Abstract
Postnatal and adult subventricular zone (SVZ) neurogenesis is believed to be primarily controlled by neural stem cell (NSC)-intrinsic mechanisms, interacting with extracellular and niche-driven cues. Although behavioral experiments and disease states have suggested possibilities for higher level inputs, it is unknown whether neural activity patterns from discrete circuits can directly regulate SVZ neurogenesis. We identified a previously unknown population of choline acetyltransferase (ChAT)+ neurons residing in the rodent SVZ neurogenic niche. These neurons showed morphological and functional differences from neighboring striatal counterparts and released acetylcholine locally in an activity-dependent fashion. Optogenetic inhibition and stimulation of subependymal ChAT+ neurons in vivo indicated that they were necessary and sufficient to control neurogenic proliferation. Furthermore, whole-cell recordings and biochemical experiments revealed direct SVZ NSC responses to local acetylcholine release, synergizing with fibroblast growth factor receptor activation to increase neuroblast production. These results reveal an unknown gateway connecting SVZ neurogenesis to neuronal activity-dependent control and suggest possibilities for modulating neuroregenerative capacities in health and disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
10 June 2014
In the version of this article initially published online, the plus signs denoting the mean values in Figure 7e were displaced upward by ~0.1 units. The error has been corrected for the print, PDF and HTML versions of this article.
References
Kelsch, W., Sim, S. & Lois, C. Watching synaptogenesis in the adult brain. Annu. Rev. Neurosci. 33, 131–149 (2010).
Ihrie, R.A. & Alvarez-Buylla, A. Lake-front property: a unique germinal niche by the lateral ventricles of the adult brain. Neuron 70, 674–686 (2011).
Lazarini, F. & Lledo, P.M. Is adult neurogenesis essential for olfaction? Trends Neurosci. 34, 20–30 (2011).
Aboody, K., Capela, A., Niazi, N., Stern, J.H. & Temple, S. Translating stem cell studies to the clinic for CNS repair: current state of the art and the need for a Rosetta stone. Neuron 70, 597–613 (2011).
Robel, S., Berninger, B. & Gotz, M. The stem cell potential of glia: lessons from reactive gliosis. Nat. Rev. Neurosci. 12, 88–104 (2011).
Benner, E.J. et al. Protective astrogenesis from the SVZ niche after injury is controlled by Notch modulator Thbs4. Nature 497, 369–373 (2013).
Kriegstein, A. & Alvarez-Buylla, A. The glial nature of embryonic and adult neural stem cells. Annu. Rev. Neurosci. 32, 149–184 (2009).
Suh, H., Deng, W. & Gage, F.H. Signaling in adult neurogenesis. Annu. Rev. Cell Dev. Biol. 25, 253–275 (2009).
Shen, Q. et al. Endothelial cells stimulate self-renewal and expand neurogenesis of neural stem cells. Science 304, 1338–1340 (2004).
Tavazoie, M. et al. A specialized vascular niche for adult neural stem cells. Cell Stem Cell 3, 279–288 (2008).
Kuo, C.T. et al. Postnatal deletion of Numb/Numblike reveals repair and remodeling capacity in the subventricular neurogenic niche. Cell 127, 1253–1264 (2006).
Paez-Gonzalez, P. et al. Ank3-dependent SVZ niche assembly is required for the continued production of new neurons. Neuron 71, 61–75 (2011).
Miller, F.D. & Gauthier-Fisher, A. Home at last: neural stem cell niches defined. Cell Stem Cell 4, 507–510 (2009).
Bovetti, S., Gribaudo, S., Puche, A.C., De Marchis, S. & Fasolo, A. From progenitors to integrated neurons: role of neurotransmitters in adult olfactory neurogenesis. J. Chem. Neuroanat. 42, 304–316 (2011).
Young, S.Z., Taylor, M.M. & Bordey, A. Neurotransmitters couple brain activity to subventricular zone neurogenesis. Eur. J. Neurosci. 33, 1123–1132 (2011).
