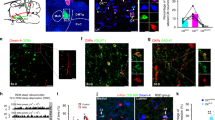
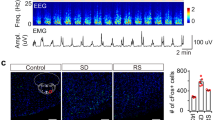
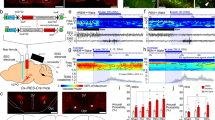
Abstract
Rapid-eye movement (REM) sleep correlates with neuronal activity in the brainstem, basal forebrain and lateral hypothalamus. Lateral hypothalamus melanin-concentrating hormone (MCH)-expressing neurons are active during sleep, but their effects on REM sleep remain unclear. Using optogenetic tools in newly generated Tg(Pmch-cre) mice, we found that acute activation of MCH neurons (ChETA, SSFO) at the onset of REM sleep extended the duration of REM, but not non-REM, sleep episodes. In contrast, their acute silencing (eNpHR3.0, archaerhodopsin) reduced the frequency and amplitude of hippocampal theta rhythm without affecting REM sleep duration. In vitro activation of MCH neuron terminals induced GABAA-mediated inhibitory postsynaptic currents in wake-promoting histaminergic neurons of the tuberomammillary nucleus (TMN), and in vivo activation of MCH neuron terminals in TMN or medial septum also prolonged REM sleep episodes. Collectively, these results suggest that activation of MCH neurons maintains REM sleep, possibly through inhibition of arousal circuits in the mammalian brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jouvet, M. Research on the neural structures and responsible mechanisms in different phases of physiological sleep. Arch. Ital. Biol. 100, 125–206 (1962).
Saper, C.B., Fuller, P.M., Pedersen, N.P., Lu, J. & Scammell, T.E. Sleep state switching. Neuron 68, 1023–1042 (2010).
McGinty, D.J. & Harper, R.M. Dorsal raphe neurons: depression of firing during sleep in cats. Brain Res. 101, 569–575 (1976).
Aston-Jones, G. & Bloom, F.E. Activity of norepinephrine-containing locus coeruleus neurons in behaving rats anticipates fluctuations in the sleep-waking cycle. J. Neurosci. 1, 876–886 (1981).
McCarley, R.W. & Hobson, J.A. Neuronal excitability modulation over the sleep cycle: a structural and mathematical model. Science 189, 58–60 (1975).
Hobson, J.A., McCarley, R.W. & Wyzinski, P.W. Sleep cycle oscillation: reciprocal discharge by two brainstem neuronal groups. Science 189, 55–58 (1975).
Pace-Schott, E.F. & Hobson, J.A. The neurobiology of sleep: genetics, cellular physiology and subcortical networks. Nat. Rev. Neurosci. 3, 591–605 (2002).
Sapin, E. et al. Localization of the brainstem GABAergic neurons controlling paradoxical (REM) sleep. PLoS ONE 4, e4272 (2009).
Boissard, R., Fort, P., Gervasoni, D., Barbagli, B. & Luppi, P.-H. Localization of the GABAergic and non-GABAergic neurons projecting to the sublaterodorsal nucleus and potentially gating paradoxical sleep onset. Eur. J. Neurosci. 18, 1627–1639 (2003).
Verret, L., Fort, P., Gervasoni, D., Leger, L. & Luppi, P.-H. Localization of the neurons active during paradoxical (REM) sleep and projecting to the locus coeruleus noradrenergic neurons in the rat. J. Comp. Neurol. 495, 573–586 (2006).
Lu, J., Sherman, D., Devor, M. & Saper, C.B. A putative flip-flop switch for control of REM sleep. Nature 441, 589–594 (2006).
Luppi, P.-H., Clément, O. & Fort, P. Paradoxical (REM) sleep genesis by the brainstem is under hypothalamic control. Curr. Opin. Neurobiol. doi:10.1016/j.conb.2013.02.006 (2013).10.1016/j.conb.2013.02.006
Lu, J. et al. Selective activation of the extended ventrolateral preoptic nucleus during rapid eye movement sleep. J. Neurosci. 22, 4568–4576 (2002).
