Abstract
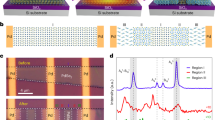
To be able to control the functions of engineered multicomponent nanomaterials, a detailed understanding of heterogeneous nucleation at the nanoscale is essential. Here, by using in situ synchrotron X-ray scattering, we show that in the heterogeneous nucleation and growth of Au on Pt or Pt-alloy seeds the heteroepitaxial growth of the Au shell exerts high stress (∼2 GPa) on the seed by forming a core/shell structure in the early stage of the reaction. The development of lattice strain and subsequent strain relaxation, which we show using atomic-resolution transmission electron microscopy to occur through the slip of {111} layers, induces morphological changes from a core/shell to a dumbbell structure, and governs the nucleation and growth kinetics. We also propose a thermodynamic model for the nucleation and growth of dumbbell metallic heteronanostructures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tada, H., Mitsui, T., Kiyonaga, T., Akita, T. & Tanaka, K. All-solid-state Z-scheme in CdS–Au–TiO2 three-component nanojunction system. Nature Mater. 5, 782–786 (2006).
Amirav, L. & Alivisatos, A. P. Luminescence studies of individual quantum dot photocatalysts. J. Am. Chem. Soc. 135, 13049–13053 (2013).
Si, R. & Flytzani-Stephanopoulos, M. Shape and crystal-plane effects of nanoscale ceria on the activity of Au–CeO2 catalysts for the water–gas shift reaction. Angew. Chem. Int. Ed. 47, 2884–2887 (2008).
Lee, Y., Garcia, M. A., Huls, N. A. F. & Sun, S. Synthetic tuning of the catalytic properties of Au-Fe3O4 nanoparticles. Angew. Chem. Int. Ed. 49, 1271–1274 (2010).
Lim, B. et al. Pd–Pt bimetallic nanodendrites with high activity for oxygen reduction. Science 324, 1302–1305 (2009).
Yamada, Y. et al. Nanocrystal bilayer for tandem catalysis. Nature Chem. 3, 372–376 (2011).
Wang, L., Nemoto, Y. & Yamauchi, Y. Direct synthesis of spatially-controlled Pt-on-Pd bimetallic nanodendrites with superior electrocatalytic activity. J. Am. Chem. Soc. 133, 9674–9677 (2011).
Zhang, H., Watanabe, T., Okumura, M., Haruta, M. & Toshima, N. Catalytically highly active top gold atom on palladium nanocluster. Nature Mater. 11, 49–52 (2012).
Cui, C., Gan, L., Heggen, M., Rudi, S. & Strasser, P. Compositional segregation in shaped Pt alloy nanoparticles and their structural behaviour during electrocatalysis. Nature Mater. 12, 765–771 (2013).
Wu, H. et al. Formation of heterodimer nanocrystals: UO2/In2O3 and FePt/In2O3 . J. Am. Chem. Soc. 133, 14327–14337 (2011).
Costi, R., Saunders, A. E. & Banin, U. Colloidal hybrid nanostructures: A new type of functional materials. Angew. Chem. Int. Ed. 49, 4878–4897 (2010).
Carbone, L. & Cozzoli, P. D. Colloidal heterostructured nanocrystals: Synthesis and growth mechanisms. Nano Today 5, 449–493 (2010).
Wang, C., Xu, C., Zeng, H. & Sun, S. Recent progress in syntheses and applications of dumbbell-like nanoparticles. Adv. Mater. 21, 3045–3052 (2009).
Reiss, P., Protière, M. & Li, L. Core/shell semiconductor nanocrystals. Small 5, 154–168 (2009).
Mazumder, V., Chi, M., More, K. L. & Sun, S. Synthesis and characterization of multimetallic Pd/Au and Pd/Au/FePt core/shell nanoparticles. Angew. Chem. Int. Ed. 49, 9368–9372 (2010).
Podsiadlo, P. et al. How “hollow” are hollow nanoparticles? J. Am. Chem. Soc. 135, 2435–2438 (2013).