Brazel, C.Y., Nunez, J.L., Yang, Z. & Levison, S.W. Glutamate enhances survival and proliferation of neural progenitors derived from the subventricular zone. Neuroscience 131, 55–65 (2005).
Platel, J.C. et al. NMDA receptors activated by subventricular zone astrocytic glutamate are critical for neuroblast survival prior to entering a synaptic network. Neuron 65, 859–872 (2010).
Lin, C.W. et al. Genetically increased cell-intrinsic excitability enhances neuronal integration into adult brain circuits. Neuron 65, 32–39 (2010).
Liu, X., Wang, Q., Haydar, T.F. & Bordey, A. Nonsynaptic GABA signaling in postnatal subventricular zone controls proliferation of GFAP-expressing progenitors. Nat. Neurosci. 8, 1179–1187 (2005).
Alfonso, J., Le Magueresse, C., Zuccotti, A., Khodosevich, K. & Monyer, H. Diazepam binding inhibitor promotes progenitor proliferation in the postnatal SVZ by reducing GABA signaling. Cell Stem Cell 10, 76–87 (2012).
Young, S.Z., Platel, J.C., Nielsen, J.V., Jensen, N.A. & Bordey, A. GABA(A) increases calcium in subventricular zone astrocyte-like cells through L- and T-type voltage-gated calcium channels. Front. Cell. Neurosci. 4, 8 (2010).
Van Kampen, J.M., Hagg, T. & Robertson, H.A. Induction of neurogenesis in the adult rat subventricular zone and neostriatum following dopamine D3 receptor stimulation. Eur. J. Neurosci. 19, 2377–2387 (2004).
O'Keeffe, G.C. et al. Dopamine-induced proliferation of adult neural precursor cells in the mammalian subventricular zone is mediated through EGF. Proc. Natl. Acad. Sci. USA 106, 8754–8759 (2009).
Banasr, M., Hery, M., Printemps, R. & Daszuta, A. Serotonin-induced increases in adult cell proliferation and neurogenesis are mediated through different and common 5-HT receptor subtypes in the dentate gyrus and the subventricular zone. Neuropsychopharmacology 29, 450–460 (2004).
Tong, C.K. et al. Axonal control of the adult neural stem cell niche. Cell Stem Cell 14, 500–511 (2014).
Cooper-Kuhn, C.M., Winkler, J. & Kuhn, H.G. Decreased neurogenesis after cholinergic forebrain lesion in the adult rat. J. Neurosci. Res. 77, 155–165 (2004).
Mudò, G., Belluardo, N., Mauro, A. & Fuxe, K. Acute intermittent nicotine treatment induces fibroblast growth factor-2 in the subventricular zone of the adult rat brain and enhances neuronal precursor cell proliferation. Neuroscience 145, 470–483 (2007).
Misgeld, T. et al. Roles of neurotransmitter in synapse formation: development of neuromuscular junctions lacking choline acetyltransferase. Neuron 36, 635–648 (2002).
de Castro, B.M. et al. The vesicular acetylcholine transporter is required for neuromuscular development and function. Mol. Cell. Biol. 29, 5238–5250 (2009).
Zhou, D. et al. AnkyrinG is required for clustering of voltage-gated Na channels at axon initial segments and for normal action potential firing. J. Cell Biol. 143, 1295–1304 (1998).
Kordeli, E., Lambert, S. & Bennett, V. AnkyrinG. A new ankyrin gene with neural-specific isoforms localized at the axonal initial segment and node of Ranvier. J. Biol. Chem. 270, 2352–2359 (1995).
Patel, J.C., Rossignol, E., Rice, M.E. & Machold, R.P. Opposing regulation of dopaminergic activity and exploratory motor behavior by forebrain and brainstem cholinergic circuits. Nat. Commun. 3, 1172 (2012).
Guzman, M.S. et al. Elimination of the vesicular acetylcholine transporter in the striatum reveals regulation of behaviour by cholinergic-glutamatergic co-transmission. PLoS Biol. 9, e1001194 (2011).
