Abstract
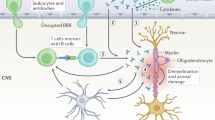
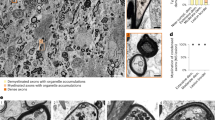
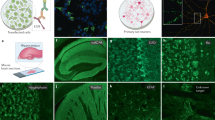
The molecular mechanisms underlying myelin sheath destruction in multiple sclerosis lesions remain unresolved. With immunogold–labeled peptides of myelin antigens and high–resolution microscopy, techniques that can detect antigen–specific antibodies in situ, we have identified autoantibodies specific for the central nervous system myelin antigen myelin/oligodendrocyte glycoprotein. These autoantibodies were specifically bound to disintegrating myelin around axons in lesions of acute multiple sclerosis and the marmoset model of allergic encephalomyelitis. These findings represent direct evidence that autoantibodies against a specific myelin protein mediate target membrane damage in central nervous system demyelinating disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Brosnan, C.F. & Raine, C.S. Mechanisms of immune injury in multiple sclerosis. Brain Pathol. 6, 243– 257 (1996).
Hohlfeld, R. Biotechnological agents for the immunotherapy of multiple sclerosis. Principles, problems and perspectives. Brain 120, 865–916 (1997).
Wekerle, H., Linington, C., Lassmann, H. & Meyermann, R. Cellular immune reactivity within the CNS. Trends Neurosci. 9, 271–277 (1986).
Willenborg, D.O. & Prowse, S.J. Immunoglobulin–deficient rats fail to develop experimental allergic encephalomyelitis. J. Neuroimmunol. 5, 99–109 (1983).
Piddlesden, S.J., Lassmann, H., Zimprich, F., Morgan, B.P. & Linington, C. The demyelinating potential of antibodies to myelin oligodendrocyte glycoprotein is related to their ability to fix complement. Am. J. Pathol. 143, 555–564 (1993).
Wolf, S., Dittel, B., Hardardottir, F. & Janeway, C.J. Experimental autoimmune encephalomyelitis induction in genetically B cell–deficient mice. J. Exp. Med. 184, 2271– 2278 (1996).
Raine, C.S. & Bornstein, M.B. Experimental allergic encephalomyelitis: an ultrastructural study of demyelination in vitro. J. Neuropathol. Exp. Neurol. 29, 177–191 (1970).
Raine, C.S., Hummelgard, A., Swanson, E. & Bornstein, M.B. Multiple sclerosis: serum–induced demyelination in vitro. A light and electron microscope study. J. Neurol. Sci. 20, 127–148 (1973).
Genain, C.P. et al. Antibody facilitation of multiple sclerosis–like lesions in a non human primate. J. Clin. Invest. 96, 2966–2974 (1995).
Kirschner, D.A. & Blaurock, A.E. in Myelin: Biology and Chemistry (ed. Martenson, R.E.) 3– 78 (CRC, Boca Raton, Florida, 1992).
Brunner, C., H. Lassman, Waehneldt, T.V., Matthieu, J.M. & Linington, C. Differential ultrastructural localization of myelin basic protein, myelin/oligodendroglial glycoprotein, and 2'3' cyclic nucleotide phosphodiesterase in the CNS of adult rats. J. Neurochem. 52, 296–304 (1989).
Lassmann, H., Brunner, C., Bradl, M. & Linington, C. Experimental allergic encephalomyelitis: the balance between encephalitogenic T lymphocytes and demyelinating antibodies determines size and structure of demyelinated lesions. Acta Neuropathol. (Berl) 75, 566–576 (1988).
Takisawa, T. & Robinson, J.M. Use of 1.4–nm immunogold particles for immunocytochemistry on ultra–thin–cryo–sections. J. Histochem. Cytochem. 43, 1615– 1623 (1994).
Genain, C.P. et al. Late complications of immune deviation therapy in a non human primate. Science 274, 2054– 2057 (1996).
Tabira, T. & Kira, J.–I. in Myelin: Biology and Chemistry (ed. Martenson, R.E.) 783–799 (CRC, Boca Raton, Florida, 1992).
Warren, K.G., Catz, I. & Steinman, L. Fine specificity of the antibody response to myelin basic protein in the central nervous system in multiple sclerosis: the minimal B cell epitope and a model of its features. Proc. Natl. Acad. Sci. USA 92, 11061–11065 (1995).
Raine, C.S., Snyder, D.H., Valsamis, M.P. & Stone, S.H. Chronic experimental allergic encephalomyelitis in inbred guinea pigs. An ultrastructural study. Lab. Invest. 31, 369–380 (1974).
Dal Canto, M.C., Wisniewski, H.M., Johnson, A.B., Brostoff, S.W. & Raine, C.S. Vesicular disruption of myelin in autoimmune demyelination. J. Neurol. Sci. 24, 313–319 (1975).
