Abstract
Astrocytes respond to chemical, electrical and mechanical stimuli with transient increases in intracellular calcium concentration ([Ca2+]i). We now show that astrocytes in situ display intrinsic [Ca2+]i oscillations that are not driven by neuronal activity. These spontaneous astrocytic oscillations can propagate as waves to neighboring astrocytes and trigger slowly decaying NMDA receptor-mediated inward currents in neurons located along the wave path. These findings show that astrocytes in situ can act as a primary source for generating neuronal activity in the mammalian central nervous system.
Similar content being viewed by others
Main
Until recently, glia have been regarded as having merely supportive roles for neurons in the CNS. This view shifted with the discovery that astrocytes in culture respond with increases in [Ca2+]i to synaptic stimulation and the application of neurotransmitters1,2,3. These evoked increases in [Ca2+]i propagate as Ca2+ waves through gap-junctionally coupled astrocytes in organotypic slice culture, prompting the suggestion that astrocytes can participate in a form of slow, long-distance information processing3.
Studies of co-cultured neurons and astrocytes4,5,6,7 and investigations in situ8,9,10 have shown that evoked [Ca2+]i increases in astrocytes can modulate the activity of adjacent neurons, and further studies have extended the potential roles of astrocytes in the CNS by demonstrating that, following stimulation, astrocytes release neurotransmitters such as glutamate11,12,13, through a probable exocytotic mechanism14.
These data have led to the recognition of bidirectional astrocyte–neuron communication10, and the emergence of the concept of the 'tripartite synapse'9,15,16, in which the astrocyte, in addition to pre- and postsynaptic neuronal elements, is a functional component of the synapse. However, in this scenario, astrocytes are still reactive elements that respond to synaptic activity with rises in [Ca2+]i and subsequently release neurotransmitters to have a feedback effect on synaptic transmission.
Here we describe spontaneous astrocytic [Ca2+]i oscillations in situ that are independent of synaptic activity. These oscillations underlie excitatory signaling to adjacent neurons, suggesting that astrocytes can generate neuronal activity, which significantly extends the known roles of astrocytes in the CNS.
Note: Supplementary information can be found on the Nature Neuroscience web site (http://neuroscience.nature.com/web_specials/).
Results
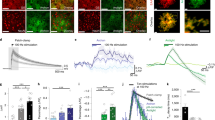
To investigate changes in astrocytic [Ca2+]i, freshly prepared slices of the rat ventrobasal (VB) thalamus were loaded with the cell-permeable forms of the Ca2+ indicator dyes Fluo-3 or Fluo-4. This method and preparation is a convenient way of investigating astrocytic [Ca2+]i activity because, due to their distinct morphology, astrocytes and neurons in the VB can be readily distinguished under phase contrast microscopy. In the presence of tetrodotoxin (TTX) to block synaptic activity, spontaneous [Ca2+]i increases were observed in astrocytes in all slices (n = 201) used in this study. Analysis of the spatial and temporal pattern of the astrocytic activity showed that spontaneous [Ca2+]i transients had a half-maximal duration of 15 ± 1 s (n = 56) and displayed a variety of activity profiles (astrocytes 1 to 12, Fig. 1a) throughout the imaged area (center, Fig. 1a). Most (67%) astrocytes (in 24 slices) exhibited two or more spontaneous [Ca2+]i transients during the recording period, which was usually 10 minutes long (astrocytes 2, 3 and 9, Fig. 1a), whereas the other active astrocytes displayed single spontaneous events (astrocytes 4, 5, 6 and 12, Fig. 1a). In all active astrocytes (n = 161) present in these 24 slices, a [Ca2+]i transient was seen on average every 333 ± 32 seconds, corresponding to a frequency of about 0.003 Hz, whereas in those astrocytes (n = 108) that showed multiple [Ca2+]i increases, the mean interval between these events was 98.9 ± 4 seconds, corresponding to an oscillation frequency of approximately 0.01 Hz.
