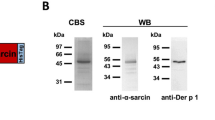
Abstract
Animal allergens are an important cause of asthma and allergic rhinitis. We designed and tested a chimeric human-cat fusion protein composed of a truncated human IgG Fcγ1 and the major cat allergen Fel d1, as a proof of concept for a new approach to allergy immunotherapy. This Fcγ-Fel d1 protein induced dose-dependent inhibition of Fel d1-driven IgE-mediated histamine release from cat-allergic donors' basophils and sensitized human cord blood-derived mast cells. Such inhibition was associated with altered Syk and ERK signaling. The Fcγ-Fel d1 protein also blocked in vivo reactivity in FcεRIα transgenic mice passively sensitized with human IgE antibody to cat and in Balb/c mice actively sensitized against Fel d1. The Fcγ-Fel d1 protein alone did not induce mediator release. Chimeric human Fcγ-allergen fusion proteins may provide a new therapeutic platform for the immune-based therapy of allergic disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Leung, D.Y.M. et al. Effect of anti-IgE therapy in patients with peanut allergy. N. Engl. J. Med. 348, 986–993 (2003).
Oliver, J.M., Kepley, C.L., Ortega, E. & Wilson, B.S. Immunologically mediated signaling in basophils and mast cells: finding therapeutic targets for allergic diseases in the human FcεRI signaling pathway. Immunopharmacology 48, 269–281 (2000).
Daeron, M. Fc receptor biology. Annu. Rev. Immunol. 15, 203–234 (1997).
Kepley, C.L. et al. Negative regulation of FcεRI signaling by FcγR II costimulation in human blood basophils. J. Allergy Clin. Immunol. 106, 337–348 (2000).
Daeron, M. et al. The same tyrosine-based inhibition motif, in the intracytoplasmic domain of FcγRIIB, regulates negatively BCR-, TCR-, and FcR-dependent cell activation. Immunity 3, 635–646 (1995).
Malbec, O. et al. Fcε Receptor I-associated lyn-dependent phosphorylation of Fcγ Receptor IIB during negative regulation of mast cell activation. J. Immunol. 160, 1647–1658 (1998).
Fong, D.C. et al. Selective in vivo recruitment of the phosphatidylinositol phosphatase SHIP by phosphorylated FcγR IIB during negative regulation of IgE-dependent mouse mast cell activation. Immunol. Lett. 54, 83–91 (1996).
Ott, V.L. & Cambier, J.C. Activating and inhibitory signaling in mast cells: New opportunities for therapeutic intervention? J. Allergy Clin. Immunol. 106, 429–440 (2000).
Ono, M., Bolland, S., Tempst, P. & Ravetch, J.V. Role of the inositol phosphatase SHIP in negative regulation of the immune system by the receptor FcγRIIB. Nature 383, 263–266 (1996).
Zhu, D., Kepley, C.L., Zhang, M., Zhang, K. & Saxon, A. A novel human immunoglobulin Fcγ-Fcε bifunctional fusion protein inhibits FcεRI-mediated degranulation. Nat. Med. 8, 518–521 (2002).
Tam, S.W., Demissie, S., Thomas, D. & Daeron, M. A bispecific antibody against human IgE and human FcγRII that inhibits antigen-induced histamine release by human mast cells and basophils. Allergy 59, 772–780 (2004).
Kepley, C.L. et al. Co-aggregation of FcγRII With FcεRI on human mast cells inhibits antigen-induced secretion and involves SHIP-Grb2-Dok complexes. J. Biol. Chem. 279, 35139–35149 (2004).
Daeron, M. et al. Regulation of high-affinity IgE receptor-mediated mast cell activation by murine low-affinity IgG receptors. J. Clin. Invest. 95, 577–585 (1995).
Morgenstern, J.P. et al. Amino acid sequence of Fel d1, the major allergen of the domestic cat: protein sequence analysis and cDNA cloning. Proc. Natl. Acad. Sci. USA 88, 9690–9694 (1991).
Vailes, L.D. et al. High-level expression of immunoreactive recombinant cat allergen (Fel d1): Targeting to antigen-presenting cells. J. Allergy Clin. Immunol. 110, 757–762 (2002).