Clément, O. et al. The lateral hypothalamic area controls paradoxical (REM) sleep by means of descending projections to brainstem GABAergic neurons. J. Neurosci. 32, 16763–16774 (2012).
Verret, L. et al. A role of melanin-concentrating hormone producing neurons in the central regulation of paradoxical sleep. BMC Neurosci. 4, 19 (2003).
Sapin, E. et al. A very large number of GABAergic neurons are activated in the tuberal hypothalamus during paradoxical (REM) sleep hypersomnia. PLoS ONE 5, e11766 (2010).
Monti, J.M., Torterolo, P. & Lagos, P. Melanin-concentrating hormone control of sleep-wake behavior. Sleep Med. Rev. 17, 293–298 (2013).
Konadhode, R.R. et al. Optogenetic stimulation of MCH neurons increases sleep. J. Neurosci. 33, 10257–10263 (2013).
Modirrousta, M., Mainville, L. & Jones, B.E. Orexin and MCH neurons express c-Fos differently after sleep deprivation vs. recovery and bear different adrenergic receptors. Eur. J. Neurosci. 21, 2807–2816 (2005).
Hanriot, L. et al. Characterization of the melanin-concentrating hormone neurons activated during paradoxical sleep hypersomnia in rats. J. Comp. Neurol. 505, 147–157 (2007).
Hassani, O.K., Lee, M.G. & Jones, B.E. Melanin-concentrating hormone neurons discharge in a reciprocal manner to orexin neurons across the sleep-wake cycle. Proc. Natl. Acad. Sci. USA 106, 2418–2422 (2009).
Willie, J.T., Sinton, C.M., Maratos-Flier, E. & Yanagisawa, M. Abnormal response of melanin-concentrating hormone deficient mice to fasting: hyperactivity and rapid eye movement sleep suppression. Neuroscience 156, 819–829 (2008).
Adamantidis, A. et al. Sleep architecture of the melanin-concentrating hormone receptor 1-knockout mice. Eur. J. Neurosci. 27, 1793–1800 (2008).
Tsai, H.-C. et al. Phasic firing in dopaminergic neurons is sufficient for behavioral conditioning. Science 324, 1080–1084 (2009).
Atasoy, D., Aponte, Y., Su, H.H. & Sternson, S.M.A. FLEX switch targets channelrhodopsin-2 to multiple cell types for imaging and long-range circuit mapping. J. Neurosci. 28, 7025–7030 (2008).
Gunaydin, L.A. et al. Ultrafast optogenetic control. Nat. Neurosci. 13, 387–392 (2010).
Bittencourt, J.C. et al. The melanin-concentrating hormone system of the rat brain: an immuno- and hybridization histochemical characterization. J. Comp. Neurol. 319, 218–245 (1992).
van den Pol, A.N., Acuna-Goycolea, C., Clark, K.R. & Ghosh, P.K. Physiological properties of hypothalamic MCH neurons identified with selective expression of reporter gene after recombinant virus infection. Neuron 42, 635–652 (2004).
Burdakov, D., Gerasimenko, O. & Verkhratsky, A. Physiological changes in glucose differentially modulate the excitability of hypothalamic melanin-concentrating hormone and orexin neurons in situ. J. Neurosci. 25, 2429–2433 (2005).
Berndt, A., Yizhar, O., Gunaydin, L.A., Hegemann, P. & Deisseroth, K. Bi-stable neural state switches. Nat. Neurosci. 12, 229–234 (2009).
Zhang, F. et al. Multimodal fast optical interrogation of neural circuitry. Nature 446, 633–639 (2007).
Gradinaru, V. et al. Molecular and cellular approaches for diversifying and extending optogenetics. Cell 141, 154–165 (2010).
Han, X. & Boyden, E.S. Multiple-color optical activation, silencing and desynchronization of neural activity, with single-spike temporal resolution. PLoS ONE 2, e299 (2007).
Chow, B.Y. et al. High-performance genetically targetable optical neural silencing by light-driven proton pumps. Nature 463, 98–102 (2010).