Bennett, B.D. & Wilson, C.J. Spontaneous activity of neostriatal cholinergic interneurons in vitro. J. Neurosci. 19, 5586–5596 (1999).
Ding, J.B., Guzman, J.N., Peterson, J.D., Goldberg, J.A. & Surmeier, D.J. Thalamic gating of corticostriatal signaling by cholinergic interneurons. Neuron 67, 294–307 (2010).
Nguyen, Q.T. et al. An in vivo biosensor for neurotransmitter release and in situ receptor activity. Nat. Neurosci. 13, 127–132 (2010).
Doetsch, F., Garcia-Verdugo, J.M. & Alvarez-Buylla, A. Cellular composition and three-dimensional organization of the subventricular germinal zone in the adult mammalian brain. J. Neurosci. 17, 5046–5061 (1997).
Bertran-Gonzalez, J., Chieng, B.C., Laurent, V., Valjent, E. & Balleine, B.W. Striatal cholinergic interneurons display activity-related phosphorylation of ribosomal protein S6. PLoS ONE 7, e53195 (2012).
Knight, Z.A. et al. Molecular profiling of activated neurons by phosphorylated ribosome capture. Cell 151, 1126–1137 (2012).
Nagode, D.A., Tang, A.H., Karson, M.A., Klugmann, M. & Alger, B.E. Optogenetic release of ACh induces rhythmic bursts of perisomatic IPSCs in hippocampus. PLoS ONE 6, e27691 (2011).
Ma, W. et al. Acetylcholine stimulates cortical precursor cell proliferation in vitro via muscarinic receptor activation and MAP kinase phosphorylation. Eur. J. Neurosci. 12, 1227–1240 (2000).
Campbell, J.S., Wenderoth, M.P., Hauschka, S.D. & Krebs, E.G. Differential activation of mitogen-activated protein kinase in response to basic fibroblast growth factor in skeletal muscle cells. Proc. Natl. Acad. Sci. USA 92, 870–874 (1995).
Shingo, T. et al. Pregnancy-stimulated neurogenesis in the adult female forebrain mediated by prolactin. Science 299, 117–120 (2003).
Wang, W. et al. Extracellular signal-regulated kinase 5 (ERK5) mediates prolactin-stimulated adult neurogenesis in the subventricular zone and olfactory bulb. J. Biol. Chem. 288, 2623–2631 (2013).
Mak, G.K. et al. Male pheromone-stimulated neurogenesis in the adult female brain: possible role in mating behavior. Nat. Neurosci. 10, 1003–1011 (2007).
Mak, G.K. & Weiss, S. Paternal recognition of adult offspring mediated by newly generated CNS neurons. Nat. Neurosci. 13, 753–758 (2010).
Markwardt, S.J., Dieni, C.V., Wadiche, J.I. & Overstreet-Wadiche, L. Ivy/neurogliaform interneurons coordinate activity in the neurogenic niche. Nat. Neurosci. 14, 1407–1409 (2011).
Song, J. et al. Neuronal circuitry mechanism regulating adult quiescent neural stem-cell fate decision. Nature 489, 150–154 (2012).
Song, J. et al. Parvalbumin interneurons mediate neuronal circuitry-neurogenesis coupling in the adult hippocampus. Nat. Neurosci. 16, 1728–1730 (2013).
Young, S.Z. et al. NKCC1 knockdown decreases neuron production through GABAA-regulated neural progenitor proliferation and delays dendrite development. J. Neurosci. 32, 13630–13638 (2012).
Gong, S. et al. Targeting Cre recombinase to specific neuron populations with bacterial artificial chromosome constructs. J. Neurosci. 27, 9817–9823 (2007).
Gong, S. et al. A gene expression atlas of the central nervous system based on bacterial artificial chromosomes. Nature 425, 917–925 (2003).
Jenkins, P.M. et al. E-cadherin polarity is determined by a multifunction motif mediating lateral membrane retention through ankyrin-G and apical-lateral transcytosis through clathrin. J. Biol. Chem. 288, 14018–14031 (2013).
