Abstract
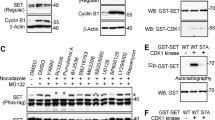
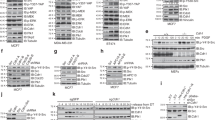
The cyclin-dependent kinase inhibitor p27kip1 is a putative tumor suppressor for human cancer. The mechanism underlying p27kip1 deregulation in human cancer is, however, poorly understood. We demonstrate that the serine/threonine kinase Akt regulates cell proliferation in breast cancer cells by preventing p27kip1-mediated growth arrest. Threonine 157 (T157), which maps within the nuclear localization signal of p27kip1, is a predicted Akt-phosphorylation site. Akt-induced T157 phosphorylation causes retention of p27kip1 in the cytoplasm, precluding p27kip1-induced G1 arrest. Conversely, the p27kip1-T157A mutant accumulates in cell nuclei and Akt does not affect p27kip1–T157A-mediated cell cycle arrest. Lastly, T157-phosphorylated p27kip1 accumulates in the cytoplasm of primary human breast cancer cells coincident with Akt activation. Thus, cytoplasmic relocalization of p27kip1, secondary to Akt-mediated phosphorylation, is a novel mechanism whereby the growth inhibitory properties of p27kip1 are functionally inactivated and the proliferation of breast cancer cells is sustained.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Morgan, D.O. Principles of Cdk regulation. Nature 374, 131–134 (1995).
Sherr, C.J. & Roberts, J.M. CDK inhibitors: Positive and negative regulators of G1-phase progression. Genes Dev. 13, 1501–1512 (1999).
Hanahan, D. & Weinberg, R.A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Sherr, C.J. Cancer cell cycles. Science 274, 1672–1677 (1996).
Hunter, T. & Pines, J. Cyclins and cancer. II: Cyclin D and CDK inhibitors come of age. Cell 79, 573–582 (1994).
Weinstat-Saslow, D. et al. Overexpression of cyclin D mRNA distinguishes invasive and in situ breast carcinomas from non-malignant lesions. Nature Med. 1, 1257–1260 (1995).
Porter, P.L. et al. Expression of cell-cycle regulators p27kip1 and cyclin E, alone and in combination, correlate with survival in young breast cancer patients. Nature Med. 3, 222–225 (1997).
Baldassarre, B. et al. Overexpressed cyclin D3 contributes to retaining the growth inhibitor p27 in the cytoplasm of thyroid tumor cells. J. Clin. Invest. 10, 865–874 (1999).
Rocco, J.W. & Sidransky, D. p16(MTS-1/CDKN2/INK4a) in cancer progression. Exp. Cell Res. 264, 42–55 (2001).
Otsuki, T., Clark, H.M., Wellmann, A., Jaffe, E.S. & Raffeld, M. Involvement of CDKN2 p16INK4A/MTS1) and p15INK4B/MTS2 in human leukemias and lymphomas. Cancer Res. 55, 1436–1440 (1995).
Sgambato, A., Cittadini, A., Faraglia, B. & Weinstein, I.B. Multiple functions of p27Kip1 and its alterations in tumor cells: A review. J. Cell Physiol. 183, 18–27 (2000).
Fero, M.L., Randel, E., Gurley, K.E., Roberts, J.M. & Kemp, C.J. The murine gene p27Kip1 is haplo-insufficient for tumor suppression. Nature 396, 177–180 (1998).
Catzavelos, C. et al. Decreased levels of the cell-cycle inhibitor p27Kip1 protein: Prognostic implications in primary breast cancer. Nature Med. 3, 227–230 (1997).
Lloyd, R.V., Jin, L., Qian, X. & Kulig, E. Aberrant p27Kip1 expression in endocrine and other tumors. Am. J. Pathol. 150, 401–407 (1997).