Elias, C.F. et al. Characterization of CART neurons in the rat and human hypothalamus. J. Comp. Neurol. 432, 1–19 (2001).
Del Cid-Pellitero, E. & Jones, B.E. Immunohistochemical evidence for synaptic release of GABA from melanin-concentrating hormone containing varicosities in the locus coeruleus. Neuroscience 223, 269–276 (2012).
Fort, P. et al. The satiety molecule nesfatin-1 is co-expressed with melanin concentrating hormone in tuberal hypothalamic neurons of the rat. Neuroscience 155, 174–181 (2008).
Tan, C.P. et al. Melanin-concentrating hormone receptor subtypes 1 and 2: species-specific gene expression. Genomics 79, 785–792 (2002).
Takahashi, K., Lin, J.-S. & Sakai, K. Neuronal activity of histaminergic tuberomammillary neurons during wake-sleep states in the mouse. J. Neurosci. 26, 10292–10298 (2006).
Schöne, C. et al. Optogenetic probing of fast glutamatergic transmission from hypocretin/orexin to histamine neurons in situ. J. Neurosci. 32, 12437–12443 (2012).
van den Pol, A.N. Neuropeptide transmission in brain circuits. Neuron 76, 98–115 (2012).
Lagos, P., Torterolo, P., Jantos, H. & Monti, J.M. Immunoneutralization of melanin-concentrating hormone (MCH) in the dorsal raphe nucleus: effects on sleep and wakefulness. Brain Res. 1369, 112–118 (2011).
Saito, Y., Cheng, M., Leslie, F.M. & Civelli, O. Expression of the melanin-concentrating hormone (MCH) receptor mRNA in the rat brain. J. Comp. Neurol. 435, 26–40 (2001).
Chee, M.J.S., Pissios, P. & Maratos-Flier, E. Neurochemical characterization of neurons expressing melanin-concentrating hormone receptor 1 in the mouse hypothalamus. J. Comp. Neurol. 521, 2208–2234 (2013).
Lee, M.G., Hassani, O.K. & Jones, B.E. Discharge of identified orexin/hypocretin neurons across the sleep-waking cycle. J. Neurosci. 25, 6716–6720 (2005).
Mileykovskiy, B.Y., Kiyashchenko, L.I. & Siegel, J.M. Behavioral correlates of activity in identified hypocretin/orexin neurons. Neuron 46, 787–798 (2005).
Lu, Z.-H. et al. Melanin concentrating hormone induces hippocampal acetylcholine release via the medial septum in rats. Peptides 44, 32–39 (2013).
Wu, M., Dumalska, I., Morozova, E., van den Pol, A. & Alreja, M. Melanin-concentrating hormone directly inhibits GnRH neurons and blocks kisspeptin activation, linking energy balance to reproduction. Proc. Natl. Acad. Sci. USA 106, 17217–17222 (2009).
Adamantidis, A. et al. Disrupting the melanin-concentrating hormone receptor 1 in mice leads to cognitive deficits and alterations of NMDA receptor function. Eur. J. Neurosci. 21, 2837–2844 (2005).
Pachoud, B. et al. Major impairments of glutamatergic transmission and long-term synaptic plasticity in the hippocampus of mice lacking the melanin-concentrating hormone receptor-1. J. Neurophysiol. 104, 1417–1425 (2010).
Warming, S., Costantino, N., Court, D.L., Jenkins, N.A. & Copeland, N.G. Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res. 33, e36 (2005).
Grimm, D. et al. In vitro and in vivo gene therapy vector evolution via multispecies interbreeding and retargeting of adeno-associated viruses. J. Virol. 82, 5887–5911 (2008).
Adamantidis, A.R., Zhang, F., Aravanis, A.M., Deisseroth, K. & de Lecea, L. Neural substrates of awakening probed with optogenetic control of hypocretin neurons. Nature 450, 420–424 (2007).
Franken, P., Chollet, D. & Tafti, M. The homeostatic regulation of sleep need is under genetic control. J. Neurosci. 21, 2610–2621 (2001).
