Abstract
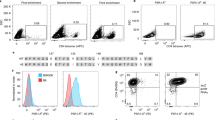
The direct assessment of T helper (TH)-cell responses specific for antigens is essential to evaluate pathogenic and protective immunity. Presently, analysis and isolation of antigen-specific TH cells is restricted to cells that produce cytokines, or can be performed only with a rare selection of specific peptide major histocompatibility complex class II (MHC II) multimers. Here we report a new method that enables the assessment and isolation of TH cells specific for a defined antigen according to CD154 expression induced after stimulation in vitro. We show that antigen-induced CD154 expression is highly sensitive and specific for human and mouse antigen-specific TH cells. Moreover, the isolation of antigen-specific CD154+ TH cells necessitates only surface staining with antibodies, thereby enabling the fast generation of antigen-specific TH cell lines. Our approach allows assessment of TH cells with a defined specificity for the combined quantitative and qualitative analysis of TH-cell immunity as well as for the isolation of specific TH cells for targeted cellular immunotherapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Altman, J.D. et al. Phenotypic analysis of antigen-specific T lymphocytes. Science 274, 94–96 (1996).
Casares, S., Bona, C.A. & Brumeanu, T.D. Enzymatically mediated engineering of multivalent MHC class II-peptide chimeras. Protein Eng. 14, 195–200 (2001).
Casares, S. et al. Down-regulation of diabetogenic CD4+ T cells by a soluble dimeric peptide-MHC class II chimera. Nat. Immunol. 3, 383–391 (2002).
Hackett, C.J. & Sharma, O.K. Frontiers in peptide-MHC class II multimer technology. Nat. Immunol. 3, 887–889 (2002).
Manz, R., Assenmacher, M., Pfluger, E., Miltenyi, S. & Radbruch, A. Analysis and sorting of live cells according to secreted molecules, relocated to a cell-surface affinity matrix. Proc. Natl. Acad. Sci. USA 92, 1921–1925 (1995).
Waldrop, S.L., Pitcher, C.J., Peterson, D.M., Maino, V.C. & Picker, L.J. Determination of antigen-specific memory/effector CD4+ T cell frequencies by flow cytometry: evidence for a novel, antigen-specific homeostatic mechanism in HIV-associated immunodeficiency. J. Clin. Invest. 99, 1739–1750 (1997).
Suni, M.A., Picker, L.J. & Maino, V.C. Detection of antigen-specific T cell cytokine expression in whole blood by flow cytometry. J. Immunol. Methods 212, 89–98 (1998).
Brosterhus, H. et al. Enrichment and detection of live antigen-specific CD4(+) and CD8(+) T cells based on cytokine secretion. Eur. J. Immunol. 29, 4053–4059 (1999).
Maino, V.C., Suni, M.A. & Ruitenberg, J.J. Rapid flow cytometric method for measuring lymphocyte subset activation. Cytometry 20, 127–133 (1995).
Kahi, S. et al. A rapid flow cytometric method to explore cellular immunity against Toxoplasma gondii in humans. Clin. Diagn. Lab. Immunol. 5, 745–748 (1998).
Michalek, J. et al. Definitive separation of graft-versus-leukemia- and graft-versus-host-specific CD4+ T cells by virtue of their receptor beta loci sequences. Proc. Natl. Acad. Sci. USA 100, 1180–1184 (2003).
Thiel, A., Scheffold, A. & Radbruch, A. Antigen-specific cytometry–new tools arrived!. Clin. Immunol. 111, 155–161 (2004).
Armitage, R.J. et al. Molecular and biological characterization of a murine ligand for CD40. Nature 357, 80–82 (1992).
Graf, D., Korthauer, U., Mages, H.W., Senger, G. & Kroczek, R.A. Cloning of TRAP, a ligand for CD40 on human T cells. Eur. J. Immunol. 22, 3191–3194 (1992).
Yellin, M.J. et al. CD40 molecules induce down-modulation and endocytosis of T cell surface T cell-B cell activating molecule/CD40-L. Potential role in regulating helper effector function. J. Immunol. 152, 598–608 (1994).
