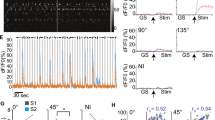
Abstract
Repeated viewing of an image over days and weeks induces a marked reduction in the strength with which neurons in monkey inferotemporal cortex respond to it. The processing advantage that attaches to this reduction is unknown. One possibility is that truncation of the response to a familiar image leaves neurons in a state of readiness to respond to ensuing images and thereby enhances their ability to track rapidly changing displays. We explored this possibility by assessing neuronal responses to familiar and novel images in rapid serial visual displays. Inferotemporal neurons responded more strongly to familiar than to novel images in such displays. The effect was stronger among putative inhibitory neurons than among putative excitatory neurons. A comparable effect occurred at the level of the scalp potential in humans. We conclude that long-term familiarization sharpens the response dynamics of neurons in both monkey and human extrastriate visual cortex.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ungerleider, L.G. & Mishkin, M. Two cortical visual systems. in Analysis of Visual Behavior (eds. D.J. Ingle, M.A. Goodale & R.J.W. Mansfield) 549–586 (MIT Press, Cambridge, Massachusetts, 1982).
Sobotka, S. & Ringo, J. Investigation of long-term recognition and association memory in unit responses from inferotemporal cortex. Exp. Brain Res. 96, 28–38 (1993).
Fahy, F.L., Riches, I.P. & Brown, M.W. Neuronal activity related to visual recognition memory: long-term memory and the encoding of recency and familiarity information in the primate anterior and medial inferior temporal and rhinal cortex. Exp. Brain Res. 96, 457–472 (1993).
Xiang, J.Z. & Brown, M.W. Differential neuronal encoding of novelty, familiarity and recency in regions of the anterior temporal lobe. Neuropharmacology 37, 657–676 (1998).
Freedman, D.J., Riesenhuber, M., Poggio, T. & Miller, E.K. Experience-dependent sharpening of visual shape selectivity in inferior temporal cortex. Cereb. Cortex 16, 1631–1644 (2006).
Mruczek, R.E.B. & Sheinberg, D.L. Context familiarity enhances target processing by inferior temporal cortex neurons. J. Neurosci. 27, 8533–8545 (2007).
Anderson, B., Mruczek, R.E.B., Kawasaki, K. & Sheinberg, D. Effects of familiarity on neural activity in monkey inferior temporal lobe. Cereb. Cortex 18, 2540–2552 (2008).
Woloszyn, L. & Sheinberg, D.L. Effects of long-term visual experience on responses of distinct classes of single units in inferior temporal cortex. Neuron 74, 193–205 (2012).
Anderson, B. & Sheinberg, D.L. Effects of temporal context and temporal expectancy on neural activity in inferior temporal cortex. Neuropsychologia 46, 947–957 (2008).
Sobotka, S. & Ringo, J. Investigation of long term recognition and association memory in unit responses from inferotemporal cortex. Exp. Brain Res. 96, 28–38 (1993).
De Baene, W. & Vogels, R. Effects of adaptation on the stimulus selectivity of macaque inferior temporal spiking activity and local field potentials. Cereb. Cortex 20, 2145–2165 (2010).
Desimone, R. Neural mechanisms for visual memory and their role in attention. Proc. Natl. Acad. Sci. USA 93, 13494–13499 (1996).
Li, L., Miller, E.K. & Desimone, R. The representation of stimulus familiarity in anterior inferior temporal cortex. J. Neurophysiol. 69, 1918–1929 (1993).
Liu, Y., Murray, S.O. & Jagadeesh, B. Time course and stimulus dependence of repetition-induced response suppression in inferotemporal cortex. J. Neurophysiol. 101, 418–436 (2009).
Lueschow, A., Miller, E.K. & Desimone, R. Inferior temporal mechanisms for invariant object recognition. Cereb. Cortex 4, 523–531 (1994).
McMahon, D.B.T. & Olson, C.R. Repetition suppression in monkey inferotemporal cortex: relation to behavioral priming. J. Neurophysiol. 97, 3532–3543 (2007).
Miller, E.K. & Desimone, R. Parallel neuronal mechanisms for short-term memory. Science 263, 520–522 (1994).
Miller, E.K., Gochin, P.M. & Gross, C.G. Habituation-like decrease in the responses of neurons in inferior temporal cortex of the macaque. Vis. Neurosci. 7, 357–362 (1991).
Miller, E.K., Li, L. & Desimone, R. A neural mechanism for working and recognition memory in inferior temporal cortex. Science 254, 1377–1379 (1991).
Miller, E.K., Li, L. & Desimone, R. Activity of neurons in anterior inferior temporal cortex during a short-term memory task. J. Neurosci. 13, 1460–1478 (1993).
Sawamura, H., Orban, G. & Vogels, R. Selectivity of neuronal adaptation does not match response selectivity: a single-cell study of the FMRI adaptation paradigm. Neuron 49, 307–318 (2006).