Zhang, J., Tang, Y., Lee, K. & Ouyang, M. Nonepitaxial growth of hybrid core-shell nanostructures with large lattice mismatches. Science 327, 1634–1638 (2010).
Pellegrino, T. et al. Heterodimers based on CoPt3–Au nanocrystals with tunable domain size. J. Am. Chem. Soc. 128, 6690–6698 (2006).
Krylova, G. et al. Probing the surface of transition-metal nanocrystals by chemiluminesence. J. Am. Chem. Soc. 132, 9102–9110 (2010).
Krylova, G. et al. Study of nucleation and growth mechanism of the metallic nanodumbbells. J. Am. Chem. Soc. 134, 4384–4392 (2012).
Mokari, T., Rothenberg, E., Popov, I., Costi, R. & Banin, U. Selective growth of metal tips onto semiconductor quantum rods and tetrapods. Science 304, 1787–1790 (2004).
Mokari, T., Sztrum, C. G., Salant, A., Rabani, E. & Banin, U. Formation of asymmetric one-sided metal-tipped semiconductor nanocrystal dots and rods. Nature Mater. 4, 855–863 (2005).
Halpert, J. E., Porter, V. J., Zimmer, J. P. & Bawendi, M. G. Synthesis of CdSe/CdTe nanobarbells. J. Am. Chem. Soc. 128, 12590–12591 (2006).
Carbone, L. et al. Synthesis and micrometer-scale assembly of colloidal CdSe/CdS nanorods prepared by a seeded growth approach. Nano Lett. 7, 2942–2950 (2007).
Talapin, D. V. et al. Seeded growth of highly luminescent CdSe/CdS nanoheterostructures with rod and tetrapod morphologies. Nano Lett. 7, 2951–2959 (2007).
Fiore, A. et al. Tetrapod-shaped colloidal nanocrystals of II–VI semiconductors prepared by seeded growth. J. Am. Chem. Soc. 131, 2274–2282 (2009).
Pimpinelli, A. & Villain, J. Physics of Crystal Growth (Cambridge Univ. Press, 1999).
Wang, C. et al. Rational synthesis of heterostructured nanoparticles with morphology control. J. Am. Chem. Soc. 132, 6524–6529 (2010).
Habas, S. E., Lee, H., Radmilovic, V., Somorjai, G. A. & Yang, P. Shaping binary metal nanocrystals through epitaxial seeded growth. Nature Mater. 6, 692–697 (2007).
Smith, A. M., Mohs, A. M. & Nie, S. Tuning the optical and electronic properties of colloidal nanocrystals by lattice strain. Nature Nanotech. 4, 56–63 (2009).
Wanjala, B. N., Luo, J., Fang, B., Mott, D. & Zhong, C. J. Gold–platinum nanoparticles: Alloying and phase segregation. J. Mater. Chem. 21, 4012–4020 (2011).
Guinier, A. & Fournet, G. Small-Angle Scattering of X-rays (Wiley, 1955).
Roe, R-J. Method of X-ray and Neutron Scattering in Polymer Science (Oxford Univ. Press, 2000).
Winkler, P. M. et al. Heterogeneous nucleation experiments bridging the scale from molecular ion clusters to nanoparticles. Science 319, 1374–1377 (2008).
Fletcher, N. H. Size effect in heterogeneous nucleation. J. Chem. Phys. 29, 572–576 (1958).
Matthews, J. W. & Blakeslee, A. E. Defects in epitaxial multilayers. I. Misfit dislocations. J. Crystal Growth 27, 118–125 (1974).
Jesser, W. A. & Kuhlmann-Wilsdorf, D. On the theory of interfacial energy and elastic strain of epitaxial overgrowths in parallel alignment on single crystal substrates. Phys. Status Solidi 19, 95–105 (1967).
Wang, C., Wei, Y., Jiang, H. & Sun, S. Tug-of-war in nanoparticles: Competitive growth of Au on Au-Fe3O4 nanoparticles. Nano Lett. 9, 4544–4547 (2009).
Topor, L. & Kleppa, O. J. Thermochemistry of binary liquid gold alloys: The systems Au–Ni, Au–Co, Au–Fe, and Au–Mn. Metall. Trans. 15, 573–580 (1984).
