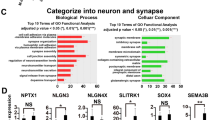
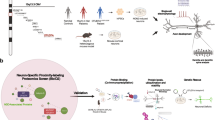
Abstract
Individuals with 22q11.2 microdeletions have cognitive deficits and a high risk of developing schizophrenia. Here we provide evidence that primary hippocampal neurons from a mouse model of 22q11.2 deletion (Df(16)A+/− mice) have decreased density of dendritic spines and glutamatergic synapses, as well as impaired dendritic growth. These deficits were prevented by introduction of the enzymatically active ZDHHC8 palmitoyltransferase encoded by a gene in the 22q11.2 locus, and they were also observed in primary cultures from Zdhhc8-deficient mice. Many of these deficits were also present in the hippocampi of adult Df(16)A+/− and Zdhhc8-deficient mice. Finally, we provide evidence that PSD95 is one of the substrates of ZDHHC8. Our analysis reveals that 22q11.2 microdeletion results in deficits in neuronal development and suggests that impaired neuronal protein palmitoylation contributes to many of these deficits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
McDonald-McGinn, D.M. et al. Phenotype of the 22q11.2 deletion in individuals identified through an affected relative: cast a wide FISHing net!. Genet. Med. 3, 23–29 (2001).
Woodin, M. et al. Neuropsychological profile of children and adolescents with the 22q11.2 microdeletion. Genet. Med. 3, 34–39 (2001).
Bearden, C.E. et al. The neurocognitive phenotype of the 22q11.2 deletion syndrome: selective deficit in visual-spatial memory. J. Clin. Exp. Neuropsychol. 23, 447–464 (2001).
Sobin, C. et al. Neuropsychological characteristics of children with the 22q11 Deletion Syndrome: a descriptive analysis. Child Neuropsychol. 11, 39–53 (2005).
Chow, E.W., Watson, M., Young, D.A. & Bassett, A.S. Neurocognitive profile in 22q11 deletion syndrome and schizophrenia. Schizophr. Res. 87, 270–278 (2006).
Pulver, A.E. et al. Psychotic illness in patients diagnosed with velo-cardio-facial syndrome and their relatives. J. Nerv. Ment. Dis. 182, 476–478 (1994).
Karayiorgou, M. et al. Schizophrenia susceptibility associated with interstitial deletions of chromosome 22q11. Proc. Natl. Acad. Sci. USA 92, 7612–7616 (1995).
Xu, B. et al. Strong association of de novo copy number mutations with sporadic schizophrenia. Nat. Genet. 40, 880–885 (2008).
Edelmann, L., Pandita, R.K. & Morrow, B.E. Low-copy repeats mediate the common 3-Mb deletion in patients with velo-cardio-facial syndrome. Am. J. Hum. Genet. 64, 1076–1086 (1999).
Gothelf, D. et al. COMT genotype predicts longitudinal cognitive decline and psychosis in 22q11.2 deletion syndrome. Nat. Neurosci. 8, 1500–1502 (2005).
Mukai, J. et al. Evidence that the gene encoding ZDHHC8 contributes to the risk of schizophrenia. Nat. Genet. 36, 725–731 (2004).
Paterlini, M. et al. Transcriptional and behavioral interaction between 22q11.2 orthologs modulates schizophrenia-related phenotypes in mice. Nat. Neurosci. 8, 1586–1594 (2005).
Raux, G. et al. Involvement of hyperprolinemia in cognitive and psychiatric features of the 22q11 deletion syndrome. Hum. Mol. Genet. 16, 83–91 (2007).
Fukata, M., Fukata, Y., Adesnik, H., Nicoll, R.A. & Bredt, D.S. Identification of PSD95 palmitoylating enzymes. Neuron 44, 987–996 (2004).
Linder, M.E. & Deschenes, R.J. Model organisms lead the way to protein palmitoyltransferases. J. Cell Sci. 117, 521–526 (2004).
Roth, A.F., Feng, Y., Chen, L. & Davis, N.G. The yeast DHHC cysteine-rich domain protein Akr1p is a palmitoyl transferase. J. Cell Biol. 159, 23–28 (2002).
Smotrys, J.E. & Linder, M.E. Palmitoylation of intracellular signaling proteins: regulation and function. Annu. Rev. Biochem. 73, 559–587 (2004).
