Abstract
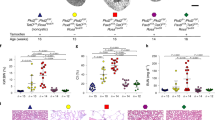
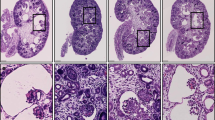
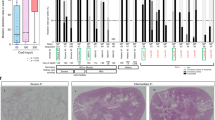
Autosomal dominant polycystic kidney disease is an important cause of end-stage renal disease, for which there is no proven therapy1. Mutations in PKD1 (the gene encoding polycystin-1) are the principal cause of this disease. The disease begins in utero2 and is slowly progressive, but it is not known whether cystogenesis is an ongoing process during adult life. We now show that inactivation of Pkd1 in mice before postnatal day 13 results in severely cystic kidneys within 3 weeks, whereas inactivation at day 14 and later results in cysts only after 5 months. We found that cellular proliferation was not appreciably higher in cystic specimens than in age-matched controls, but the abrupt change in response to Pkd1 inactivation corresponded to a previously unrecognized brake point during renal growth and significant changes in gene expression. These findings suggest that the effects of Pkd1 inactivation are defined by a developmental switch that signals the end of the terminal renal maturation process. Our studies show that Pkd1 regulates tubular morphology in both developing and adult kidney, but the pathologic consequences of inactivation are defined by the organ's developmental status. These results have important implications for clinical understanding of the disease and therapeutic approaches.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Boucher, C. & Sandford, R. Autosomal dominant polycystic kidney disease (ADPKD, MIM 173900, PKD1 and PKD2 genes, protein products known as polycystin-1 and polycystin-2). Eur. J. Hum. Genet. 12, 347–354 (2004).
Pretorius, D.H. et al. Diagnosis of autosomal dominant polycystic kidney disease in utero and in the young infant. J. Ultrasound Med. 6, 249–255 (1987).
Piontek, K.B. et al. A functional floxed allele of Pkd1 that can be conditionally inactivated in vivo. J. Am. Soc. Nephrol. 15, 3035–3043 (2004).
Lu, W. et al. Perinatal lethality with kidney and pancreas defects in mice with a targetted Pkd1 mutation. Nat. Genet. 17, 179–181 (1997).
Bukanov, N.O., Smith, L.A., Klinger, K.W., Ledbetter, S.R. & Ibraghimov-Beskrovnaya, O. Long-lasting arrest of murine polycystic kidney disease with CDK inhibitor roscovitine. Nature 444, 949–952 (2006).
Lanoix, J., D'Agati, V., Szabolcs, M. & Trudel, M. Dysregulation of cellular proliferation and apoptosis mediates human autosomal dominant polycystic kidney disease (ADPKD). Oncogene 13, 1153–1160 (1996).
Woo, D. Apoptosis and loss of renal tissue in polycystic kidney diseases. N. Engl. J. Med. 333, 18–25 (1995).
Ashburner, M. et al. Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat. Genet. 25, 25–29 (2000).
Chotteau-Lelievre, A. et al. PEA3 transcription factors are expressed in tissues undergoing branching morphogenesis and promote formation of duct-like structures by mammary epithelial cells in vitro. Dev. Biol. 259, 241–257 (2003).
Grantham, J.J., Geiser, J.L. & Evan, A.P. Cyst formation and growth in autosomal dominant polycystic kidney disease. Kidney Int. 31, 1145–1152 (1987).
Nadasdy, T. et al. Proliferative activity of cyst epithelium in human renal cystic diseases. J. Am. Soc. Nephrol. 5, 1462–1468 (1995).
Chang, M.Y. et al. Haploinsufficiency of Pkd2 is associated with increased tubular cell proliferation and interstitial fibrosis in two murine Pkd2 models. Nephrol. Dial. Transplant. 21, 2078–2084 (2006).
Shillingford, J.M. et al. The mTOR pathway is regulated by polycystin-1, and its inhibition reverses renal cystogenesis in polycystic kidney disease. Proc. Natl. Acad. Sci. USA 103, 5466–5471 (2006).
Sweeney, W.E. Jr. et al. Combination treatment of PKD utilizing dual inhibition of EGF-receptor activity and ligand bioavailability. Kidney Int. 64, 1310–1319 (2003).
Muskhelishvili, L., Latendresse, J.R., Kodell, R.L. & Henderson, E.B. Evaluation of cell proliferation in rat tissues with BrdU, PCNA, Ki-67 (MIB-5) immunohistochemistry and in situ hybridization for histone mRNA. J. Histochem. Cytochem. 51, 1681–1688 (2003).
Simons, M. et al. Inversin, the gene product mutated in nephronophthisis type II, functions as a molecular switch between Wnt signaling pathways. Nat. Genet. 37, 537–543 (2005).