(a) Center, image from an experiment showing the position of astrocytes exhibiting spontaneous [Ca2+]i transients in an area of VB thalamus. The surrounding graphs show percent ΔF versus time for the ringed and lettered astrocytes in the image; color codings denote groups of astrocytes that showed correlated activity. The central image was taken 260 s after beginning of the recording session, and thus astrocytes 6 and 7 exhibit high fluorescence, whereas astrocytes 3, 4 and 5 do not. (b) Probability profile of the incidence of experimental astrocytic activity patterns for all possible pairs of astrocytes (n = 874) against the probability of such combinations (bin width, 0.01). Data points from correlated activity combinations are plotted in blue, showing that lower-probability (≤0.48) cases are exclusively due to correlated activity, whereas uncorrelated activity (black bars) has exclusively higher probability (>0.48). A similar pattern of exclusive probability separation between correlated and uncorrelated combinations was also observed for larger groups of astrocytes. (c) Theoretical probability profile constructed according to derived theoretical probability distributions. Data points from activity combinations showing correlations are plotted in blue. (d) The difference in experimental (exp) and theoretical (th) incidence shows that there are more correlated combinations than expected. (e) Ratio of experimental to theoretical profiles, illustrating that, for example, correlated activity patterns at probabilities less than 0.01 in astrocyte pairs have an incidence 27 times greater than predicted. (f–h) Ratios of experimental to predicted probability profiles (shown for the regions corresponding exclusively to correlated activity patterns) for groups of 3, 4 and 5 astrocytes (bin widths, 0.01, 0.001 and 0.0001, respectively). Comparison of plots (e–h) indicates that, for larger groups of astrocytes, correlated activity has much lower probability, and so the experimental/theoretical ratio can be extremely large.
Neighboring astrocytes often exhibited coincident [Ca2+]i transients (astrocytes 3–5, 6–9 and 10–12, Fig. 1a). To determine whether the incidence of observed coincident astrocytic [Ca2+]i increases was significantly greater than would be expected by chance, we analyzed the relative times of [Ca2+]i peaks in all active astrocytes in a slice (n = 24 slices; Fig 1b–e). The incidence of experimentally observed patterns of events was plotted against their probability, thus producing the experimental probability profile (Fig. 1b). Astrocyte pairs were considered correlated if their activity patterns exhibited at least one coincident peak (peaks within 10 s of each other in the pair; see Methods). As expected from the relatively low frequency of [Ca2+]i transients (above), correlated activity patterns occupied an exclusively lower probability (≤0.48) region in the experimental probability profile (Fig. 1b, blue bars), whereas uncorrelated patterns occupied an exclusively higher probability (>0.48) region (Fig. 1b, black bars). The experimental profile was then compared to the theoretical probability profile (Fig. 1c), which was calculated according to the theoretical probability distributions (see Methods). The difference between the experimental and theoretical distributions (Fig. 1d) illustrates that there are more correlated patterns than expected by chance. The ratio of experimental to theoretical probability profiles (Fig. 1e) indicates that overall, correlated patterns in astrocyte pairs occurred 2.5 times more often than expected by chance. The same analysis done for all possible groups of 3, 4 and 5 astrocytes (Fig. 1f, g and h; n = 2690, 6156 and 10710) showed that the incidence of correlated patterns in groups of these sizes was 12, 59 and 316 times greater, respectively, than expected by chance. These results confirmed that coincident [Ca2+]i increases between groups of astrocytes do not occur solely by chance. For correlated pairs of astrocytes, the proportion of astrocytic [Ca2+]i transients in one cell that led to a transient in the other cell was 31 ± 3% (n = 24 slices), and the distance between correlated pairs of astrocytes was 73.01 ± 1.86 μm (n = 297), whereas that between uncorrelated pairs was 83.5 ± 0.68 μm (n = 577, p < 0.001).
To determine the origin and mechanism of the spontaneous astrocytic [Ca2+]i transients, we measured activity following the application of a number of pharmacological agents. There was no contribution of neuronal firing and action potential-evoked transmitter release on spontaneous astrocytic activity, as the number of active astrocytes was similar in the presence and absence of TTX (92 ± 11% of control, n = 4; Fig. 2). (TTX was present in all pharmacological experimental groups and all subsequent experiments in this study). In addition, in the presence of the broad spectrum glutamate receptor antagonist kynurenic acid (1 mM) and the GABAA receptor antagonist bicuculline (30 μM) the activity was 97 ± 16% of control (n = 5), and the further inclusion of the purinergic antagonist suramin (50 μM) brought this activity to 130 ± 10% of control (n = 4; Fig. 2). A block of low-voltage-activated Ca2+ channels by Ni2+ (1 mM) did not affect the number of [Ca2+]i transients (90 ± 9% of control, n = 12), whereas the N, P, Q high-voltage-activated Ca2+ channel blocker MVIIC (5 μM) produced a small reduction (to 68 ± 18%, p = 0.23, n = 5; Fig. 2). In contrast, the L-type Ca2+ channel blocker nifedipine (1 μM) caused a marked reduction in astrocytic activity (decrease to 35 ± 11%, p < 0.01, n = 7), and [Ca2+]i transients were virtually abolished by the non-specific Ca2+ channel blocker Co2+ (1 mM; 12 ± 10%, p < 0.001, n = 4) and by the intracellular calcium store depletors thapsigargin (1 μM; 13 ± 9%, p < 0.0001, n = 6) and cyclopiazonic acid (10 μM; 0%, p < 0.0001, n = 4; Fig. 2). The role of extracellular Ca2+ was investigated in the presence of 0 mM [Ca2+]o (and 2 mM EGTA), and spontaneous transients were virtually abolished under this experimental condition (10.7 ± 2.4%, p < 0.001, n = 9). However, single Ca2+ transients were observed in astrocytes many minutes following substitution with the 0 mM [Ca2+]o medium (3.6 ± 0.5 min, n = 30 cells), indicating that extracellular [Ca2+]o was not required for their expression. Similar results were obtained upon addition of Co2+ (3.7 ± 1.0 min, n = 9 cells). Therefore, the astrocytic [Ca2+]i transients observed in situ are spontaneous in nature, and their underlying mechanism involves [Ca2+]i release with a possible dependence on store-refilling via plasma membrane, dihydropyridine-sensitive high-voltage-activated Ca2+ channels.
