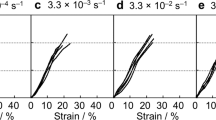
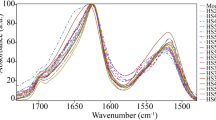
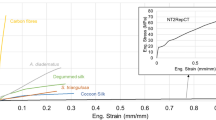
Abstract
Spider silks possess nature’s most exceptional mechanical properties, with unrivalled extensibility and high tensile strength. Unfortunately, our understanding of silks is limited because the complete elastic response has never been measured—leaving a stark lack of essential fundamental information. Using non-invasive, non-destructive Brillouin light scattering, we obtain the entire stiffness tensors (revealing negative Poisson’s ratios), refractive indices, and longitudinal and transverse sound velocities for major and minor ampullate spider silks: Argiope aurantia, Latrodectus hesperus, Nephila clavipes, Peucetia viridans. These results completely quantify the linear elastic response for all possible deformation modes, information unobtainable with traditional stress–strain tests. For completeness, we apply the principles of Brillouin imaging to spatially map the elastic stiffnesses on a spider web without deforming or disrupting the web in a non-invasive, non-contact measurement, finding variation among discrete fibres, junctions and glue spots. Finally, we provide the stiffness changes that occur with supercontraction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cranford, S. W., Tarakanova, A., Pugno, N. M. & Buehler, M. J. Nonlinear material behavior of spider silk yields robust webs. Nature 482, 72–76 (2012).
Keten, S., Xu, Z., Ihle, B. & Buehler, M. J. Nanoconfinement controls stiffness, strength and mechanical toughness of β-sheet crystals in silk. Nature Mater. 9, 359–367 (2010).
Vollrath, F. Silk evolution untangled. Nature 466, 319 (2010).
Vollrath, F. Spider webs and silks. Sci. Am. 266, 70–76 (1992).
Work, R. W. Viscoelastic behaviour and wet supercontraction of major ampullate silk fibres of certain orb-web-building spiders (Araneae). J. Exp. Biol. 118, 379–404 (1985).
Blackledge, T. A., Swindeman, J. E. & Hayashi, C. Y. Quasistatic and continuous dynamic characterization of the mechanical properties of silk from the cobweb of the black widow spider Latrodectus hesperus. J. Exp. Biol. 208, 1937–1949 (2005).
Gosline, J. M., Guerette, P. A., Ortlepp, C. S. & Savage, K. N. The mechanical design of spider silks: from fibroin sequence to mechanical function. J. Exp. Biol. 202, 3295–3303 (1999).
Blackledge, T. A., Cardullo, R. A. & Hayashi, C. Y. Polarized light microscopy, variability in spider silk diameters, and the mechanical characterization of spider silk. Invertebr. Biol. 124, 165–173 (2005).
Liu, Y., Shao, Z. & Vollrath, F. Relationships between supercontraction and mechanical properties of spider silk. Nature Mater. 4, 901–905 (2005).
Emile, O., Le Floch, A. & Vollrath, F. Biopolymers: Shape memory in spider draglines. Nature 440, 621 (2006).
Eisoldt, L., Smith, A. & Scheibel, T. Decoding the secrets of spider silk. Mater. Today 14, 80–86 (March, 2011).
Omenetto, F. G. & Kaplan, D. L. New opportunities for an ancient material. Science 329, 528–531 (2010).
Speziale, S. et al. Sound velocity and elasticity of tetragonal lysozyme crystals by Brillouin spectroscopy. Biophys. J. 85, 3202–3213 (2003).
Randall, S. J. & Vaughan, J. M. Brillouin scattering in systems of biological significance [and discussion]. Phil. Trans. R. Soc. Lond. A 293, 341–348 (1979).
Cusack, S. & Miller, A. Determination of the elastic constants of collagen by Brillouin light scattering. J. Mol. Biol. 135, 39–51 (1979).
Harley, R., James, D., Miller, A. & White, J. W. Phonons and the elastic moduli of collagen and muscle. Nature 267, 285–287 (1977).
Berovic, N., Thomas, N., Thornhill, R. A. & Vaughan, J. M. Observation of Brillouin scattering from single muscle fibres. Eur. Biophys. J. 17, 69–74 (1989).
Koski, K. J. & Yarger, J. L. Brillouin imaging. Appl. Phys. Lett. 87, 061903 (2005).
Arecchi, F. T. et al. Laser Handbook (North-Holland, 1972).
Saravanan, D. Spider silk—structure, properties and spinning. J. Text. Apparel Technol. Manage. 5, 1–20 (2006).
Work, R. W. Dimensions, birefringences, and force-elongation behavior of major and minor ampullate silk fibers from orb-web-spinning spiders—the effects of wetting on these properties. Tex. Res. J. 47, 650–662 (1977).
Zemlin, J. C. Technical Report 69-29-CM: A Study of the Mechanical Behavior of Spider Silks (Collaborative Research, 1968).
Vincent, J. F. V. Structural Materials 1–378 (Macmillan, 1982).
Fossey, S. A. & Kaplan, D. L. in Polymer Data Handbook (ed. Mark, J. E.) (Oxford Univ. Press, 1999).
Little, D. J. & Kane, D. M. Hybrid immersion-polarization method for measuring birefringence applied to spider silks. Opt. Lett. 36, 4098–4100 (2011).
Ko, F. K. & Jovicic, J. Modeling of mechanical properties and structural design of spider web. Biomacromolecules 5, 780–785 (2004).