Bijlmakers, M.J. & Marsh, M. The on-off story of protein palmitoylation. Trends Cell Biol. 13, 32–42 (2003).
el-Husseini, A.E. & Bredt, D.S. Protein palmitoylation: a regulator of neuronal development and function. Nat. Rev. Neurosci. 3, 791–802 (2002).
Chen, W.Y. et al. Case-control study and transmission disequilibrium test provide consistent evidence for association between schizophrenia and genetic variation in the 22q11 gene ZDHHC8. Hum. Mol. Genet. 13, 2991–2995 (2004).
Glaser, B. et al. No association between the putative functional ZDHHC8 single nucleotide polymorphism rs175174 and schizophrenia in large European samples. Biol. Psychiatry 58, 78–80 (2005).
Puech, A. et al. Comparative mapping of the human 22q11 chromosomal region and the orthologous region in mice reveals complex changes in gene organization. Proc. Natl. Acad. Sci. USA 94, 14608–14613 (1997).
Stark, K.L. et al. Altered brain microRNA biogenesis contributes to phenotypic deficits in a 22q11-deletion mouse model. Nat. Genet. 40, 751–760 (2008).
El-Husseini, A.E., Schnell, E., Chetkovich, D.M., Nicoll, R.A. & Bredt, D.S. PSD95 involvement in maturation of excitatory synapses. Science 290, 1364–1368 (2000).
Sala, C. et al. Regulation of dendritic spine morphology and synaptic function by Shank and Homer. Neuron 31, 115–130 (2001).
Charych, E.I. et al. Activity-independent regulation of dendrite patterning by postsynaptic density protein PSD95. J. Neurosci. 26, 10164–10176 (2006).
Chakravarthy, S. et al. Postsynaptic TrkB signaling has distinct roles in spine maintenance in adult visual cortex and hippocampus. Proc. Natl. Acad. Sci. USA 103, 1071–1076 (2006).
Bekkers, J.M. & Stevens, C.F. Excitatory and inhibitory autaptic currents in isolated hippocampal neurons maintained in cell culture. Proc. Natl. Acad. Sci. USA 88, 7834–7838 (1991).
Cho, K.O., Hunt, C.A. & Kennedy, M.B. The rat brain postsynaptic density fraction contains a homolog of the Drosophila discs-large tumor suppressor protein. Neuron 9, 929–942 (1992).
Kornau, H.C., Schenker, L.T., Kennedy, M.B. & Seeburg, P.H. Domain interaction between NMDA receptor subunits and the postsynaptic density protein PSD95. Science 269, 1737–1740 (1995).
Bellocchio, E.E., Reimer, R.J., Fremeau, R.T. Jr. & Edwards, R.H. Uptake of glutamate into synaptic vesicles by an inorganic phosphate transporter. Science 289, 957–960 (2000).
Ohno, Y., Kihara, A., Sano, T. & Igarashi, Y. Intracellular localization and tissue-specific distribution of human and yeast DHHC cysteine-rich domain-containing proteins. Biochim. Biophys. Acta 1761, 474–483 (2006).
Gauthier-Campbell, C., Bredt, D.S., Murphy, T.H. & El-Husseini Ael, D. Regulation of dendritic branching and filopodia formation in hippocampal neurons by specific acylated protein motifs. Mol. Biol. Cell 15, 2205–2217 (2004).
Feng, G. et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 41–51 (2000).
Tada, T. & Sheng, M. Molecular mechanisms of dendritic spine morphogenesis. Curr. Opin. Neurobiol. 16, 95–101 (2006).
Vessey, J.P. & Karra, D. More than just synaptic building blocks: scaffolding proteins of the post-synaptic density regulate dendritic patterning. J. Neurochem. 102, 324–332 (2007).
el-Husseini, A.E. et al. Synaptic strength regulated by palmitate cycling on PSD95. Cell 108, 849–863 (2002).
Topinka, J.R. & Bredt, D.S. N-terminal palmitoylation of PSD95 regulates association with cell membranes and interaction with K+ channel Kv1.4. Neuron 20, 125–134 (1998).
Ehrlich, I., Klein, M., Rumpel, S. & Malinow, R. PSD95 is required for activity-driven synapse stabilization. Proc. Natl. Acad. Sci. USA 104, 4176–4181 (2007).
