Abstract
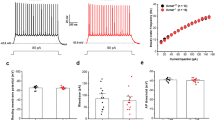
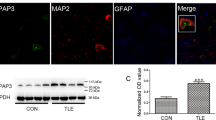
Dysfunction of fast-spiking, parvalbumin-positive (FS-PV) interneurons is implicated in the pathogenesis of epilepsy. ErbB4, a key Neuregulin 1 (NRG1) receptor, is mainly expressed in this type of interneurons, and recent studies suggest that parvalbumin interneurons are a major target of NRG1-ErbB4 signaling in adult brain. Thus, we hypothesized that downregulation of NRG1-ErbB4 signaling in FS-PV interneurons is involved in epilepsy. We found that NRG1, through its receptor ErbB4, increased the intrinsic excitability of FS-PV interneurons. This effect was mediated by increasing the near-threshold responsiveness and decreasing the voltage threshold for action potentials through Kv1.1, a voltage-gated potassium channel. Furthermore, mice with specific deletion of ErbB4 in parvalbumin interneurons were more susceptible to pentylenetetrazole- and pilocarpine-induced models of epilepsy. Exogenous NRG1 delayed the onset of seizures and decreased their incidence and stage. Moreover, expression of ErbB4, but not ErbB2, was downregulated in human epileptogenic tissue. Together, our findings suggest that NRG1–ErbB4 signaling contributes to human epilepsy through regulating the excitability of FS-PV interneurons. ErbB4 may be a new target for anticonvulsant drugs in epilepsy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Perucca, E., Alexandre, V. Jr. & Tomson, T. Old versus new antiepileptic drugs: the SANAD study. Lancet 370, 313; author reply 315–316 (2007).
Kawaguchi, Y. & Kubota, Y. GABAergic cell subtypes and their synaptic connections in rat frontal cortex. Cereb. Cortex 7, 476–486 (1997).
Lau, D. et al. Impaired fast-spiking, suppressed cortical inhibition, and increased susceptibility to seizures in mice lacking Kv3.2 K+ channel proteins. J. Neurosci. 20, 9071–9085 (2000).
Ogiwara, I. et al. Na(v)1.1 localizes to axons of parvalbumin-positive inhibitory interneurons: a circuit basis for epileptic seizures in mice carrying an Scn1a gene mutation. J. Neurosci. 27, 5903–5914 (2007).
Buonanno, A. The neuregulin signaling pathway and schizophrenia: from genes to synapses and neural circuits. Brain Res. Bull. 83, 122–131 (2010).
Mei, L. & Xiong, W.C. Neuregulin 1 in neural development, synaptic plasticity and schizophrenia. Nat. Rev. Neurosci. 9, 437–452 (2008).
Corfas, G., Roy, K. & Buxbaum, J.D. Neuregulin 1-erbB signaling and the molecular/cellular basis of schizophrenia. Nat. Neurosci. 7, 575–580 (2004).
Lai, C. & Lemke, G. An extended family of protein-tyrosine kinase genes differentially expressed in the vertebrate nervous system. Neuron 6, 691–704 (1991).
Fazzari, P. et al. Control of cortical GABA circuitry development by Nrg1 and ErbB4 signalling. Nature 464, 1376–1380 (2010).
Huang, Y.Z. et al. Regulation of neuregulin signaling by PSD-95 interacting with ErbB4 at CNS synapses. Neuron 26, 443–455 (2000).
Yau, H.J., Wang, H.F., Lai, C. & Liu, F.C. Neural development of the neuregulin receptor ErbB4 in the cerebral cortex and the hippocampus: preferential expression by interneurons tangentially migrating from the ganglionic eminences. Cereb. Cortex 13, 252–264 (2003).
Vullhorst, D. et al. Selective expression of ErbB4 in interneurons, but not pyramidal cells, of the rodent hippocampus. J. Neurosci. 29, 12255–12264 (2009).
Woo, R.S. et al. Neuregulin-1 enhances depolarization-induced GABA release. Neuron 54, 599–610 (2007).
Wen, L. et al. Neuregulin 1 regulates pyramidal neuron activity via ErbB4 in parvalbumin-positive interneurons. Proc. Natl. Acad. Sci. USA 107, 1211–1216 (2010).
Chen, Y.J. et al. ErbB4 in parvalbumin-positive interneurons is critical for neuregulin 1 regulation of long-term potentiation. Proc. Natl. Acad. Sci. USA 107, 21818–21823 (2010).
Kwon, O.B., Longart, M., Vullhorst, D., Hoffman, D.A. & Buonanno, A. Neuregulin-1 reverses long-term potentiation at CA1 hippocampal synapses. J. Neurosci. 25, 9378–9383 (2005).
Pitcher, G.M. et al. Schizophrenia susceptibility pathway neuregulin 1-ErbB4 suppresses Src upregulation of NMDA receptors. Nat. Med. 17, 470–478 (2011).