Boutry, C., Rezac, M. & Blackledge, T. A. Plasticity in major ampullate silk production in relation to spider phylogeny and ecology. PLOS ONE 6, e22467 (2011).
Heim, M., Keerl, D. & Scheibel, T. Spider silk: From soluble protein to extraordinary fiber. Angew. Chem. Int. Ed. 48, 3584–3596 (2009).
Moore, A. M. F. & Tran, K. Material properties of cobweb silk from the black widow spider Latrodectus hesperus. Int. J. Biol. Macromol. 24, 277–282 (1999).
Nova, A., Keten, S., Pugno, N. M., Redaelli, A. & Buehler, M. J. Molecular and nanostructural mechanisms of deformation, strength, and toughness of spider silk fibrils. Nano Lett. 10, 2626–2634 (2010).
Denny, M. The physical properties of spider’s silk and their role in the design of orb-webs. J. Exp. Biol. 65, 483–506 (1976).
Gosline, J. M., Demont, M. E. & Denny, M. W. The structure and properties of spider silk. Endeavour 10, 37–43 (1986).
Vollrath, F. Biology of spider silk. Int. J. Biol. Macromol. 24, 81–88 (1999).
Sahni, V., Blackledge, T. A. & Dhinojwala, A. Viscoelastic solids explain spider web stickiness. Nature Commun. 1, 1–4 (2010).
Blackledge, T. A., Summers, A. P. & Hayashi, C. Y. Gumfooted lines in black widow cobwebs and the mechanical properties of spider capture silk. Zoology 108, 41–46 (2005).
Sahni, V., Blackledge, T. A. & Dhinojwala, A. Changes in the adhesive properties of spider aggregate glue during the evolution of cobwebs. Sci. Rep. 1, 1–41 (2011).
Xu, M. & Lewis, R. M. Structure of a protein superfiber: Spider dragline silk. Proc. Natl Acad. Sci. USA 87, 7120–7124 (1990).
Bell, F. I., McEwen, I. J. & Viney, C. Fibre science: Supercontraction stress in wet spider dragline. Nature 416, 37 (2002).
Blackledge, T. A. et al. How super is supercontraction? Persistent versus cyclic responses to humidity in spider dragline silk. J. Exp. Biol. 212, 1981–1989 (2009).
Van Beek, J. D., Hess, S., Vollrath, F. & Meier, B. H. The molecular structure of spider dragline silk: Folding and orientation of the protein backbone. Proc. Natl Acad. Sci. USA 99, 10266–10271 (2002).
Simmons, A. H., Michal, C. A. & Jelinski, L. W. Molecular orientation and two-component nature of the crystalline fraction of spider dragline silk. Science 271, 84–87 (1996).
Eles, P.T. & Michal, C. A. Strain dependent local phase transitions observed during controlled supercontraction reveal mechanisms in spider silk. Macromolecules 37, 1342–1345 (2004).
Boutry, C. & Blackledge, T. A. Evolution of supercontraction in spider silk: structure function relationship from tarantulas to orb-weavers. J. Exp. Biol. 213, 3505–3514 (2010).
Guinea, G. V., Elices, M., Prez-Rigueiro, J. & Plaza, G. R. Stretching of supercontracted fibers: A link between spinning and the variability of spider silk. J. Exp. Biol. 208, 25–30 (2005).
Savage, K. N., Guerette, P. A. & Gosline, J. M. Supercontraction stress in spider webs. Biomacromolecules 5, 675–679 (2004).
Work, R. W. A comparative study of the supercontraction of major ampullate silk fibers of orb-web-building spiders (Araneae). J. Arachnol. 9, 299–308 (1981).
Fossey, S. A. & Kaplan, D. L. Polymer Data Handbook 970–974 (Oxford Univ. Press, 1999).
Work, R. W. & Emerson, P. D. J. An apparatus and technique for the forcible silking of spiders. J. Arachnol. 10, 1–10 (1982).
Fedorov, F. I. Theory of Elastic Waves in Crystals (Plenum, 1958).
Nye, J. F. Physical Properties of Crystals 144–145 (Clarendon, 1957).
Acknowledgements
J.L.Y. would like to acknowledge spider silk research support from the Department of Defense, AFOSR (FA9550-10-1-0275) and the US National Science Foundation (CHE-1011937).
Author information
Authors and Affiliations
Contributions
K.J.K. and J.L.Y. designed the experiments and wrote this manuscript. P.A. and J.L.Y. provided samples. K.J.K., P.A., K.M. and J.L.Y. performed the experiments. K.J.K. analysed the results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 660 kb)
Rights and permissions
About this article
Cite this article
Koski, K., Akhenblit, P., McKiernan, K. et al. Non-invasive determination of the complete elastic moduli of spider silks. Nature Mater 12, 262–267 (2013). https://doi.org/10.1038/nmat3549
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3549
This article is cited by
-
Brillouin light scattering anisotropy microscopy for imaging the viscoelastic anisotropy in living cells
Nature Photonics (2024)
-
Pulsed stimulated Brillouin microscopy enables high-sensitivity mechanical imaging of live and fragile biological specimens
Nature Methods (2023)
-
High-resolution line-scan Brillouin microscopy for live imaging of mechanical properties during embryo development
Nature Methods (2023)
-
Tunable and light-weight phononic lattices inspired by spider capture silk
Applied Physics A (2023)
-
Measuring mechanical anisotropy of the cornea with Brillouin microscopy
Nature Communications (2022)