Misumi, Y., Miki, K., Takatsuki, A., Tamura, G. & Ikehara, Y. Novel blockade by brefeldin A of intracellular transport of secretory proteins in cultured rat hepatocytes. J. Biol. Chem. 261, 11398–11403 (1986).
Choi, Y.W. et al. Interaction of Staphylococcus aureus toxin “superantigens” with human T cells. Proc. Natl. Acad. Sci. USA 86, 8941–8945 (1989).
Hong, S.C., Waterbury, G. & Janeway, C.A., Jr. Different superantigens interact with distinct sites in the Vbeta domain of a single T cell receptor. J. Exp. Med. 183, 1437–1446 (1996).
Murphy, K.M., Heimberger, A.B. & Loh, D.Y. Induction by antigen of intrathymic apoptosis of CD4+CD8+TCRlo thymocytes in vivo. Science 250, 1720–1723 (1990).
Lindgren, H., Axcrona, K. & Leanderson, T. Regulation of transcriptional activity of the murine CD40 ligand promoter in response to signals through TCR and the costimulatory molecules CD28 and CD2. J. Immunol. 166, 4578–4585 (2001).
Stinski, M.F. Sequence of protein synthesis in cells infected by human cytomegalovirus: early and late virus-induced polypeptides. J. Virol. 26, 686–701 (1978).
Wills, M.R. et al. The human cytotoxic T-lymphocyte (CTL) response to cytomegalovirus is dominated by structural protein pp65: frequency, specificity, and T-cell receptor usage of pp65-specific CTL. J. Virol. 70, 7569–7579 (1996).
Beninga, J., Kropff, B. & Mach, M. Comparative analysis of fourteen individual human cytomegalovirus proteins for helper T cell response. J. Gen. Virol. 76, 153–160 (1995).
Czerkinsky, C.C., Nilsson, L.A., Nygren, H., Ouchterlony, O. & Tarkowski, A. A solid-phase enzyme-linked immunospot (ELISPOT) assay for enumeration of specific antibody-secreting cells. J. Immunol. Methods 65, 109–121 (1983).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Ludewig, B., Henn, V., Schroder, J.M., Graf, D. & Kroczek, R.A. Induction, regulation, and function of soluble TRAP (CD40 ligand) during interaction of primary CD4+ CD45RA+ T cells with dendritic cells. Eur. J. Immunol. 26, 3137–3143 (1996).
Acknowledgements
We thank A. Radbruch for critical discussion and U. Klein for critical reading of the manuscript. We thank Q. Phan, F. Kellndorfer and D. Huscher for technical help. This work was supported by the European Sixth Framework Program, grants LSHP-CT-2003-503240, LSHG-CT-2005-005203 and LSHB-CT-2004-512074, grants PTJ 0311597 and PTJ 0312102 (all grants were to A.T. and A.S.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Combined qualitative and quantitative assessment of Th cells specific for different antigens. (PDF 872 kb)
Rights and permissions
About this article
Cite this article
Frentsch, M., Arbach, O., Kirchhoff, D. et al. Direct access to CD4+ T cells specific for defined antigens according to CD154 expression. Nat Med 11, 1118–1124 (2005). https://doi.org/10.1038/nm1292
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1292
This article is cited by
-
Immune-checkpoint expression in antigen-presenting cells (APCs) of cytomegaloviruses infection after transplantation: as a diagnostic biomarker
Archives of Microbiology (2023)
-
CD4 T cell epitope abundance in ferritin core potentiates responses to hemagglutinin nanoparticle vaccines
npj Vaccines (2022)
-
Interleukin-1 promotes autoimmune neuroinflammation by suppressing endothelial heme oxygenase-1 at the blood–brain barrier
Acta Neuropathologica (2020)
-
SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19
Nature (2020)
-
Strategies of adoptive T -cell transfer to treat refractory viral infections post allogeneic stem cell transplantation
Journal of Hematology & Oncology (2019)