Goldberg, E.M. et al. K+ channels at the axon initial segment dampen near-threshold excitability of neocortical fast-spiking GABAergic interneurons. Neuron 58, 387–400 (2008).
Chattopadhyaya, B. et al. Experience and activity-dependent maturation of perisomatic GABAergic innervation in primary visual cortex during a postnatal critical period. J. Neurosci. 24, 9598–9611 (2004).
Di Cristo, G. et al. Subcellular domain-restricted GABAergic innervation in primary visual cortex in the absence of sensory and thalamic inputs. Nat. Neurosci. 7, 1184–1186 (2004).
Law, A.J., Shannon Weickert, C., Hyde, T.M., Kleinman, J.E. & Harrison, P.J. Neuregulin-1 (NRG-1) mRNA and protein in the adult human brain. Neuroscience 127, 125–136 (2004).
Ting, A.K. et al. Neuregulin 1 promotes excitatory synapse development and function in GABAergic interneurons. J. Neurosci. 31, 15–25 (2011).
Bjarnadottir, M. et al. Neuregulin1 (NRG1) signaling through Fyn modulates NMDA receptor phosphorylation: differential synaptic function in NRG1+/− knock-outs compared with wild-type mice. J. Neurosci. 27, 4519–4529 (2007).
Gu, Z., Jiang, Q., Fu, A.K., Ip, N.Y. & Yan, Z. Regulation of NMDA receptors by neuregulin signaling in prefrontal cortex. J. Neurosci. 25, 4974–4984 (2005).
Li, B., Woo, R.S., Mei, L. & Malinow, R. The neuregulin-1 receptor erbB4 controls glutamatergic synapse maturation and plasticity. Neuron 54, 583–597 (2007).
Fukazawa, R. et al. Neuregulin-1 protects ventricular myocytes from anthracycline-induced apoptosis via erbB4-dependent activation of PI3-kinase/Akt. J. Mol. Cell. Cardiol. 35, 1473–1479 (2003).
García-Rivello, H. et al. Dilated cardiomyopathy in Erb-b4-deficient ventricular muscle. Am. J. Physiol. Heart Circ. Physiol. 289, H1153–H1160 (2005).
Arber, S., Ladle, D.R., Lin, J.H., Frank, E. & Jessell, T.M. ETS gene Er81 controls the formation of functional connections between group Ia sensory afferents and motor neurons. Cell 101, 485–498 (2000).
Hippenmeyer, S. et al. A developmental switch in the response of DRG neurons to ETS transcription factor signaling. PLoS Biol. 3, e159 (2005).
Fisahn, A., Neddens, J., Yan, L. & Buonanno, A. Neuregulin-1 modulates hippocampal gamma oscillations: implications for schizophrenia. Cereb. Cortex 19, 612–618 (2009).
Neddens, J. & Buonanno, A. Selective populations of hippocampal interneurons express ErbB4 and their number and distribution is altered in ErbB4 knockout mice. Hippocampus 20, 724–744 (2010).
Coetzee, W.A. et al. Molecular diversity of K+ channels. Ann. NY Acad. Sci. 868, 233–285 (1999).
Guan, D. et al. Expression and biophysical properties of Kv1 channels in supragranular neocortical pyramidal neurones. J. Physiol. (Lond.) 571, 371–389 (2006).
Robertson, B., Owen, D., Stow, J., Butler, C. & Newland, C. Novel effects of dendrotoxin homologues on subtypes of mammalian Kv1 potassium channels expressed in Xenopus oocytes. FEBS Lett. 383, 26–30 (1996).
Ziyatdinova, S. et al. Spontaneous epileptiform discharges in a mouse model of Alzheimer′s disease are suppressed by antiepileptic drugs that block sodium channels. Epilepsy Res. 94, 75–85 (2011).
Dreyfus, J. A Remarkable Medicine Has Been Overlooked: Including an Autobiography and the Clinical Section of the Broad Range of Use of Phenytoin (Continuum International Publishing Group, 1998).
Perucca, E. Pharmacological and therapeutic properties of valproate: a summary after 35 years of clinical experience. CNS Drugs 16, 695–714 (2002).
Gibson, J.R., Beierlein, M. & Connors, B.W. Two networks of electrically coupled inhibitory neurons in neocortex. Nature 402, 75–79 (1999).
Wang, H., Kunkel, D.D., Schwartzkroin, P.A. & Tempel, B.L. Localization of Kv1.1 and Kv1.2, two K channel proteins, to synaptic terminals, somata, and dendrites in the mouse brain. J. Neurosci. 14, 4588–4599 (1994).
Tanouye, M.A., Ferrus, A. & Fujita, S.C. Abnormal action potentials associated with the Shaker complex locus of Drosophila. Proc. Natl. Acad. Sci. USA 78, 6548–6552 (1981).