Loda, M. et al. Increased proteasome-dependent degradation of the cyclin-dependent kinase inhibitor p27Kip1 in aggressive colorectal carcinomas. Nature Med. 3, 231–234 (1997).
Ciaparrone, M. et al. Localization and expression of p27Kip1 in multistage colorectal carcinogenesis. Cancer Res. 58, 114–122 (1998).
Singh, S. et al. Loss or altered subcellular localization of p27Kip1 in Barrett's associated adenocarcinoma. Cancer Res. 58, 1730–1735 (1998).
Jiang, Y., Zhao, R.C. & Verfaillie, C.M. Abnormal integrin-mediated regulation of chronic myelogenous leukemia CD34+ cell proliferation: BCR/ABL upregulates the cyclin-dependent kinase inhibitor, p27Kip1, which is relocated to the cell cytoplasm and incapable of regulating cdk2 activity. Proc. Natl. Acad. Sci. USA 97, 10538–10543 (2000).
Soucek, T., Yeung, R.S. & Hengstschlager, M. Inactivation of the cyclin-dependent kinase inhibitor p27Kip1 upon loss of the tuberous sclerosis complex gene-2. Proc. Natl. Acad. Sci. USA 95, 15653–15658 (1998).
Orend, G., Hunter, T. & Ruoslahti, E. Cytoplasmic displacement of cyclin E-cdk2 inhibitors p21 and p27 in anchorage-independent cells. Oncogene 16, 2575–2583 (1998).
Yaroslavskiy, B., Watkins, S., Donnenberg, A.D., Patton, T.J. & Steinman, R.A. Subcellular and cell-cycle expression profiles of CDK-inhibitors in normal differentiating myeloid cells. Blood 93, 2907–2917 (1999).
Tomoda, K., Kubota, Y. & Kato, J. Degradation of the cyclin-dependent-kinase inhibitor p27Kip1 is instigated by Jab1. Nature 398, 160–165 (1999).
Testa, J.R. & Bellacosa, A. AKT plays a central role in tumorigenesis. Proc. Natl. Acad. Sci. USA 98, 10983–10985 (2001).
Datta, S.R., Brunet, A. & Greenberg, M.E. Cellular survival: A play in three Akts. Genes Dev. 13, 2905–2927 (1999).
Datta, S.R. et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91, 231–241 (1997).
Cardone, M.H. et al. Regulation of cell death protease caspase-9 by phosphorylation. Science 282, 1318–1321 (1998).
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999).
Kops, G.J. et al. Direct control of the forkhead transcription factor AFX by protein kinase B. Nature 398, 630–634 (1999).
Zhou, B.P. et al. (2001) Cytoplasmic localization of p21Cip1/WAF1 by Akt-induced phosphorylation in HER-2/neu-overexpressing cells. Nature Cell Biol. 3, 245–252 (2001).
Coffer, P.J., Jin, J. & Woodgett, J.R. Protein kinase B (c-Akt): A multifunctional mediator of phosphatidylinositol 3-kinase activation. Biochem. J. 335, 1–12 (1998).
Di Cristofano, A. & Pandolfi, P.P. The multiple roles of PTEN in tumor suppression. Cell 100, 387–390 (2000).
Lobenhofer, E.K., Huper, G., Iglehart, J.D. & Marks, J.R. Inhibition of mitogen-activated protein kinase and phosphatidylinositol 3-kinase activity in MCF-7 cells prevents estrogen-induced mitogenesis. Cell Growth Differ. 11, 99–110 (2000).
Tsai, E.M, Wang, S.C., Lee, J.N. & Hung, MC . Akt Activation by estrogen in estrogen receptor-negative breast cancer cells. Cancer Res. 61, 8390–8392 (2001).
Lenferink, A.E., Busse, D., Flanagan, W.M., Yakes, F.M. & Arteaga, C.L. ErbB2/neu kinase modulates cellular p27Kip1 and cyclin D1 through multiple signaling pathways. Cancer Res. 61, 6583–6591 (2001).