Bokil, H., Andrews, P., Kulkarni, J.E., Mehta, S. & Mitra, P.P. Chronux: a platform for analyzing neural signals. J. Neurosci. Methods 192, 146–151 (2010).
Schöne, C. et al. Optogenetic probing of fast glutamatergic transmission from hypocretin/orexin to histamine neurons in situ. J. Neurosci. 32, 12437–12443 (2012).
Acknowledgements
We thank B. Jones, M.E. Carter and L. de Lecea for helpful comments on a previous version of the manuscript. We thank the members of the Adamantidis laboratory for their technical help and comments. Optogenetic plasmids were kindly provided by K. Deisseroth (Stanford University). S.D.G. was supported by the Fonds de la Recherche du Québec - Santé. D.B. and A.R.A. were supported by the Human Frontier Science Program. A.R.A. was supported by the Douglas Foundation, McGill University, Canadian Fund for Innovation, Canadian Research Chair (Tier 2), Canadian Institute for Health Research and the Natural Science and Engineering Council of Canada.
Author information
Authors and Affiliations
Contributions
All of the authors designed the experiments. S.J., S.D.G., C.G.H., M.E. and S.J.R. collected data and performed analysis. M.E and J.F. generated the transgenic mouse model. All of the authors discussed the results and S.J., S.D.G., D.B. and A.R.A. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Photomicrographs of coronal brain sections from a Tg(Pmch-Cre) mouse injected with AAVdj-ChETA-EYFP in the LH-ZI area.
Pictures show dense EYFP-expressing MCH neuron projection terminating within sleep-related structures including the medial septum (a), neocortex (b), raphe nucleus (c), and the locus coeruleus (d). Abbreviations used: LC: Locus coeruleus. Scale bar: 200 μm.
Supplementary Figure 2 MCH neurons transfected with AAVdj-ChETA-EYFP increase their firing rate in response to blue light pulses.
a, Schematic of the optrode recording system. Note that the optic fiber is placed in the glass pipette. b, In vivo extracellular recording of ChETA-expressing MCH neuron in the LH showing tonic firing evoked by 1 Hz light pulse trains. Note the spontaneous firing rate of MCH neuron in the absence of light. c, Extracellular waveforms recorded from putative MCH neurons before (black), during (green) and after (red) optical stimulation of LH area. Note the strong similarity between spontaneous and light-evoked waveforms. d, Quantification of the discharge of MCH neurons before, during and after the light stimulation. MCH neurons firing was significantly increased by the 1Hz light pulse trains (n = 4 cells, 4 animals, 12 optical stimulations). Number of spike per second are represented as mean ± SEM. ***: p < 0.001 compared to the firing evoked by light pulse trains, one-way ANOVA within subject design, followed by Tukey post-hoc test.
Supplementary Figure 3 Spontaneous sleep-wake cycle of Tg(Pmch-cre) and Tg(Pmch-cre); Mchr1–/–.
a. Spontaneous duration of wake, NREM and REM sleep (expressed as a percentage of time) of Tg(Pmch-Cre) transduced with ChETA-EYFP (blue, n = 8), SSFO-EYFP (orange, n = 4), NpHR3.0-EYFP (red, n = 6) virus, and their EYFP controls (black, n = 8) during the light and dark phases, and 24h of the light/dark cycle. No significant differences were found between Tg(Pmch-Cre) animals expressing the opsin and their controls demonstrating that expression of the opsin in MCH neurons does not alter the spontaneous sleep-wake cycle of the mice b. Spontaneous duration of wake, NREM and REM sleep (expressed as a percentage of time) of Tg(Pmch-Cre) animals (n = 8) compared to [Tg(Pmch-Cre) X MCHR-1-/- ] animals (n = 8) showing a hyperactive phenotype during the dark phase for the double transgenic animals associated with a significant decrease of both NREM sleep and REM sleep. No significant differences during the light-dark phases and the 24h were found. Mean durations are represented as mean ± SEM. *: p < 0.05, ***: p < 0.001 one-way ANOVA between subject design for viral transduction, followed by Tukey post-hoc test or unpaired two-tailed t test.