Seil, F.J., Falk, G.A., Kies, M.W. & Alvord, E.C. The in vitro demyelinating activity of sera from guinea pigs sensitized with whole CNS and with purified encephalitogen. Exp. Neurol. 22, 545–555 (1968).
Raine, C.S., Johnson, A.B., Marcus, D.M., Suzuki, A. & Bornstein, M.B. Demyelination in vitro. Absorption studies demonstrate that galactocerebroside is a major target. J. Neurol. Sci. 52, 117– 131 (1981).
Brosnan, C.F., Traugott, U. & Raine, C.S. Analysis of humoral and cellular events and the role of lipid haptens during CNS demyelination. Acta Neuropathol. (Berlin) Suppl. 9, 59–70 (1983).
Linington, C. & Lassmann, H. Antibody responses in chronic relapsing experimental allergic encephalomyelitis: correlation of serum demyelinating activity with antibody titre to the myelin/oligodendrocyte glycoprotein (MOG). J. Neuroimmunol. 17, 61– 69 (1987).
Gerritse, K. et al. The involvement of specific anti myelin basic protein antibody–forming cells in multiple sclerosis immunopathology. J. Neuroimmunol. 49, 153–159 (1994).
Sun, J. et al. T and B cell responses to myelin oligodendrocyte glycoprotein in multiple sclerosis. J. Immunol. 146, 1490–1495 (1991).
Benveniste, E.N. Role of macrophages/microglia in multiple sclerosis and experimental allergic encephalomyelitis. J. Mol. Med. 75, 165– 173 (1997).
Morris, G. in Epitope mapping protocols 1–416 (Humana Press, Totowa, 1996).
Amor, S. et al. Identification of epitopes of myelin oligodendrocyte glycoprotein for the induction of experimental allergic encephalomyelitis in SJL and Biozzi AB/H mice. J. Immunol. 153, 4349– 4356 (1994).
Ichikawa, M., Johns, T.G., Liu, J. & Bernard, C.C.A. Analysis of the fine B–cell specificity during the chronic/relapsing course of a multiple sclerosis–like disease in Lewis rats injected with the encephalitogenic myelin oligodendrocyte glycoprotein peptide 35–55. J. Immunol 157, 919–926 (1996).
Lassmann, H., Raine, C.S., Antel, J. & Prineas, J. Immunopathology of multiple sclerosis: report on an international meeting held at the Institute of Neurology of the University of Vienna. J. Neuroimmunol. 86, 213–217 (1998).
Aleu, F.P., Katzman, R. & Terry, R.D. Fine structure and electrolyte analysis of cerebral edema induced by alkyl–tin intoxication. J. Neuropath. Exp. Neurology 22, 403– (1963).
Compston, A., Scolding, N., Wren, D. & Noble, M. The pathogenesis of demyelinating disease: insights from cell biology. Trends Neurosci. 14, 175–182 (1991).
Prineas, J.W. & Connell, F. The fine structure of chronically active multiple sclerosis plaques. Neurology 28, 68–75 (1978).
Epstein, L.G., Prineas, J.W. & Raine, C.S. Attachment of myelin to coated pits on macrophages in experimental allergic encephalomyelitis. J. Neurol. Sci. 61, 341–348 (1983).
Moore, G.R.W. & Raine, C.S. Immunogold localization and analysis of IgG during immune–mediated demyelination. Lab. Invest. 59, 641–648 (1988).
Pham–Dinh, D. et al. Characterization and expression of the cDNA coding for the human myelin/oligodendrocyte glycoprotein. J. Neurochem. 63, 2353–2356 (1994).
Acknowledgements
We thank E.P. Reich (Anergen, Redwood City, California) for the gift of MSA peptide, and E. Swanson, M. Pakingan and H. Finch for technical assistance. This work was supported by grants from the National Institutes of Health (NS 08952, NS 11920 to C.S.R.; AI 43073 to S.L.H.), the National Multiple Sclerosis Society (RG 1001–I–9 to C.S.R.), and the Nancy Davis Center Without Walls. C.P.G. is a Harry Weaver Neuroscience Scholar of the National Multiple Sclerosis Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Genain, C., Cannella, B., Hauser, S. et al. Identification of autoantibodies associated with myelin damage in multiple sclerosis. Nat Med 5, 170–175 (1999). https://doi.org/10.1038/5532
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/5532
This article is cited by
-
High throughput method for detecting murine brain atrophy using a clinical 3T MRI
BMC Medical Imaging (2023)
-
Detection of antibodies against the huntingtin protein in human plasma
Cellular and Molecular Life Sciences (2023)
-
Ocrelizumab effect on humoral and cellular immunity in multiple sclerosis and its clinical correlates: a 3-year observational study
Journal of Neurology (2023)
-
B cells in central nervous system disease: diversity, locations and pathophysiology
Nature Reviews Immunology (2022)
-
Anti-CD20 therapies for multiple sclerosis: current status and future perspectives
Journal of Neurology (2022)