Abstract
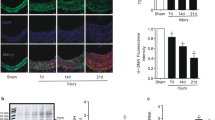
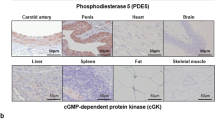
Injury of the arterial wall induces the formation of the neoin-tima1. This structure is generated by the growth of mitogenically activated smooth muscle cells of the arterial wall2–5. The molecular mechanism underlying the formation of the neointima involves deregulated cell growth, primarily triggered by the injury of the arterial wall6–9. The activated gene products transmitting the injury-induced mitogenic stimuli have been identified and inhibited by several means: transdominant negative expression vectors, antisense oligodeoxynucleotides, adenovirus-mediated gene transfer, antibodies and inactivating drugs8,10–12. Results of our study show that local administration of 3′,5′-cyclic AMP and phosphodiesterase-inhibitor drugs (aminophylline and amrinone) to rats markedly inhibits neointima formation after balloon injury in vivo and in smooth muscle cells in vitro. The growth inhibitory effect of aminophylline was completely reversed by the inhibition of cAMP-dependent protein kinase A (PKA). These findings indicate an alternative approach to the treatment of diseases associated with injury-induced cell growth of the arterial wall, as stimulation of cAMP signaling is pharmacologically feasible in the clinical setting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Schwartz, S.M., Heimark, R.L. & Majesky, M.W. Developmental mechanisms underlying pathology of arteries. Physiol. Rev. 70, 1177–1209 (1990).
Ross, R. The pathogenesis of atherosclerosis: A perspective for 1990. Nature 362, 801–809 (1993).
Virmani, R., Farb, A. & Burke, A.P. Coronary angioplasty from the prospective of atherosclerotic plaque: Morphologic predictors of immediate success and restenosis. Am. Heart J. 127, 163–179 (1994).
Clowes, A.W., Reidy, M.A. & Clowes, M.M. Mechanisms of stenosis after arterial injury. Lab. Invest. 49, 208–215 (1983).
Clowes, A.W., Reidy, M.A. & Clowes, M.M. Kinetics of cellular proliferation after arterial injury. I. Smooth muscle growth in the absence of endothelium. Lab. Invest. 49, 327–333 (1983).
Edelman, E.R., Nugent, M.A., Smith, L.T. & Karnovsky, M.J. Basic fibroblast growth factor enhances the coupling of intimal hyperplasia and proliferation of vasa vasorum in injured rat arteries. J. Clin. Invest. 89, 465–473 (1992).
Jawien, A., Bowen-Pope, D.F., Lindner, V., Schwartz, S.M. & Clowes, A.W. Platelet-derived growth factor promotes smooth muscle migration and intimal thickening in a rat model of balloon angioplasty. J. Clin. Invest. 89, 507–511 (1992).
Indolfi, C. et al. Inhibition of cellular ras prevents smooth muscle cell proliferation after vascular injury in vivo. Nature Med. 1, 541–545 (1995).
Indolfi, C., Chiariello, M. & Awedimento, E.V. Selective gene therapy for proliferative disorders: Sense and antisense. Nature Med. 2, 634–635 (1996).
Chang, M.V. et al. Adenovirus-mediated transfer of the herpes simplex virus thymidine kinase inhibits vascular smooth muscle cell proliferation and neointima formation following balloon angioplasty of the rat carotid artery. Mol. Med. 1, 172–181 (1995).
Chang, M.V. et al. Cytostatic gene therapy for vascular proliferative disorder with a constitutively active form of the retinoblastoma gene product. Science 267 518–522 (1995).
Simons, M., Edelman, E.R., DeKeyser, J.L., Langer, R. & Rosenberg, R.D. Antisense c-myb oligonucleotides inhibit intimal arterial smooth muscle cell accumulation in vivo. Nature 359, 67–70 (1992).
Assender, J.W., Southgate, K.M., Hallett, M.B. & Newby, A.C. Inhibition of proliferation, but not of Ca++ mobilization, by cyclic AMP and GMP in rabbit aortic smooth muscle cells. Biochem.J. 288, 527–532 (1992).
Boynton, A.L. & Whitfield, J.F. The role of cAMP in cell proliferation: A critical as sessment of the evidence. Adv. Cyclic Nudeotide Res. 15, 193–294 (1983).
Cornwell, T.L., Arnold, E., Boerth, N.J. & Lincoln, T.M. Inhibition of smooth muscle cell growth by nitric oxide and activation of cAMP-dependent protein kinase by cGMP. Am. J. Physiol. 36, 1405–1413 (1994).
Yokozaki, H. et al. Unhydrolyzable analogues of adenosine 3′:5′-monophosphate demonstrating growth inhibition and differentiation in human cancer cells. Cancer Res. 52, 2504–2508 (1992).
Skalhegg, B.S. et al. Cyclic AMP-dependent protein kinase type I mediates the inhibitory effects of 3′ 5′-cyclic adenosine monophosphate on cell replication in human T lymphocytes. J. Biol. Chem. 267, 15707–15714 (1992).
Sutherland, E.W. Studies on the mechanism of hormone action. Science 177, 401–408 (1972).
Schwartz, D.A. & Rubin, C.S. Regulation of cAMP-dependent protein kinase subunit levels in friend erythroleukemic cells. J. Biol. Chem. 258, 777–784 (1983).
Pastan, I.H., Johnson, G.S. & Anderson, W.B. Role of cyclic nucleotides in growth control. Annu. Rev. Biochem. 44, 491–522 (1975).