Fischer, E. et al. Defective planar cell polarity in polycystic kidney disease. Nat. Genet. 38, 21–23 (2006).
Ciruna, B., Jenny, A., Lee, D., Mlodzik, M. & Schier, A.F. Planar cell polarity signalling couples cell division and morphogenesis during neurulation. Nature 439, 220–224 (2006).
Torres, V.E. et al. Effective treatment of an orthologous model of autosomal dominant polycystic kidney disease. Nat. Med. 10, 363–364 (2004).
Wu, G. et al. Somatic inactivation of Pkd2 results in polycystic kidney disease. Cell 93, 177–188 (1998).
Osathanondh, V. & Potter, E.L. Pathogenesis of polycystic kidneys. Historical Survey. Arch. Pathol. 77, 459–465 (1964).
Heggo, O. A microdissection study of cystic disease of the kidneys in adults. J. Pathol. Bacteriol. 91, 311–315 (1966).
Baert, L. Hereditary polycystic kidney disease (adult form): a microdissection study of two cases at an early stage of the disease. Kidney Int. 13, 519–525 (1978).
Calvet, J.P. Injury and development in polycystic kidney disease. Curr. Opin. Nephrol. Hypertens. 3, 340–348 (1994).
Nauli, S.M. et al. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat. Genet. 33, 129–137 (2003).
Davenport, J.R. et al. Disruption of intraflagellar transport in adult mice leads to obesity and slow-onset cystic kidney disease. Curr. Biol. 17, 1586–1594 (2007).
Gentleman, R.C. et al. Bioconductor: open software development for computational biology and bioinformatics. Genome Biol. 5, R80 (2004).
Smyth, G. LIMMA: linear models for microarray data. in Bioinformatics and Computational Biology Solutions Using R and Bioconductor (Eds. Gentleman, R., Carey, V.J., Huber, W., Irizarry, R.A., Dudoit, S.) 397–420 (Springer, New York City, 2005).
Stegmann, A. et al. Metabolome, transcriptome, and bioinformatic cis-element analyses point to HNF-4 as a central regulator of gene expression during enterocyte differentiation. Physiol. Genomics 27, 141–155 (2006).
Dennis, G., Jr. et al. DAVID: database for annotation, visualization, and integrated discovery. Genome Biol. 4, P3 (2003).
Acknowledgements
This work was supported by grants from the US National Institutes of Health (DK48006, DK51259) and the US National Kidney Foundation (L.F.M.). G.G.G. is the Irving Blum Scholar of the Johns Hopkins University School of Medicine. The authors wish to thank members of the Germino, Watnick, Montell, Qian and Sutters laboratories (Johns Hopkins University School of Medicine), A. Tousson (University of Alabama at Birmingham) and S. Kuo (Johns Hopkins University School of Medicine) for helpful advice, C. Cheadle (Johns Hopkins Bayview Medical Campus Genomics Core) for assistance with the microarray studies, and M. Knepper (US National Institutes of Health) and J. Hoyer (Children's Hospital of Philadelphia) for the antibodies to aquaporin-2 and Tamm-Horsfall glycoprotein, respectively.
Author information
Authors and Affiliations
Contributions
K.P. designed and performed the experiments and contributed to the writing of the manuscript. L.F.M. assisted in the experimental design, helped to analyze the histopathology, performed the microarray analyses and assisted in the writing of the manuscript. M.A.G.-G. helped perform the experiments and contributed conceptually to the project. D.L.H. helped with the histopathology. G.G.G. directed the project and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–3 and Supplementary Table 1 (PDF 1484 kb)
Supplementary Tables 2–5
Supplementary Tables 2–5 (XLS 220 kb)
Rights and permissions
About this article
Cite this article
Piontek, K., Menezes, L., Garcia-Gonzalez, M. et al. A critical developmental switch defines the kinetics of kidney cyst formation after loss of Pkd1. Nat Med 13, 1490–1495 (2007). https://doi.org/10.1038/nm1675
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1675
This article is cited by
-
Genetic interactions between polycystin-1 and Wwtr1 in osteoblasts define a novel mechanosensing mechanism regulating bone formation in mice
Bone Research (2023)
-
Mechanism of cystogenesis by Cd79a-driven, conditional mTOR activation in developing mouse nephrons
Scientific Reports (2023)
-
Loss of Pkd1 limits susceptibility to colitis and colorectal cancer
Oncogenesis (2023)
-
Dehydration Accelerates Cytogenesis and Cyst Growth in Pkd1−/− Mice by Regulating Macrophage M2 Polarization
Inflammation (2023)
-
Rapid target validation in a Cas9-inducible hiPSC derived kidney model
Scientific Reports (2021)