Firestein, B.L., Craven, S.E. & Bredt, D.S. Postsynaptic targeting of MAGUKs mediated by distinct N-terminal domains. Neuroreport 11, 3479–3484 (2000).
Kanaani, J., Diacovo, M.J., El-Husseini Ael, D., Bredt, D.S. & Baekkeskov, S. Palmitoylation controls trafficking of GAD65 from Golgi membranes to axon-specific endosomes and a Rab5a-dependent pathway to presynaptic clusters. J. Cell Sci. 117, 2001–2013 (2004).
Kang, R. et al. Presynaptic trafficking of synaptotagmin I is regulated by protein palmitoylation. J. Biol. Chem. 279, 50524–50536 (2004).
Washbourne, P. et al. Cysteine residues of SNAP-25 are required for SNARE disassembly and exocytosis, but not for membrane targeting. Biochem. J. 357, 625–634 (2001).
Berthiaume, L. & Resh, M.D. Biochemical characterization of a palmitoyl acyltransferase activity that palmitoylates myristoylated proteins. J. Biol. Chem. 270, 22399–22405 (1995).
El-Husseini, A.E. et al. Dual palmitoylation of PSD95 mediates its vesiculotubular sorting, postsynaptic targeting, and ion channel clustering. J. Cell Biol. 148, 159–172 (2000).
Karayiorgou, M. & Gogos, J.A. The molecular genetics of the 22q11-associated schizophrenia. Brain Res. Mol. Brain Res. 132, 95–104 (2004).
Valdez-Taubas, J. & Pelham, H. Swf1-dependent palmitoylation of the SNARE Tlg1 prevents its ubiquitination and degradation. EMBO J. 24, 2524–2532 (2005).
Huang, K. et al. Huntingtin-interacting protein HIP14 is a palmitoyl transferase involved in palmitoylation and trafficking of multiple neuronal proteins. Neuron 44, 977–986 (2004).
Arguello, P.A. & Gogos, J.A. Modeling madness in mice: one piece at a time. Neuron 52, 179–196 (2006).
Gu, J.G. & MacDermott, A.B. Activation of ATP P2X receptors elicits glutamate release from sensory neuron synapses. Nature 389, 749–753 (1997).
Acknowledgements
We thank M. Sribour, A. Abrams-Downey, A. Garçia-Williams and D. Swor for technical support and assistance with the mouse colony; and G. Feng (Duke University) and P. Scheiffele (Columbia University) for constructs. This research was supported in part by the US National Institutes of Health (MH67068 to M.K. and J.A.G. and MH077235 to J.A.G.), a McKnight Brain Disorders Award, an EJLB Scholar award (to J.A.G.) and two NARSAD awards (to J.A.G. and J.M.).
Author information
Authors and Affiliations
Contributions
J.M., A.D., L.J.D. and L.C. conducted research; K.L.S. contributed reagents; J.M., A.D., L.J.D., L.C. and A.B.M. analyzed data; M.K. and J.A.G. supervised the project; and J.M., A.D., L.J.D., A.B.M., M.K. and J.A.G. wrote the paper.
Note: Supplementary information is available on the Nature Neuroscience website.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Note, Supplementary Data and Supplementary Methods (PDF 4837 kb)
Rights and permissions
About this article
Cite this article
Mukai, J., Dhilla, A., Drew, L. et al. Palmitoylation-dependent neurodevelopmental deficits in a mouse model of 22q11 microdeletion. Nat Neurosci 11, 1302–1310 (2008). https://doi.org/10.1038/nn.2204
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2204
This article is cited by
-
DiGeorge syndrome critical region gene 2 (DGCR2), a schizophrenia risk gene, regulates dendritic spine development through cell adhesion
Cell & Bioscience (2023)
-
The molecular pathology of schizophrenia: an overview of existing knowledge and new directions for future research
Molecular Psychiatry (2023)
-
The synaptic hypothesis of schizophrenia version III: a master mechanism
Molecular Psychiatry (2023)
-
Current advancements of modelling schizophrenia using patient-derived induced pluripotent stem cells
Acta Neuropathologica Communications (2022)
-
Roles of palmitoylation in structural long-term synaptic plasticity
Molecular Brain (2021)