Scheffler, B. et al. Phenotypic and functional characterization of adult brain neuropoiesis. Proc. Natl. Acad. Sci. USA 102, 9353–9358 (2005).
Sheikh, F., Fandrich, R.R., Kardami, E. & Cattini, P.A. Overexpression of long or short FGFR-1 results in FGF-2–mediated proliferation in neonatal cardiac myocyte cultures. Cardiovasc. Res. 42, 696–705 (1999).
Lim, D.A. et al. Chromatin remodeling factor Mll1 is essential for neurogenesis from postnatal neural stem cells. Nature 458, 529–533 (2009).
Mirzadeh, Z., Merkle, F.T., Soriano-Navarro, M., Garcia-Verdugo, J.M. & Alvarez-Buylla, A. Neural stem cells confer unique pinwheel architecture to the ventricular surface in neurogenic regions of the adult brain. Cell Stem Cell 3, 265–278 (2008).
Kao, Y.H. & Sterling, P. Matching neural morphology to molecular expression: single cell injection following immunostaining. J. Neurocytol. 32, 245–251 (2003).
Mirzadeh, Z., Doetsch, F., Sawamoto, K., Wichterle, H. & Alvarez-Buylla, A. The subventricular zone en-face: wholemount staining and ependymal flow. J. Vis. Exp. 39, 1938 (2010).
Mank, M. et al. A genetically encoded calcium indicator for chronic in vivo two-photon imaging. Nat. Methods 5, 805–811 (2008).
Beaulieu, J.M. et al. A beta-arrestin 2 signaling complex mediates lithium action on behavior. Cell 132, 125–136 (2008).
Abdi, K.M. & Bennett, V. Adducin promotes micrometer-scale organization of beta2-spectrin in lateral membranes of bronchial epithelial cells. Mol. Biol. Cell 19, 536–545 (2008).
Acknowledgements
We thank A. Gittis (Carnegie Mellon), J. Yakel (National Institute of Environmental Health Sciences), J. Ting (Massachusetts Institute of Technology), M. Prado (Robarts Research Institute), N. Calakos, M. Caron and V. Bennett for helpful discussions, D. Kleinfeld (University of California, San Diego) for M1-CNiFER cells, N. Kessaris (University College London) for Gsx2-Cre driver, Duke Pathology EM Facility for help with sample preparation, Cold Spring Harbor Laboratory Ion Channel Physiology Course instructors N. Golding, A. Lee and M. Nolan for inspiration, K. Abdi, G. Lyons, Q. Xiao, M. Rinehart and D. Fromme for project assistance, and J. Grandl and G. Pitt for manuscript comments. This work was supported by the David & Lucile Packard Foundation, US National Institutes of Health grant R01NS078192, and a George & Jean Brumley Endowment (C.T.K.).
Author information
Authors and Affiliations
Contributions
P.P.-G. designed and performed the anatomical, IHC staining and biochemical experiments. B.A. and P.P.-G. designed and performed the in vivo optogenetic experiments. B.A., E.R. and C.T.K. designed and performed the electrophysiological recordings. B.A. and C.T.K. designed and performed the imaging experiments. C.T.K. conceived the project. P.P.-G., B.A. and E.R. assembled the figures. P.P.-G., B.A. and C.T.K. wrote the paper. All of the authors discussed results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Neurotransmitter effects on DCX+ neuroblast production in vitro.
(a) Representative DCX staining of differentiated SVZ adherent cultures treated pharmacologically. (b) Western blot analyses of protein lysates from individually treated culture plate wells, showing titratable increase in DCX protein levels following acetylcholine (ACh) treatment. Ser: serotonin; Dopa: dopamine; Glut: glutamate; Muscimol: GABA agonist. (c) Western blot analyses of control or ACh-treated (10 μm) cultures in the presence of muscarinic antagonist atropine (Atrop) or nicotinic antagonist mecamylamine (Mec). Western blot analyses for each condition were repeated 5 times with consistent results. Scale bar: 50 μm.
