Abstract
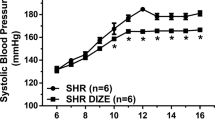
Prostaglandins (PGs) are ubiquitous lipid mediators derived from cyclooxygenase metabolism of arachidonic acid that exert a broad range of physiologic activities, including modulation of inflammation, ovulation1,2 and arterial blood pressure3,4. PGE2, a chief cyclooxygenase product, modulates blood pressure and fertility, although the specific G protein–coupled receptors5,6 mediating these effects remain poorly defined. To evaluate the physiologic role of the PGE2 EP2 receptor subtype, we created mice with targeted disruption of this gene (EP2–/–). EP2–/– mice develop normally but produce small litters and have slightly elevated baseline systolic blood pressure. In EP2–/– mice, the characteristic hypotensive effect of intravenous PGE2 infusion was absent; PGE2 infusion instead produced hypertension. When fed a diet high in salt, the EP2–/– mice developed profound systolic hypertension, whereas wild–type mice showed no change in systolic blood pressure. Analysis of wild–type and EP2–/– mice on day 5 of pregnancy indicated that the reduced litter size of EP2–/– mice is due to a pre–implantation defect. This reduction of implanted embryos could be accounted for by impaired ovulation and dramatic reductions in fertilization observed on day 2 of pregnancy. These data demonstrate that the EP2 receptor mediates arterial dilatation, salt–sensitive hypertension, and also plays an essential part in female fertility.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Smith, G., Roberts, R., Hall, C. & Nuki, G. Reversible ovulatory failure associated with the development of luteinized unruptured follicles in women with inflammatory arthritis taking non–steroidal anti–inflammatory drugs. Br. J. Rheumatol. 35, 458– 462 (1996).
Downs, S.M. & Longo, F.J. Effects of indomethacin on preovulatory follicles in immature, superovulated mice. Am. J. Anatomy 164, 265–274 (1982).
Gurwitz, J.G. et al. Initiation of antihypertensive treatment during nonsteroidal anti–inflammatory drug therapy. J. Am. Med. Assoc. 272, 781–786 (1994).
Johnson, A.G. NSAIDs and increased blood pressure. What is the clinical significance? Drug Saf. 17, 277–289 (1997).
Coleman, R.A., Kennedy, I., Humphrey, P.P.A., Bunce, K. & Lumley, P. in Comprehensive Medical Chemistry (ed. Emmer, J.C.) 643–714 (Pergammon, Oxford,1990).
Coleman, R.A. et al. A novel inhibitory prostanoid receptor in piglet saphenous vein. Prostaglandins 47, 151– 168 (1994).
Gardiner, P.J. Characterization of prostanoid relaxant/inhibitory receptors (psi) using a highly selective agonist, TR4979. Br. J. Pharmacol. 87, 45–56 (1986).
Coleman, R.A., Kennedy, I. & Sheldrick, R.L.G. New Evidence with Selective Agonists and Antagonists for the Subclassification of PGE2 –sensitive (EP) Receptors. Adv. Prostaglandin Thromboxane Leukot. Res. 17, 467–470 (1987).
Simpson, F.O. in Hypertension: Pathophysiology, Diagnosis, and Management (eds. Laragh, J.H. & Brenner, B.M.) 273–280 (Raven, New York, 1995).
Challis, J.R.G. Prostaglandins and reproduction–what do knockouts really tell us? Nature Med. 3, 1326–1327 (1997).
Priddy, A.R. & Killick, S.R. Eicosanoids and ovulation. Prostaglandins Leukot. Essent. Fatty Acids 49, 827– 831 (1993).
Weitlauf, H.M. in The Physiology of Reproduction (eds. Knobil, E. & Neill, J.) pp 391–440 (Raven, New York, 1994).
Bonventre, J.V. et al. Reduced fertility and postischaemic brain injury in mice deficient in cytosolic phospholipase A2. Nature 390, 622–625 (1997).
Uozumi, N. et al. Role of cytosolic phospholipase A2 in allergic response and parturition. Nature 390, 618– 622 (1997).
Lau, I.F., Saksena, S.K. & Chang, M.C. Pregancy blockade by indomethacin, an inhibitor of prostaglandin synthesis: its reversal by prostaglandins and progesterone in mice. Prostaglandins 4, 795– 803 (1973).
Hoffman, L. Antifertility effect of indomethacin during early pregnancy in the rabbit. Biol. Reprod. 18, 148– 153 (1978).
Kennedy, T.G. Evidence for a role for prostaglandins in the initiation of blastocyst implantation in the rat. Biol. Reprod. 16, 286– 291 (1977).
Lim, H., Paria, B.C., Das, S.K., Dinchuk, J.E., Langenbach, R., Trzaskos, J.M., and Dey, S.K. Multiple female reproductive failures in cyclooxygenase 2–deficient mice. Cell 91, 197–208 (1997).
Regan, J.W. et al. Cloning of a novel human prostaglandin receptor with characteristics of the pharmacologically defined EP2 subtype. Mol. Pharmacol. 46, 213–220 (1994).
Tybulewicz, V. & Mulligan, R. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c–abl proto–oncogene. Cell 65, 1153–1163 (1991).
Acknowledgements
We thank A. George, T. Inagami and D. Threadgill for critical reading of the manuscript. Gene targeting and embryonic stem cell microinjection were done by the Vanderbilt Transgenic/ES Cell Shared Resource. Support for this project was provided in part by NIH grants GM–15431 (J.A.O. and R.M.B.), DK–46205 (R.M.B.) and DK–37097 (M.D.B.). C.R.J.K. is the recipient of an NKF fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kennedy, C., Zhang, Y., Brandon, S. et al. Salt–sensitive hypertension and reduced fertility in mice lacking the prostaglandin EP2 receptor. Nat Med 5, 217–220 (1999). https://doi.org/10.1038/5583
Issue Date:
DOI: https://doi.org/10.1038/5583
This article is cited by
-
Neuroinflammatory mediators in acquired epilepsy: an update
Inflammation Research (2023)
-
Metabolism pathways of arachidonic acids: mechanisms and potential therapeutic targets
Signal Transduction and Targeted Therapy (2021)
-
COX-2-derived PGE2 triggers hyperplastic renin expression and hyperreninemia in aldosterone synthase-deficient mice
Pflügers Archiv - European Journal of Physiology (2018)
-
Prostaglandins E2 signal mediated by receptor subtype EP2 promotes IgE production in vivo and contributes to asthma development
Scientific Reports (2016)
-
Prostaglandin E2 and programmed cell death 1 signaling coordinately impair CTL function and survival during chronic viral infection
Nature Medicine (2015)