The number of active astrocytes in a 350 × 260 μm area over 15 min in the presence of the indicated pharmacological agents is compared to the number in control conditions with no blockers. TTX, tetrodotoxin; Ni2+, nickel; kyn, kynurenic acid; bic, bicuculline; sur, suramin; nif, nifedipine; Co2+, cobalt; thap, thapsigargin; CPA, cyclopiazonic acid. TTX was present in all experimental conditions except the control groups, as indicated by the horizontal gray bar. *p < 0.01, **p < 0.0001.
The astrocytic identity of spontaneously active cells was confirmed after recording by immunohistochemical processing for the astrocytic marker vimentin17. Filling thalamocortical (TC) neurons in imaged areas with lucifer yellow and biocytin, using patch pipettes, allowed the mapping of cells showing [Ca2+]i transients within the imaged field (Fig. 3a). Subsequent immunohistochemical staining for the intraneuronally injected biocytin and the astrocytic marker vimentin showed that Ca2+ transients (Fig. 3d and e) were localized to the vimentin-stained astrocytes (18 of 27 active cells in 5 slices were stained; Fig. 3b and c). Furthermore, filling cells exhibiting [Ca2+]i transients with lucifer yellow via patch pipette always (n = 13/13) revealed cells with a characteristic small soma (8.88 ± 0.42 μm, n = 13) and restricted nebulous fine processes (Fig. 3f), which were morphologically distinct from the large-soma (18.55 ± 1.29 μm, n = 10) TC neurons.
(a) Fluorescent image of a VB slice, loaded with Fluo-4 AM, shows the position of spontaneously active cells (indicated by 1, 2 and 3) around a TC neuron (n) that had been filled with lucifer yellow and biocytin. Scale bar, 20 μm. (b) The same field as in (a) following processing of the slice for biocytin (purple TC neuron staining, n) and astrocyte-specific vimentin (brown staining). 1, 2 and 3, same astrocytes as in (a), stained with vimentin. (c) Higher magnification image of vimentin-stained astrocyte 3. Scale bar, 5 μm. (d) Percent ΔF over time for astrocyte 3. (e) Pseudocolor images of percent ΔF at the indicated times, superimposed over a monochrome image of astrocyte 3. Scale bar, 10 μm. (f) Fluorescence image of a spontaneously active astrocyte filled with lucifer yellow. Filling pipette can be seen to the right of soma. Scale bar, 10 μm.
One of the most characteristic and well-described features of astrocytic Ca2+ signaling in culture studies is the propagation of intercellular Ca2+ waves through astrocytic networks18. Our finding that astrocytes exhibit correlated [Ca2+]i increases indicates that there is communication among astrocytes. Imaging of astrocytes at high magnification showed that Ca2+ signals propagated along astrocytic processes (Fig. 4a). Spontaneous activity at astrocytic processes was correlated but delayed relative to the activity at the astrocyte soma (Fig. 4b–d), and the calculated speed of this intra-astrocytic Ca2+ wave propagation was 3.2 ± 0.72 μm/s (n = 6 astrocytes). Spontaneous [Ca2+]i increases also propagated between astrocytes. Activity in neighboring astrocytes was correlated and delayed (Figs. 4e–l and 5b, c and f), consistent with the propagation of activity between astrocytes. The speed of propagation of spontaneously occurring waves between astrocytes was 3.67 ± 0.92 μm/s (range, 0.6–8 μm/s, n = 12; Figs. 4 and 5), comparable to propagation velocities in culture studies19. Astrocytes in culture are normally assumed to be coupled via gap junctions7,18, though chemical transmission has also been suggested20. In our slices, filling single astrocytes with lucifer yellow via patch pipettes failed to show dye-coupling in adjacent astrocytes (n = 13/13), indicating a probable chemical rather than gap junctional mediated transmission mechanism.