Huston, J.S. et al. Protein engineering of antibody binding site: recovery of specific activity in an anti-digoxin single-chain Fv analogue produced in Escherichia coli. Proc. Natl. Acad. Sci. USA 85, 5879–5883 (1988).
Hulett, M.D., Witort, E., Brinkworth, R.I., Mckenzie, I.F. & Hogarth, P.M. Identification of the IgG binding site of the human low affinity receptor for IgG FcγRII. J. Biol. Chem. 269, 15287–15293 (1994).
Wedi, B., Lewrick, H., Butterfield, J.H. & Kapp, A. Human HMC-1 mast cells exclusively express the FcγR II subtype of IgG receptor. Arch. Dermatol. Res. 289, 21–27 (1996).
Kepley, C.L., Youssef, L., Andrews, R.P., Wilson, B.S. & Oliver, J.M. Multiple defects in FcεRI signaling in Syk-deficient nonreleaser basophils and IL-3-induced recovery of Syk expression and secretion. J. Immunol. 165, 5913–5920 (2000).
Suzuki, H. et al. Early and late events in FcεRI signal transduction in human cultured mast cells. J. Immunol. 159, 5881–5888 (1997).
Dombrowicz, D. et al. Anaphylaxis mediated through a humanized high affinity IgE receptor. J. Immunol. 157, 1645–1651 (1996).
Fung-Leung, W.P. et al. Transgenic mice expressing the human high-affinity immunoglobulin (Ig) E receptor α chain respond to human IgE in mast cell degranulation and in allergic reactions. J. Exp. Med. 183, 49–56 (1996).
Zhang, K. et al. Inhibition of allergen-specific IgE reactivity by a human Ig Fcγ-Fcε bifunctional protein. J. Allergy Clin. Immunol. 114, 321–327 (2004).
Dombrowicz, D., Flamand, V., Brigman, K.K., Koller, B.H. & Kinet, J.P. Abolition of anaphylaxis by targeted disruption of the high affinity immunoglobulin E receptor alpha chain gene. Cell 75, 969–976 (1993).
Witteman, A.M. et al. Fel d1-specific IgG antibodies induced by natural exposure have blocking activity in skin test. Int. Arch. Allergy Immunol. 109, 369–375 (1996).
Platts-Mills, T., Vaughan, J., Squillace, S., Woodfolk, J. & Sporik, R. Sensitization, asthma, and a modified Th2 response in children exposed to cat allergen: a population-based cross-sectional study. Lancet 357, 752–756 (2001).
Kepley, C.L., Craig, S.S. and Schwartz, L.B. Identification and partial characterization of a unique marker for human basophils. J. Immunol. 154, 6548–6555 (1995).
Schuessler, T.F., and Bates, J.H. A computer-controlled research ventilator for small animals: design and evaluation. IEEE Trans. Biomed. Eng. 42, 860–866 (1995).
Acknowledgements
Supported by an USPHS-NIH grant, AI-15251 to A.S. We thank S.L. Morrison, R. Trinh and L. A. Chan for technical advice and for providing some experimental materials. C.L.K. was supported by a grant from the American Lung Association and the AD Williams Foundation at VCU, and the Food Allergy and Anaphylaxis Network. We also thank J.P. Kinet for providing the human FcεRIα transgenic mice. We are grateful to T.H. Sulahian and P.M. Guyre for the Fel d1 cDNA construct. We thank M. Jyrala, L. Zhang, and M. Rainof for technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Daocheng Zhu, Zhang Ke and Andrew Saxon are coinventors on the patent held by the University of California for gamma-allergen proteins.
Rights and permissions
About this article
Cite this article
Zhu, D., Kepley, C., Zhang, K. et al. A chimeric human-cat fusion protein blocks cat-induced allergy. Nat Med 11, 446–449 (2005). https://doi.org/10.1038/nm1219
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1219
This article is cited by
-
An update on molecular cat allergens: Fel d 1 and what else? Chapter 1: Fel d 1, the major cat allergen
Allergy, Asthma & Clinical Immunology (2018)
-
Treating cat allergy with monoclonal IgG antibodies that bind allergen and prevent IgE engagement
Nature Communications (2018)
-
Modified Allergens for Immunotherapy
Current Allergy and Asthma Reports (2018)
-
Potential effector and immunoregulatory functions of mast cells in mucosal immunity
Mucosal Immunology (2015)
-
New treatments for allergen immunotherapy
World Allergy Organization Journal (2014)