Supplementary Figure 4 Sleep-specific stimulation during NREM and REM sleep.
a, timeline of NREM and REM sleep-specific stimulation experiments (right). B, Representative EEG/EMG recordings showing optical stimulation of ChETA (lower trace) and EYFP (control, upper trace) animals time-locked to NREM sleep episode. Note the transition from NREM to REM sleep occurring in ChETA animals after blue light stimulation. Horizontal blue bar (dashed) represents optical stimulations. C, Representative EEG/EMG recordings showing optical stimulation of ChETA (lower trace) and EYFP (control, upper trace) animals time-locked to REM sleep episode. Note that optical stimulation of EYFP animal starts at the onset of REM sleep (i.e., after the transition from NREM to REM sleep is complete). Wakefulness signals the termination of REM sleep. Horizontal blue bar (dashed) represents optical stimulations.
Supplementary Figure 5 Activation of SSFO in MCH neurons leads to increased excitability in response to a replayed current trace.
a, To assess whether SSFO activation resulted in increased excitability, a series of EPSCs and IPSCs in an identified MCH neuron were recorded. The current trace (black) was then replayed to MCH cells transfected with SSFO before (red) and during (blue) activation with a brief pulse of blue light (50 ms pulse width). b, Spike waveforms from MCH neurons before (red) and during (blue) SSFO activation are similar. c, Average data show that activation of SSFO resulted in a 4.5-fold increase in the number of spikes elicited during replay compared to baseline (control : 2.5±1.3 spikes, SSFO: 11.3±0.9 spikes, p = 0.0008, n = 4, at least 3x30 s sweep per cell). *** p<0.001, paired two-tailed t-test.
Supplementary Figure 6 MCH neurons silencing decreases the stability of theta oscillations during REM sleep
a, ArchT-expressing MCH neurons show a persistent outward current (bottom) and concomitant hyperpolarization (top) upon 30 s constant yellow illumination in voltage and current clamp, respectively. Note that cells were depolarized to above-threshold potentials using constant steady-state current injection to elicit spiking. b, Quantification of membrane hyperpolarization (Mean : 34.0 ±8.1 mV) and outward currents (166.2±83.4 pA) of ArchT-expressing MCH neurons upon optical silencing (n=5 cells in 4 slices across 2 animals).
Supplementary Figure 7 Recorded neurons (represented by biocytin labeling in green) colocalized with histamine decarboxylase–positive cells (in red).
Scale bar: 50 μm.
Supplementary Figure 8 Optical fiber positions across experimental conditions over the LH, the TMN, the MS and the DR for Tg(Pmch-cre) and Tg(Pmch-cre); Mchr1–/– mice.
Drawings were generated according to the mouse brain atlas{Paxinos:2004ts}. Scale bar: 500 μm. Abbreviations used: ZI: Zona Incerta, LH: lateral hypothalamus, 3V: third ventricle, Arc: Arcuate hypothalamic nucleus, f: fornix, ns: nigrostriatal tract, LV: lateral ventricle, MS: medial septal nucleus, aca: anterior commisure, Aq: Aqueduct, DR: doral raphe nucleus, MnR: Median raphe nucleus, VTM: ventral tuberomammillary nucleus.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 15080 kb)
Rights and permissions
About this article
Cite this article
Jego, S., Glasgow, S., Herrera, C. et al. Optogenetic identification of a rapid eye movement sleep modulatory circuit in the hypothalamus. Nat Neurosci 16, 1637–1643 (2013). https://doi.org/10.1038/nn.3522
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3522
This article is cited by
-
Probing pathways by which rhynchophylline modifies sleep using spatial transcriptomics
Biology Direct (2023)
-
Prefrontal cortical regulation of REM sleep
Nature Neuroscience (2023)
-
Sleep-mediated regulation of reward circuits: implications in substance use disorders
Neuropsychopharmacology (2023)
-
Neuro-orchestration of sleep and wakefulness
Nature Neuroscience (2023)
-
Neural Control of REM Sleep and Motor Atonia: Current Perspectives
Current Neurology and Neuroscience Reports (2023)