Yang, H.Y., Shao, R., Hung, M.C. & Lee, M.H. p27Kip1 inhibits HER2/neu-mediated cell growth and tumorigenesis. Oncogene 20, 3695–3702 (2001).
Downward, J. Mechanisms and consequences of activation of protein kinase B/Akt. Curr. Opin. Cell. Biol. 10, 262–267 (1998).
Sun, M. et al. AKT1/PKBα kinase is frequently elevated in human cancers and its constitutive activation is required for oncogenic transformation in NIH3T3 cells. Am. J. Pathol. 159, 431–437 (2001).
Diehl, J.A., Cheng, M., Roussel, M.F. & Sherr, C.J. Glycogen synthase kinase-3β regulates cyclin D1 proteolysis and subcellular localization. Genes Dev. 12, 3499–3511 (1998).
Alt, J.R., Cleveland, J.L., Hannink, M. & Diehl, J.A. Phosphorylation-dependent regulation of cyclin D1 nuclear export and cyclin D1-dependent cellular transformation. Genes Dev. 14, 3102–3114 (2000).
Liu, X. et al. Disruption of TGF-β growth inhibition by oncogenic ras is linked to p27Kip1 mislocalization. Oncogene 19, 5926–5935 (2000).
Bellacosa, A. et al. Akt activation by growth factors is a multiple-step process: The role of the PH domain. Oncogene 17, 313–325 (1998).
Baldassarre, G. et al. Key role of the cyclin-dependent kinase inhibitor p27kip1 for embryonal carcinoma cell survival and differentiation. Oncogene 18, 6241–6251 (1999).
Bruni, P. et al. PTEN expression is reduced in a subset of sporadic thyroid carcinomas: Evidence that PTEN-growth suppressing activity in thyroid cancer cells is mediated by p27kip1. Oncogene 19, 3146–3155 (2000).
Polyak, K., Lee, M., Koff, A., Roberts, J.M. & Massaguè, J. Cloning of p27Kip1 a cyclin-dependent kinase inhibitor and potential mediator of extracellular antimitogenic signals. Cell 78, 59–66 (1994).
Acknowledgements
We thank G. Baldassarre and M. Napolitano for flow-cytometry analysis; G. Tallini for help in the statistical analysis; and J.A. Gilder for editing the text. This work was supported by grants from the Associazione Italiana Ricerca sul Cancro (AIRC), Agenzia 2000 of the Consiglio Nazionale delle Ricerche (CNR) and the Progetto Finalizzato Biotecnologie of the CNR. M.L.M. and F.V. are supported by FIRC fellowships. P.B. was the recipient of a BIOGEM fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Viglietto, G., Motti, M., Bruni, P. et al. Cytoplasmic relocalization and inhibition of the cyclin-dependent kinase inhibitor p27Kip1 by PKB/Akt-mediated phosphorylation in breast cancer. Nat Med 8, 1136–1144 (2002). https://doi.org/10.1038/nm762
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm762
This article is cited by
-
TRIM58 functions as a tumor suppressor in colorectal cancer by promoting RECQL4 ubiquitination to inhibit the AKT signaling pathway
World Journal of Surgical Oncology (2023)
-
SUZ12 inhibition attenuates cell proliferation of glioblastoma via post-translational regulation of CDKN1B
Genes & Genomics (2023)
-
Inhibition of secretory leukocyte protease inhibitor (SLPI) promotes the PUMA-mediated apoptosis and chemosensitivity to cisplatin in colorectal cancer cells
Discover Oncology (2023)
-
Enhanced pro-apoptosis gene signature following the activation of TAp63α in oocytes upon γ irradiation
Cell Death & Disease (2022)
-
Bacterial protein MakA causes suppression of tumour cell proliferation via inhibition of PIP5K1α/Akt signalling
Cell Death & Disease (2022)