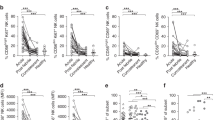
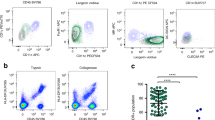
Abstract
Dengue virus (DV), an arthropod-borne flavivirus, causes a febrile illness for which there is no antiviral treatment and no vaccine1,2. Macrophages are important in dengue pathogenesis; however, the initial target cell for DV infection remains unknown. As DV is introduced into human skin by mosquitoes of the genus Aedes, we undertook experiments to determine whether human dendritic cells (DCs) were permissive for the growth of DV. Initial experiments demonstrated that blood-derived DCs were 10-fold more permissive for DV infection than were monocytes or macrophages. We confirmed this with human skin DCs (Langerhans cells and dermal/interstitial DCs). Using cadaveric human skin explants, we exposed skin DCs to DV ex vivo. Of the human leukoctye antigen DR-positive DCs that migrated from the skin, emigrants from both dermis and epidermis, 60–80% expressed DV antigens. These observations were supported by histologic findings from the skin rash of a human subject who received an attenuated tetravalent dengue vaccine. Immunohistochemistry of the skin showed CD1a-positive DCs double-labeled with an antibody against DV envelope glycoprotein. These data demonstrate that human skin DCs are permissive for DV infection, and provide a potential mechanism for the transmission of DV into human skin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Halstead, S.B. Pathogenesis of dengue: challenges to molecular biology. Science 239, 476–481 ( 1988).
Rigau-Perez, J. et al. Dengue and dengue heamorrhagic fever. Lancet 352, 971–977 (1998).
Taweechaisupapong, S. et al. Langerhans cell density and serological changes following intradermal immunisation of mice with dengue 2 virus. J. Med. Microbiol. 45, 138–145 ( 1996).
Taweechaisupapong, S., Sriurairatana, S., Angsubhakorn, S., Yoksan, S. & Bhamarapravati, N. In vivo and in vitro studies of the morphological change in the monkey epidermal Langerhans cells following exposure to dengue 2 virus. Southeast Asian J. Trop. Med. Public Health 27, 664–672 (1996).
Cella, M. et al. Maturation, activation, and protection of dendritic cells induced by double-stranded RNA. J. Exp. Med. 189, 821–829 (1999).
Rice, C.M. in Fields Virology (eds. Fields, B.N., Knipe, D.M. & Howley, P.M.) 931–959 (Lippincott-Raven, Philadelphia, 1996).
Pope, M., Betjes, M.G.H., Hirmand, H., Hoffman, L. & Steinman, R. Both dendritic cells and memory T lymphocytes emigrate from organ cultures of human skin and form distinctive dendritic-T-cell conjugates. J. Invest. Dermatol. 104 , 11–17 (1995).
Labuda, M. et al. Importance of localized skin infection in tick-borne encephalitis virus transmission. Virology 219, 357– 366 (1996).
O'Sullivan, M.K.H. The differentiation state of monocytic cells affects their susceptibility to infection and the effects of infection by dengue virus. J. Gen. Virol. 75, 2387–2392 ( 1994).
Morens, D. M., Halstead, S.B. & Marchette, N.J. Profiles of antibody-dependent enhancement of dengue virus type 2 infection. Microb. Pathog. 3, 231–237 (1987).
Morens, D.M., Larsen, L.K. & Halstead, S.B. Study of the distribution of antibody-dependent enhancement determinants on dengue 2 isolates using dengue 2-derived monoclonal antibodies . J. Med. Virol. 22, 163– 167 (1987).
Chen, Y. et al. Dengue virus infectivity depends on envelope protein binding to target cell heparin sulfate. Nature Med. 3, 866–871 (1997).
Halstead, S., O'Rourke, E. & Allison, A. Dengue viruses and mononuclear phagocytes. J. Exp. Med. 146, 218–229 (1977).
Frankel, S.S. et al. Neutralizing monoclonal antibodies block human immunodeficiency virus type 1 infection of dendritic cells and transmission to T cells. J. Virol. 72, 9788–9794 (1998).
Pope, M. et al. Human immunodeficiency virus type 1 strains of subtypes B and E replicate in cutaneous dendritic cell T-cell mixtures without displaying subtype-specific tropism. J. Virol. 71, 8001–8007 (1997).
Henchal, E.A., McCown, J.M., Burke, D.S., Seguin, M.C. & Brandt, W.E. Epitopic analysis of antigenic determinants on the surface of dengue-2 virions using monoclonal antibodies . Am. J. Trop. Med. Hyg. 34, 162– 169 (1985).
Raviprakash, K., Sinha, M., Hayes, C.G. & Porter, K.R. Conversion of dengue virus replication form RNA (RF) to replicative intermediate (RI) by nonstructural proteins NS-5 and NS-3. Am. J. Trop. Med. Hyg. 58, 90–95 (1998).
Morens, DM. Simplified plaque reduction neutralization assay for dengue viruses by semimicro methods in BHK-21 cells: Comparison of the BHK suspension test with standard plaque reduction neutralization. J. Clin. Microbiol. 22, 250–253 (1985).
Wu, S.-J.L. et al. Evaluation of the severe combined immunodeficient (SCID) mouse as an animal model for dengue viral infection. Am. J. Trop. Med. Hyg. 52, 468–476 ( 1995).
Frankel, S.S. et al. Active replication of HIV-1 at the lymphoepithelial surface of the tonsil. Am. J. Pathol. 151, 89– 96 (1997).
de Saint-Vis, B. et al. A novel lysosome-associated membrane glycoprotein, DC-LAMP, induced upon DC maturation, is transiently expressed in MHC class II compartment . Immunity 9, 325–336 (1998).
Acknowledgements
The authors thank N. Bhamarapravati, R. Steinman, S. Halstead and J. McNeil for scientific guidance and manuscript review; and D. Ford for assistance with graphics. This work was supported in part by cooperative agreement number DAMD17-93-V 3004, between the US Army Medical Research and Materiel Command and the Henry M. Jackson Foundation for the Advancement of Military Medicine and by the Military Infectious Disease Research Program ONR account 821.103. PATH.1918. The views and opinions expressed herein are those of the authors and do not purport to reflect the official policy or position of the Department of Defense. The human clinical trial was conducted in accordance with a human subjects protocol approved by The Walter Reed Army Medical Center Committee for the Protection of Human Subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, SJ., Grouard-Vogel, G., Sun, W. et al. Human skin Langerhans cells are targets of dengue virus infection. Nat Med 6, 816–820 (2000). https://doi.org/10.1038/77553
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/77553
This article is cited by
-
A mosquito salivary protein-driven influx of myeloid cells facilitates flavivirus transmission
The EMBO Journal (2024)
-
Skin muscle is the initial site of viral replication for arboviral bunyavirus infection
Nature Communications (2024)
-
Stochastic SIV in-host model of dengue virus transmission
International Journal of Dynamics and Control (2024)
-
Angiotensin II and dengue
Archives of Virology (2023)
-
A mosquito salivary protein promotes flavivirus transmission by activation of autophagy
Nature Communications (2020)