Sobótka, S. Involvement of single unit activity in inferotemporal and perirhinal cortices in recognition memory of visual objects in the macaque. Acta Neurobiol. Exp. (Warsz.) 60, 219–226 (2000).
Sobotka, S. & Ringo, J. Investigation of long term recognition and association memory in unit responses from inferotemporal cortex. Exp. Brain Res. 96, 28–38 (1993).
Sobotka, S. & Ringo, J. Mnemonic responses of single units recorded from monkey inferotemporal cortex, accessed via transcommissural versus direct pathways: a dissociation between unit activity and behavior. J. Neurosci. 16, 4222–4230 (1996).
Verhoef, B.-E., Kayaert, G., Franko, E., Vangeneugden, J. & Vogels, R. Stimulus similarity-contingent neural adaptation can be time and cortical area dependent. J. Neurosci. 28, 10631–10640 (2008).
Vogels, R., Sary, G. & Orban, G.A. How task-related are the responses of inferior temporal neurons? Vis. Neurosci. 12, 207–214 (1995).
Meyer, T. & Olson, C.R. Statistical learning of visual transitions in monkey inferotemporal cortex. Proc. Natl. Acad. Sci. USA 108, 19401–19406 (2011).
Intraub, H. Rapid conceptual identification of sequentially presented pictures. J. Exp. Psychol. Hum. Percept. Perform. 7, 604–610 (1981).
Földiák, P., Xiao, D., Keysers, C., Edwards, R. & Perrett, D.I. Rapid serial visual presentation for the determination of neural selectivity in area STSa. Prog. Brain Res. 144, 107–116 (2004).
Freiwald, W.A., Tsao, D.Y. & Livingstone, M.S. A face feature space in the macaque temporal lobe. Nat. Neurosci. 12, 1187–1196 (2009).
Kiani, R., Esteky, H., Mirpour, K. & Tanaka, K. Object category structure in response patterns of neuronal population in monkey inferior temporal cortex. J. Neurophysiol. 97, 4296–4309 (2007).
Allred, S.R. & Jagadeesh, B. Quantitative comparison between neural response in macaque inferotemporal cortex and behavioral discrimination of photographic images. J. Neurophysiol. 98, 1263–1277 (2007).
Peissig, J.J., Singer, J., Kawasaki, K. & Sheinberg, D.L. Effects of long-term object familiarity on event-related potentials in the monkey. Cereb. Cortex 17, 1323–1334 (2007).
Scott, L.S., Tanaka, J., Sheinberg, D. & Curran, T. The role of category learning in the acquisition and retention of perceptual expertise: a behavioral and neurophysiological study. Brain Res. 1210, 204–215 (2008).
Scott, L.S., Tanaka, J., Sheinberg, D. & Curran, T. A reevaluation of the electrophysiological correlates of expert object processing. J. Cogn. Neurosci. 18, 1453–1465 (2006).
Hölscher, C., Rolls, E.T. & Xiang, J. Perirhinal cortex neuronal activity related to long-term familiarity memory in the macaque. Eur. J. Neurosci. 18, 2037–2046 (2003).
Rainer, G. & Miller, E. Effects of visual experience on the representation of objects in the prefrontal cortex. Neuron 27, 179–189 (2000).
Fuhrmann, G., Markram, H. & Tsodyks, M. Spike frequency adaptation and neocortical rhythms. J. Neurophysiol. 88, 761–770 (2002).
Liu, Y.H. & Wang, X. Spike-frequency adaptation of a generalized leaky integrate-and-fire model neuron. J. Comput. Neurosci. 10, 25–45 (2001).
Sohal, V.S. & Hasselmo, M. A model for experience-dependent changes in the responses of inferotemporal neurons. Network 11, 169–190 (2000).
Bogacz, R., Brown, M. & Giraud-Carrier, C. Model of familiarity discrimination in the perirhinal cortex. J. Comput. Neurosci. 10, 5–23 (2001).
Moldakarimov, S.B., McClelland, J.L. & Ermentrout, G.B. A homeostatic rule for inhibitory synapses promotes temporal sharpening and cortical reorganization. Proc. Natl. Acad. Sci. USA 103, 16526–16531 (2006).
Ozeki, H., Finn, I.M., Schaffer, E.S., Miller, K.D. & Ferster, D. Inhibitory stabilization of the cortical network underlies visual surround suppression. Neuron 62, 578–592 (2009).
Tsodyks, M.V., Skaggs, W.E., Sejnowski, T.J. & McNaughton, B.L. Paradoxical effects of external modulation of inhibitory interneurons. J. Neurosci. 17, 4382–4388 (1997).
Tanaka, K., Saito, H., Fukada, Y. & Moriya, M. Coding visual images of objects in the inferotemporal cortex of the macaque monkey. J. Neurophysiol. 66, 170–189 (1991).
Feldman, D.E. Synaptic mechanisms for plasticity in neocortex. Annu. Rev. Neurosci. 32, 33–55 (2009).