Mitsuzawa, H. Increases in cell size at START caused by hyperactivation of the cAMP pathway in Saccharomyces cerevisae. Mol. Gen. Genet. 243, 158–165 (1994).
Goodman & Gilman's The Pharmacological Basis of Therapeutics. (McGraw-Hill, NewYork, 1995).
Indolfi, C. et al. Smooth muscle cell proliferation is proportional to the degree of balloon injury in a rat model of angioplasty. Circulation 92, 1230–1235 (1995).
Powell, J.S. et al. Inhibition of angiotensin-converting enzyme prevent myointimal proliferation after vascular injury. Science 245, 186–188 (1989).
Grieco, D., Porcellini, A., Avvedimento, E.V. & Gottesman, M.E. Requirement for cAMP-PKA pathway activation by M-phase–promoting factor in the transition from mitosis to interphase. Science 271, 1718–1723 (1996).
Bokar, J.A. et al. Characterization of the cAMP responsive elements from the genes for the alpha-subunit of glycoprotein hormones and phosphoenolpyruvate car-boxykinase (GTP): Conserved features of nuclear protein binding between tissues and species. J. Biol. Chem. 263, 19740–19747 (1988).
Krebs, E.G. & Beavo, J.A. Phosphorylation-dephosphorylation of enzymes. Annu. Rev. Biochem. 48, 923–959 (1979).
Stryer, L. & Bourne, H.R. G proteins: a family of signal transducers [Review]. Annu. Rev. Cell. Biol. 2, 391–419 (1986).
Choi, E.J., Xia, Z., Villacres, E.C. & Storm, D.R. The regulatory diversity of the mammalian adenyl cyclases [Review]. Curr. Opin. Cell Biol. 5, 269–273 (1993).
Cook, S.J. & McCormick, F. Inhibition by cAMP of Ras-dependent activation of Raf. Science 262, 1069–1072 (1993).
Holmes, D.R., Jr., et al. Restenosis after percutaneous transluminal coronary angioplasty: A report from the PTCA registry of the National Heart, Lung, and Blood Institute. Am. J. Cardiol. 53, 77C–81C (1984).
McBride, W. et al. Restenosis after successful coronary angioplasty. N. Engl. J. Med. 318, 1734–1737 (1988).
Leimgruber, P.P. et al. Restenosis after successful coronary angioplasty in patients with single-vessel disease. Circulation 73, 710–717 (1986).
Guiteras Va, P. et al. Restenosis after successful percutaneous transluminal coronary angioplasty: The Montreal Heart Institute experience. Am. J. Cardiol. 60 (Suppl.), 50B–55B (1987).
The Multicenter European Research Trial with Cilazapril after Angioplasty to Prevent Transluminal Coronary Obstruction and Restenosis (MERCATOR) Study Group. Does the new angiotensin converting enzyme inhibitor cilazapril prevent restenosis after percutaneous transluminal coronary angioplasty? Results of the MERCATOR study: A multicenter, randomized, double-blind placebo-controlled trial. Circulation 86, 100–110 (1991).
Herrmann, J.P., Hermans, W.R., Vos, J. & Serruys, P.W. Pharmacological approaches to the prevention of restenosis following angioplasty: The search for the Holy Grail? Drugs 46, 249–262 (1993).
Laird, J.R. et al. Inhibition of neointimal proliferation with low-dose irradiation from a β-particle-emitting stent. Circulation 93, 529–536 (1996).
Serruys, P.W. et al. Heparin-coated Palmaz-Schatz stents in human coronary arteries. Circulation 93, 412–422 (1996).
Nunes, G.L. et al., Inhibition of platelet-dependent thrombosis by local delivery of heparin with a hydrogel-coated balloon. Circulation 92, 1697–1700 (1995).
Rothman, A. et al. Development and characterization of a cloned rat pulmonary arterial smooth muscle cell line that maintains differentiated properties through multiple subcultures. Circulation 86, 1977–1986 (1992).
Wilkinson, L. SYSTAT, the System for Statistics. (SYSTAT, Inc., Evanston, IL, 1988).
Koumi, S., Backer, C.L., Arentzen, C.E. & Sato, R. Beta adrenergic modulation of the inwardly rectifying potassium channel in isolated human ventricular myocytes: Alteration in channel response to beta adrenergic stimulation in failing human hearts. J. Clin. Invest. 96, 2870–2881 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Indolfi, C., Vittorio Avvedimento, E., Lorenzo, E. et al. Activation of cAMP–PKA signaling in vivo inhibits smooth muscle cell proliferation induced by vascular injury. Nat Med 3, 775–779 (1997). https://doi.org/10.1038/nm0797-775
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0797-775
This article is cited by
-
Genetics of ischemic stroke functional outcome
Journal of Neurology (2024)
-
Structure‐based identification of dual ligands at the A2AR and PDE10A with anti‐proliferative effects in lung cancer cell‐lines
Journal of Cheminformatics (2021)
-
Enhanced fatty acid oxidation provides glioblastoma cells metabolic plasticity to accommodate to its dynamic nutrient microenvironment
Cell Death & Disease (2020)
-
Hindlimb Ischemia Impairs Endothelial Recovery and Increases Neointimal Proliferation in the Carotid Artery
Scientific Reports (2018)
-
Dual Role of CREB in The Regulation of VSMC Proliferation: Mode of Activation Determines Pro- or Anti-Mitogenic Function
Scientific Reports (2018)