Abstract
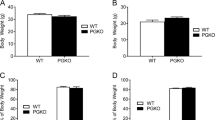
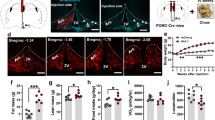
Pro-opiomelanocortin (POMC)-derived peptides (the melanocortins adrenocorticotropin, α-, β- and γ-melanocyte stimulating hormone; and the endogenous opioid β-endorphin) have a diverse array of biological activities, including roles in pigmentation, adrenocortical function and regulation of energy stores, and in the immune system and the central and peripheral nervous systems1. We show here that mice lacking the POMC-derived peptides have obesity, defective adrenal development and altered pigmentation. This phenotype is similar to that of the recently identified human POMC-deficient patients2. When treated with a stable α-melanocyte-stimulating hormone agonist, mutant mice lost more than 40% of their excess weight after 2 weeks. Our results identify the POMC-null mutant mouse as a model for studying the human POMC-null syndrome, and indicate the therapeutic use of peripheral melanocortin in the treatment of obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Smith, A.I. & Funder, J.W. Proopiomelanocortin processing in the pituitary, central nervous system, and peripheral tissues. Endocr. Rev. 9, 159–79 ( 1988).
Krude, H. et al. Severe early-onset obesity, adrenal insufficiency and red hair pigmentation caused by POMC mutations in humans. Nature Genet. 19, 155–157 ( 1998).
Notake, M. et al. Isolation and characterization of the mouse corticotropin-beta-lipotropin precursor gene and a related pseudogene. FEBS Lett. 156, 67–71 (1983).
Lu, D. et al. Agouti protein is an antagonist of the melanocyte-stimulating-hormone. Nature 371, 799–802 (1994).
Graham, M., Shutter, J.R., Sarmiento, U., Sarosi, I. & Stark, K.L. Overexpression of Agrt leads to obesity in transgenic mice. Nature Genet. 17, 273–274 (1997).
Ollmann, M.M. et al. Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science 278, 135–138 (1997).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131– 141 (1997).
Kastin, A.J., Redding, T.W., Hall, R., Besser, G.M. & Schally, A.V. Lipid mobilizing hormones of the hypothalamus and pituitary. Pharmacol. Biochem. Behav. 3, 121–126 (1975).
Richter, W.O. & Schwandt, P. Lipolytic potency of proopiomelanocorticotropin peptides in vitro. Neuropeptides 9, 59– 74 (1987).
Boston, B.A. & Cone, R.D. Characterization of melanocortin receptor subtype expression in murine adipose tissues and in the 3T3-L1 cell line. Endocrinology 137, 2043– 2050 (1996).
Jones, B.H. et al. Upregulation of adipocyte metabolism by agouti protein: possible paracrine actions in yellow mouse obesity. Am. J. Physiol. 270, E192–E196 (1996).
Xue, B., Moustaid, N., Wilkison, W.O. & Zemel, M.B. The agouti gene product inhibits lipolysis in human adipocytes via a Ca2+-dependent mechanism. FASEB J. 12, 1391– 1396 (1998).
Burchill, S.A., Thody, A.J. & Ito, S. Melanocyte-stimulating hormone, tyrosinase activity and the regulation. J. Endocrinol. 109, 15– 21 (1986).
Robbins, L.S. et al. Pigmentation phenotypes of variant extension locus alleles result from point mutations that alter MSH receptor function. Cell 72, 827–34 ( 1993).
Joerg, H., Fries, H.R., Meijerink, E. & Stranzinger, G.F. Red coat color in Holstein cattle is associated with a deletion in the MSHR gene. Mamm. Genome 7, 317– 318 (1996).
Estivariz, F.E., Iturriza, F., McLean, C., Hope, J. & Lowry, P.J. Stimulation of adrenal mitogenesis by N-terminal proopiocortin. Nature 297, 419–422 (1982).
Muglia, L., Jacobson, L., Dikkes, P. & Majzoub, J.A. Corticotropin-releasing hormone deficiency reveals major fetal but not adult glucocorticoid need. Nature 373, 427–432 (1995).
Tempel, D.L. & Leibowitz, S.F. Adrenal steroid receptors: interactions with brain neuropeptide systems in relation to nutrient intake and metabolism. J. Neuroendocrinol. 6, 479– 501 (1994).
Strack, A.M., Horsley, C.J., Sebastian, R.J., Akana, S.F. & Dallman, M.F. Glucocorticoids and insulin: complex interaction on brown adipose tissue. Am. J. Physiol. 268, R1209–R1216 (1995).
Solomon, J. & Mayer, J. The effect of adrenalectomy on the development of the obese- hyperglycemic syndrome in ob-ob mice. Endocrinology 93, 510–512 (1973).
Tokuyama, K. & Himms-Hagen, J. Adrenalectomy prevents obesity in glutamate-treated mice. Am. J.Physiol. 257, E139–E144 (1989).
Chen, W. et al. Exocrine gland dysfunction in MC5-R-deficient mice: evidence for coordinated regulation of exocrine gland function by melanocortin peptides. Cell 91, 789–798 (1997).
Cody, W.L. et al. Cyclic melanotropins. 9. 7-D-Phenylalanine analogues of the active-site sequence. J. Med. Chem. 28, 583–588 (1985).
Hogan, B., Beddington, R., Costantini, F. & Lacy, E. in Manipulating the Mouse Embryo (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1994).
Sambrook, J., Fritsch, E.F. & Maniatis, T. in Molecular Cloning. A Laboratory Manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1989).
Acknowledgements
We thank J. Elliott and S. Bui for their technical assistance, M. Glynn for art work, B. Martin for oligonucleotide synthesis, E.I. Ginns for support of the early phases of the work and M. Davisson for comments on an earlier version of the manuscript. This work was supported by a generous grant from the Board of Trustees of the Eleanor Roosevelt Institute, Denver, Colorado (M.B.B.) and by the Samuel Roberts Noble Foundation, Ardmore, Oklahoma (U.H.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yaswen, L., Diehl, N., Brennan, M. et al. Obesity in the mouse model of pro-opiomelanocortin deficiency responds to peripheral melanocortin. Nat Med 5, 1066–1070 (1999). https://doi.org/10.1038/12506
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/12506
This article is cited by
-
Hypothalamic Grb10 enhances leptin signalling and promotes weight loss
Nature Metabolism (2023)
-
Metabolic and feeding adjustments during pregnancy
Nature Reviews Endocrinology (2023)
-
The melanocortin action is biased toward protection from weight loss in mice
Nature Communications (2023)
-
Increased intrinsic and synaptic excitability of hypothalamic POMC neurons underlies chronic stress-induced behavioral deficits
Molecular Psychiatry (2023)
-
CerS6-dependent ceramide synthesis in hypothalamic neurons promotes ER/mitochondrial stress and impairs glucose homeostasis in obese mice
Nature Communications (2023)