposome-mediated DNA transfer the production of resources and the regulatory process. Gene Ther. 1, 139–147 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Caplen, N., Alton, E., Mddleton, P. et al. Liposome-mediated CFTR gene transfer to the nasal epithelium of patients with cystic fibrosis. Nat Med 1, 39–46 (1995). https://doi.org/10.1038/nm0195-39
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0195-39
This article is cited by
-
Potential therapeutic strategies for osteoarthritis via CRISPR/Cas9 mediated gene editing
Reviews in Endocrine and Metabolic Disorders (2024)
-
New perspectives in nanotherapeutics for chronic respiratory diseases
Biophysical Reviews (2017)
-
Advances in Cell and Gene-based Therapies for Cystic Fibrosis Lung Disease
Molecular Therapy (2012)
-
Nanoparticle-Based Vectors for Gene Delivery
Proceedings of the National Academy of Sciences, India Section B: Biological Sciences (2012)