Okamoto, H. & Massalski, T. B. The Au–Pt (gold–platinum) system. Bull. Alloys Phase Diagr. 6, 46–56 (1985).
Okamoto, H. & Massalski, T. B. The Au–Pd (gold–palladium) system. Bull. Alloys Phase Diagr. 6, 229–235 (1985).
Gallezot, P. & Richard, D. Selective hydrogenation of α,β-unsaturated aldehydes. Cat. Rev. 40, 81–126 (1998).
Vehkamäki, H. et al. Heterogeneous multicomponent nucleation theorems for the analysis of nanoclusters. J. Chem. Phys. 126, 174707 (2007).
Park, J., Joo, J., Kwon, S. G., Jang, Y. & Hyeon, T. Synthesis of monodisperse spherical nanocrystals. Angew. Chem. Int. Ed. 46, 4630–4660 (2007).
Shim, M., McDaniel, H. & Oh, N. Prospects for strained type-II nanorod heterostructures. J. Phys. Chem. Lett. 2, 2722–2727 (2011).
Buonsanti, R. et al. Architectural control of seeded-grown magnetic-semiconductor iron oxide–TiO2 nanorod heterostructures: The role of seeds in topology selection. J. Am. Chem. Soc. 132, 2437–2464 (2010).
Hull, D. & Bacon, D. J. Introduction to Dislocations 5th edn (Butterworth–Heinemann, 2011).
Kim, S. W. et al. Synthesis of monodisperse palladium nanoparticles. Nano Lett. 3, 1289–1291 (2003).
Kwon, S. G. et al. Capping ligands as selectivity switchers in hydrogenation reactions. Nano Lett. 12, 5382–5388 (2012).
Acknowledgements
Use of the Center for Nanoscale Materials and Advanced Photon Source was supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences, under Contract No. DE-AC02-06CH11357. MRCAT is funded by MRCAT host institutions. The authors thank C. Marshall for fruitful discussion and help in tests of catalytic activity. S.C. and T.S. would like to thank V. Zyryanov for help with experiments and C. Segre for beam-time allocation. P.J.P. and R.F.K. acknowledge support from the National Science Foundation (DMR-0959470) for the acquisition of the UIC JEOL JEMARM200CF. Support from the UIC Research Resources Center is also acknowledged. The work at GeoSoilEnviroCARS was supported by the National Science Foundation—Earth Sciences (EAR-0622171) and Department of Energy—Geosciences (DE-FG02-94ER14466).
Author information
Authors and Affiliations
Contributions
S.G.K. designed and implemented experiments, and analysed the data except for SAXS and EXAFS. G.K. carried out NP synthesis and sample preparations. P.J.P. and R.F.K. performed STEM and EDX measurements. S.C. and T.S. measured EXAFS and analysed the data. E.E.B. supervised catalytic reactions and analysis. Y.L. supervised TEM analysis. V.B.P. performed synchrotron X-ray diffraction measurements. B.L. supervised and performed SAXS/WAXS measurements and analysed SAXS data. E.V.S. supervised the project and wrote the manuscript with B.L. and S.G.K.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 4182 kb)
Rights and permissions
About this article
Cite this article
Kwon, S., Krylova, G., Phillips, P. et al. Heterogeneous nucleation and shape transformation of multicomponent metallic nanostructures. Nature Mater 14, 215–223 (2015). https://doi.org/10.1038/nmat4115
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4115
This article is cited by
-
High-entropy alloy nanopatterns by prescribed metallization of DNA origami templates
Nature Communications (2023)
-
Catalytic Potential and Utility of High Entropy Alloys
Transactions of the Indian National Academy of Engineering (2023)
-
High entropy materials based electrocatalysts for water splitting: Synthesis strategies, catalytic mechanisms, and prospects
Nano Research (2023)
-
Laser-generated high entropy metallic glass nanoparticles as bifunctional electrocatalysts
Nano Research (2022)
-
Low-temperature calcination of convenient micro-sized copper ink with surface activation and synchronous protection by in-situ chemisorbed cupric formate
Journal of Materials Science: Materials in Electronics (2022)