Lewicki, M.S. Efficient coding of natural sounds. Nat. Neurosci. 5, 356–363 (2002).
Olshausen, B.A. & Field, D.J. Sparse coding of sensory inputs. Curr. Opin. Neurobiol. 14, 481–487 (2004).
Lehky, S.R., Kiani, R., Esteky, H. & Tanaka, K. Statistics of visual responses in primate inferotemporal cortex to object stimuli. J. Neurophysiol. 106, 1097–1117 (2011).
Rust, N.C. & Dicarlo, J. Selectivity and tolerance (“invariance”) both increase as visual information propagates from cortical area V4 to IT. J. Neurosci. 30, 12978–12995 (2010).
Delorme, A. & Makeig, S. EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 134, 9–21 (2004).
Acknowledgements
We thank K. McCracken for technical assistance. This work was supported by grants from the US National Institutes of Health (RO1 EY018620, P50 MH084053, K08 MH080329) and by the Pennsylvania Department of Health's Commonwealth Universal Research Enhancement Program. Technical support was funded by grants from the US National Institutes of Health (P30 EY08098 and P41RR03631).
Author information
Authors and Affiliations
Contributions
C.R.O. provided oversight and resources for the nonhuman primate study. T.M. and C.R.O. designed the experiment. T.M. trained the monkeys and collected the data. R.Y.C. provided oversight and resources for the human study. C.W. and R.Y.C. collected and analyzed the human data. All of the authors participated in the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Population activity of 29 neurons in Monkey 1
The difference between novel and familiar image strings with regard to the amplitude of driven periodic activity, as shown in (a), achieved statistical significance (p = 0.011, Mann-Whitney U-test, n = 29). The difference between novel and familiar image sequences with respect to the strength of the response to the trailing probe, as shown in (e-f), achieved statistical significance (p = 0.031, paired t-test on firing rate 100-200 ms following offset of the leading image, n = 29). Panels (a-b) observe the conventions of panels (a-b) in Fig. 2 and panels (c-f) observe the conventions of panels (a-d) in Fig. 5.
Supplementary Figure 2 Population activity of 37 neurons in Monkey 2
The difference between novel and familiar image strings with regard to the amplitude of driven periodic activity, as shown in (a), achieved statistical significance (p = 0.0013, Mann-Whitney U-test, n = 37). The difference between novel and familiar image sequences with respect to the strength of the response to the trailing probe, as shown in (e-f), achieved statistical significance (p = 0.000013, paired t-test on firing rate 100-200 ms following offset of the leading image, n = 37). Panels (a-b) observe the conventions of panels (a-b) in Fig. 2 and panels (c-f) observe the conventions of panels (a-d) in Fig. 5.
Supplementary Figure 3 Identical images elicit strong or weak periodic responses according to whether they are familiar or novel.
In six cases, we recorded from a neuron in monkey 1 and a neuron in monkey 2 using four images that were identical short of their training status. Two of the images were familiar to monkey 1 (set 1) and the other two were familiar to monkey 2 (set 2). Images familiar to monkey 1 elicited strong periodic activity in monkey 1 - red curve in (a) - and not in monkey 2 - red curve in (b). Images familiar to monkey 2 elicited strong periodic activity in monkey 2 - blue curve in (b) - and not in monkey 1 - blue curve in (a). Even in this small number of sessions, the difference between novel and familiar image strings with regard to the amplitude of driven periodic activity achieved significance (p = 0.031, Bootstrap test based on shuffling of novel and familiar trials).
Supplementary Figure 4 Neurons were distributed bimodally with respect to action potential width.
(a) Distribution across all 66 neurons of the interval in time between the maximal and minimal voltage attained by the average waveform during the millisecond following initiation. The best-fit Gaussian function is superimposed on each mode. Red and blue indicate neurons on the fast-spiking and regular-spiking sides of the cut in the distribution. (b) Average action-potential waveforms for all 66 neurons. Red and blue indicate waveforms classified as fast-spiking and regular-spiking. There is good within-category consistency with a single exception. (c) Fast-spiking neurons (red) tended to have a lower baseline firing rate than regular-spiking neurons (blue).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 1531 kb)
Rights and permissions
About this article
Cite this article
Meyer, T., Walker, C., Cho, R. et al. Image familiarization sharpens response dynamics of neurons in inferotemporal cortex. Nat Neurosci 17, 1388–1394 (2014). https://doi.org/10.1038/nn.3794
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3794
This article is cited by
-
Predicting the temporal-dynamic trajectories of cortical neuronal responses in non-human primates based on deep spiking neural network
Cognitive Neurodynamics (2023)
-
Perirhinal circuits for memory processing
Nature Reviews Neuroscience (2019)
-
How face perception unfolds over time
Nature Communications (2019)
-
Repetition suppression to objects is modulated by stimulus-specific expectations
Scientific Reports (2017)
-
Time-compressed preplay of anticipated events in human primary visual cortex
Nature Communications (2017)