Supplementary Figure 2 Generation of Ank3-mutant ChAT+ neurons.
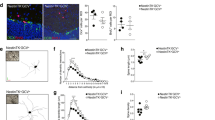
(a) Genomic map of ank3 floxed-allele showing insertion of LoxP sites around exons 22 and 23, within Spectrin binding domain of Ank3. (b) Western blot analysis of whole brain lysates from P0 animals, showing successful deletion of higher molecular weight isoforms of Ank3 in ank3KO/KO (KO) mutants (left panel). Western blot analysis of FACS-sorted ChAT+ neurons from ChATIRES-Cre/+; R26R-tdTomato mice, showing expression of higher molecular weight isoforms of Ank3 protein (right panel). (c) Ank3 IHC staining in tdTomato-labeled ChAT+ neurons in control (Ctrl) and Ank3-cKO (cKO) mice, showing proximal axonal segments (arrows). Imaris software was used to create 3D image reconstructions (lower panels). (d) Representative whole-cell, current-clamp recordings from striatal ChAT+ neurons in response to 100 ms of 250 or 500 pA current pulses in Ctrl, Het, and Ank3-cKO animals. (Right) Quantification of spike numbers to 100 ms of current pulse, 250 or 500 pA. * P < 0.002, t28 = 3.661, unpaired Student's t test, n = 15 in each group (5 animals). Box plots show mean, median, quartiles, range. Scale bar: 2 μm (c).
Supplementary Figure 3 SVZ neurogenesis in Nkx2.1-Cre; ChATflox/flox mice.
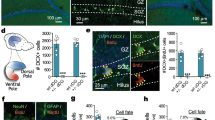
(a) Representative views of ventricular whole-mount DCX staining from P30 Nkx2.1-Cre; ChATflox/+ (Ctrl) and Nkx2.1-Cre; ChATflox/flox (cKO) mice. (b) Representative Ki67 and Mash1 IHC staining of SVZ niche from P30 Nkx2.1-Cre; ChATflox/+ (Ctrl) and Nkx2.1-Cre; ChATflox/flox (cKO) mice. Fluorescence signals inverted to black on white for clarity in both (a) and (b). (c) Quantifications of SVZ Ki67+, Mash1+, DCX+ cell numbers. n = 5. Box plots show mean, median, quartiles, range. Scale bars: 50 μm (a,b).
Supplementary Figure 4 Anatomical characteristics of subependymal ChAT+ neurons.
(a) IHC staining for tdTomato, ChAT, VAChT in ChATIRES-Cre/+; R26R-tdTomato transgenic mice, showing co-localization in subep-ChAT+ neurons. (b) Additional example traces for subependymal (Subep) or striatal ChAT+ neurons in ventricular and coronal views. (c) Quantifications of subependymal and striatal ChAT+ neuron dendritic branch point numbers. Box plots show mean, median, quartiles, range, n = 10. (d) 3D Sholl analyses of subependymal (Subep) and striatal (Str) ChAT+ neuron dendritic morphology. Box plots show mean, median, quartiles, range, n = 10. Scale bars: 20 μm (a), 50 μm (b).
Supplementary Figure 5 Comparisons of striatal and subependymal ChAT+ neuron electrophysiological properties.
(a) Representative whole-cell, current-clamp recordings of striatal and subep-ChAT+ neurons in response to 100 (red), 250 (green), or 500 pA (black) current injections for 100 ms. (Right) Number of evoked spikes during 100 ms current pulses of 100, 250, or 500 pA. Box plots show mean, median, quartiles, range, n = 15 in each group (5 animals). (b) Representative whole-cell current-clamp recordings of striatal and subep-ChAT+ neurons in response to 500 ms negative current injection (following 100 ms 20 pA positive current pulse). Dotted-lines indicate amplitude of sag potential. (c) Analyses of striatal and subep-ChAT+ neuron resting membrane potential, spike initiation threshold, and depolarization to spike, acquired by increasing depolarizing current pulses (100 ms duration) until spike initialization. Box plots show mean, median, quartiles, range, n = 15 (5 animals).