Holmes, T.C., Fadool, D.A., Ren, R. & Levitan, I.B. Association of Src tyrosine kinase with a human potassium channel mediated by SH3 domain. Science 274, 2089–2091 (1996).
Cook, K.K. & Fadool, D.A. Two adaptor proteins differentially modulate the phosphorylation and biophysics of Kv1.3 ion channel by SRC kinase. J. Biol. Chem. 277, 13268–13280 (2002).
Tucker, K. & Fadool, D.A. Neurotrophin modulation of voltage-gated potassium channels in rat through TrkB receptors is time and sensory experience dependent. J. Physiol. (Lond.) 542, 413–429 (2002).
Cascella, N.G., Schretlen, D.J. & Sawa, A. Schizophrenia and epilepsy: is there a shared susceptibility? Neurosci. Res. 63, 227–235 (2009).
Zuberi, S.M. et al. A novel mutation in the human voltage-gated potassium channel gene (Kv1.1) associates with episodic ataxia type 1 and sometimes with partial epilepsy. Brain 122, 817–825 (1999).
Liguori, R., Avoni, P., Baruzzi, A., Di Stasi, V. & Montagna, P. Familial continuous motor unit activity and epilepsy. Muscle Nerve 24, 630–633 (2001).
Schulte, U. et al. The epilepsy-linked Lgi1 protein assembles into presynaptic Kv1 channels and inhibits inactivation by Kvbeta1. Neuron 49, 697–706 (2006).
Strauss, K.A. et al. Recessive symptomatic focal epilepsy and mutant contactin-associated protein-like 2. N. Engl. J. Med. 354, 1370–1377 (2006).
Smart, S.L. et al. Deletion of the K(V)1.1 potassium channel causes epilepsy in mice. Neuron 20, 809–819 (1998).
Ostrom, R.S. et al. Receptor number and caveolar co-localization determine receptor coupling efficiency to adenylyl cyclase. J. Biol. Chem. 276, 42063–42069 (2001).
Acknowledgements
We thank L. Mei (Georgia Health Sciences University) for advice. We are grateful to T.M. Gao (Southern Medical University), Z.J. Huang (Cold Spring Harbor), X.H. Zhang (Institute of Neuroscience, Chinese Academy of Sciences) and L. Bao (Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences) for providing reagents and/or mice. We express our gratitude to the subjects and their families for their participation. We also thank T.M. Gao and I.C. Bruce for critical reading of this manuscript, and Q.L. Miao for technical assistance. This work was supported by grants from the National Natural Science Foundation of China (30970916, 31070926 and 30725047), the Major Research Program from the state Ministry of Science and Technology of China (2010CB912004, 2010CB912002), the Zhejiang Provincial Natural Science Foundation of China (Z2090127), the Foundation for the Author of National Excellent Doctoral Dissertation of China (200937), the Science Foundation of Chinese Universities (JD09023), the Zhejiang Provincial Qianjiang Talent Plan (2010R10057), the Fundamental Research Funds for the Central Universities (2011XZZX002) and Zhejiang Province Key Technology Innovation Team (2010R50049).
Author information
Authors and Affiliations
Contributions
K.-X.L. conducted the electrophysiological studies, analyzed data and wrote the manuscript; Y.-M.L. conducted the western blot analyses and wrote the manuscript; Z.-H.X. performed the studies on the mouse model of epilepsy and analyzed data; J.Z. performed the immunostaining experiments; J.-M. Zhu and J.-M. Zhang performed surgery, provided the human tissues and analyzed data; S.-X.C. conducted part of the electrophysiological recording and gene identification; X.-J.C. purified the compounds; Z.C., S.D. and J.-H.L. contributed experimental suggestions; X.-M.L. supervised all phases of the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Table 1 (PDF 485 kb)
Rights and permissions
About this article
Cite this article
Li, KX., Lu, YM., Xu, ZH. et al. Neuregulin 1 regulates excitability of fast-spiking neurons through Kv1.1 and acts in epilepsy. Nat Neurosci 15, 267–273 (2012). https://doi.org/10.1038/nn.3006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3006
This article is cited by
-
Impairments of GABAergic transmission in hippocampus mediate increased susceptibility of epilepsy in the early stage of Alzheimer’s disease
Cell Communication and Signaling (2024)
-
Control of neuronal excitation–inhibition balance by BMP–SMAD1 signalling
Nature (2024)
-
Genetic labeling reveals spatial and cellular expression pattern of neuregulin 1 in mouse brain
Cell & Bioscience (2023)
-
The plasticitome of cortical interneurons
Nature Reviews Neuroscience (2023)
-
Dysfunction of NRG1/ErbB4 Signaling in the Hippocampus Might Mediate Long-term Memory Decline After Systemic Inflammation
Molecular Neurobiology (2023)