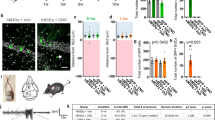
Supplementary Figure 6 M1-CNiFER experimental design.
Schematic representations of (a) M1-CNiFER cell detection of ACh; (b) M1-CNiFER cells transplanted into SVZ niche in acute slice preparation; (c) M1-CNiFER cells transplanted into SVZ niche in acute slice preparation from ChATIRES-Cre/+; R26R-ChR2EYFP mice.
Supplementary Figure 7 Nicotinic and muscarinic receptor expression in subependymal GFAP+ cells.
IHC staining of P30 SVZ coronal sections, with antibodies against α3-nicotinic (α3-nAChR), α4-nicotinic (α4-nAChR), or pan-muscarinic (mAChR) receptors (red); GFAP (blue); GFP (from ChAT-GFP transgene, green); and DAPI (red, in left panels). GFAP+ SVZ NSCs (white dashed-lines) touching the lateral ventricle (LV) are positive for nicotinic and muscarinic receptors (Imaris 3D). They are adjacent to GFP+ processes from ChAT+ neurons (arrows). Antibodies against nicotinic and muscarinic receptors that showed IHC specificity were made in rabbit, and were stained individually. Scale bar: 10 μm.
Supplementary Figure 8 Detecting activity-dependent electrical currents in SVZ cell types.
(a) Schematic representation of experimental setup in P30 SVZ acute slices from nestin-CreERtm4 (N4); R26R-tdTomato; ChAT-ChR2EYFP mice. (b) Representative images of tdTomato+ subependymal NSCs in P30 brain slices, lineage-traced via nestin-CreERtm4; R26R-tdTomato following tamoxifen injection, filled with DiO (green) through glass micropipette. (c) Representative images showing DiO injections and subsequent IHC staining with Nestin, S100β, Mash1, or DCX antibodies. (d) Representative voltage-clamp recordings from tdTomato+ ependymal niche cells, Mash1+ “C” cells, DCX+ neuroblasts, following 10 ms 473 nm light pulses @ 30 Hz for 1 second (red traces are baselines without light). Inward current in DCX+ neuroblast is sensitive to cholinergic blockers mecamylamine (Mec., 40 μM), atropine (Atrop., 5 μM). Blue bar = duration of light-stimulation train. (e) Quantifications of light-evoked current responses in (d). * P < 0.0002, F2,8 = 34.49, one-way ANOVA, n = 5. Box plots show mean, median, quartiles, range. Scale bars: 5 μm (b,c).
Supplementary Figure 9 Optogenetic modulation of subependymal ChAT+ neuron activity in vivo.
(a,c) Schematic representations of in vivo light-stimulation. LV = lateral ventricle, Ctx = cortex.(b,d) Representative Ki67, Mash1, DCX, Nestin/Ki67 IHC staining of SVZ niche from P30 ChATIRES-Cre/+; R26R-ChR2EYFP (b) or ChATIRES-Cre/+; R26R-ArchGFP (d) mice, with (+) or without (–) 48 hrs of light-stimulation in vivo (473 nm for ChR2 or 556 nm for Arch). Fluorescence signals inverted to black on white for clarity. Representative subependymal Nestin+ cells (*) co-localizing with Ki67 (arrows) in right panels. (e,f) Representative p-rpS6 IHC staining of striatal ChAT+ neurons (arrows) adjacent to the SVZ, from P30 ChATIRES-Cre/+; R26R-ChR2EYFP mice following 48 hrs of 473 nm light-stimulation (e), or from P30 ChATIRES-Cre/+; R26R-ArchGFP mice following 48 hrs of 556 nm light-stimulation (f), comparing ipsilateral to uninduced contralateral striatum after ventricular stimulation, imaged at identical settings from same section. Cy5 channel used for p-rpS6 secondary antibody staining, for clarity represented in green channel for co-localization with tdTomato. Scale bars: 20 μm (b,d), 10 μm (e,f).
Supplementary Figure 10 Assessing in vivo injury responses from light-fiber implant.
(a) Representative Iba1, CD11b, NG2 IHC staining of SVZ niche from P30 ChATIRES-Cre/+; R26R-ChR2EYFP mice, with (+) or without (–) 48 hrs of 473 nm light-stimulation in vivo. Dashed-lines = proliferating SVZ regions. (b) Quantifications of SVZ Iba1, CD11b, NG2 IHC staining data from (a). P = 0.169, F2,12 = 2.07 (NG2); P = 0.175, F2,12 = 2.02 (NG2/Ki67). One-way ANOVA, n = 5. Box plots show mean, median, quartiles, range. (c) Representative Thbs4 IHC staining on SVZ sections, 48 and 84 hrs post cortical optical fiber implantation to target the lateral ventricle (LV). Note the delayed Thbs4 upregulation in SVZ niche ipsilateral (ipsil) to fiber implantation (arrows). contra = contralateral hemisphere. Scale bars: 20 μm (a), 50 μm (c).
Supplementary Figure 11 ACh and FGFR activation.
(a) Western blot analyses on effects of ACh or FGF (12 ng/ml) in SVZ NSC FGFR phosphorylation. Anti-FGF (α-FGF) was used at 13 μg/ml, except for “low” condition in lane 8 (6.5 μg/ml). (b) Western blot analysis of FGFR phosphorylation following application of either 100 μM ACh, or 12 ng/ml FGF to SVZ NSC adherent cultures. Note the lack of ACh-mediated increase in FGFR phosphorylation 90 minutes after treatment. (c) ELISA analyses of FGF concentrations in culture media during SVZ NSC differentiation in vitro, with ACh added (100 μM once at time zero) compared to control (no ACh). * P < 0.008, z = 2.739 (24 hrs), z = 2.611 (48 hrs), Wilcoxon two-sample test, n = 5. Box plots show mean, median, quartiles, range. (d) Western blot analysis of EGFR phosphorylation in SVZ NSC adherent cultures after treatment with 100 μM ACh, 20 ng/ml EGF (EGF Cond1), or 40 ng/ml EGF (EGF Cond2). (e) Western blot analyses on the effects of ACh and FGF on DCX+ neuroblast production in SVZ NSC adherent culture. All Western analyses (a,b,d,e) were repeated 5 times with consistent results. (f) Representative IHC staining and quantifications of phosphorylated FGFR (pFGFR) co-localization with GFAP+ SVZ NSCs in P30 niche using Imaris, showing decreased expression in Ank3-cKO animals compared to littermate controls. pFGFR fluorescence signal inverted to black on white for clarity. * P < 0.008, z = 2.611 Wilcoxon two-sample test, n = 5. Box plots show mean, median, quartiles, range. Scale bar: 10 μm (f).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 (PDF 9564 kb)
Rights and permissions
About this article
Cite this article
Paez-Gonzalez, P., Asrican, B., Rodriguez, E. et al. Identification of distinct ChAT+ neurons and activity-dependent control of postnatal SVZ neurogenesis. Nat Neurosci 17, 934–942 (2014). https://doi.org/10.1038/nn.3734
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3734
This article is cited by
-
Possible Engagement of Nicotinic Acetylcholine Receptors in Pathophysiology of Brain Ischemia-Induced Cognitive Impairment
Journal of Molecular Neuroscience (2022)
-
Locomotion dependent neuron-glia interactions control neurogenesis and regeneration in the adult zebrafish spinal cord
Nature Communications (2021)
-
Impaired olfactory neurogenesis affects the performance of olfactory-guided behavior in aged female opossums
Scientific Reports (2021)
-
Ependymal cells-CSF flow regulates stress-induced depression
Molecular Psychiatry (2021)
-
Extrinsic neuromodulation in the rodent olfactory bulb
Cell and Tissue Research (2021)