(a) Example of spontaneous intra-astrocyte wave propagation. Fluorescence image of an astrocyte and processes. Scale bar, 10 μm. (To view supplementary movies of wave propagation, see the Nature Neuroscience web site, http://neurosci.nature.com/web_specials.) (b) Percent ΔF for soma and astrocyte process marked in (a). (c) Normalized ΔF plots displaying relative times of peaks in soma (blue line) and astrocyte process (red line) for (b). (d) Cross-correlation of activity in process against that in soma. (e) Fluorescence image of two adjacent astrocytes. Scale bars, 10 μm. (To view movie, see supplementary information.) (f) ΔF percent for astrocyte soma 1 and astrocytic process, and adjacent astrocyte soma 2. (g) Normalized ΔF plots displaying relative timings of peaks in astrocyte soma 1 (blue) and adjacent astrocyte soma 2 (green). (h) Cross-correlation of activity in the soma of astrocyte 1 and 2. (i) Fluorescence image showing astrocyte somas 1, 2 and 3. Scale bar, 20 μm. Percent ΔF for astrocyte somas 1, 2 and 3. (To view movie, see supplementary information.) (k) Normalized ΔF displaying relative timings of peaks in astrocyte somas 1 (blue), 2 (red) and 3 (green). (l) Cross-correlation of activity in somas 2 and 3 against that in astrocyte soma 1.
(a) Position of active astrocytes 1, 2 and 3, and the position of the recorded TC neuron soma (n). (b) Consecutive pseudocolor images (at times indicated) superimposed on a monochrome image, showing percent ΔF during a spontaneous wave that starts in astrocyte 1 and propagates over the position of recorded neuron n to astrocyte 2 and 3. (c) Percent ΔF for astrocytes 1, 2 and 3, and the current recorded from the neuron n. As the spontaneous wave passes through the position of the recorded neuron, a large inward current occurs (dotted lines in plots in c denote the time of current peak). Stimulation of a Ca2+ wave by local application of trans-ACPD (at site indicated by X in a, at time indicated by arrow in c) causes [Ca2+]i increases in astrocytes 1, 2 and 3, and an inward current in the recorded neuron (asterisk in current trace). (d) Expanded traces of spontaneously generated and trans-ACPD-evoked currents (shown in c) are normalized to the same amplitude for comparison. (e) Cross-correlations showing peaks close to zero for inward currents recorded in neuron (n) against ΔF in astrocytes 1 (blue), 2 (green) and 3 (red). (f) Relationship of astrocytic [Ca2+]i signal propagation to inward current. The time of the peak inward current is taken as time zero, and the position of the neuron as distance zero. The peak of astrocytic events occurring before the current have negative time values; those occurring afterward have positive time values. Astrocytes lying between the point of wave origin and the neuron soma are assigned negative distance values; those beyond the neuron in the direction of wave propagation are assigned positive distance values. Top, relationship for the experiment illustrated in (a), for astrocytes 1, 2 and 3. Bottom, data for 14 similar experiments, with values binned in distances of 10 μm. Data were fitted by linear regression (r2 = 0.98 and r2 = 0.84 for top and bottom plot, respectively). Experiments described in this figure were done in 0 mM Mg2+.
Astrocytic [Ca2+]i increases evoked by mechanical and chemical stimulation lead to the generation of large-scale Ca2+ waves and elicit neurotransmitter release, both of which have effects on adjacent neurons4,5,8. We therefore investigated whether TC neurons were activated during spontaneous astrocytic [Ca2+]i waves in the VB thalamus. Patch-clamp recordings from TC neurons in the absence of Mg2+, while simultaneously monitoring astrocytic [Ca2+]i activity, revealed the presence of large inward currents in 70% of recorded neurons (19/27). The inward currents were correlated with [Ca2+]i increases (see below; n = 15 of 19 neurons) and propagating waves in neighboring astrocytes (Fig. 5a and b). The magnitude of the currents (159 ± 35 pA; range, 40–435 pA; n = 17, in 15 neurons; Fig. 5c) would be expected to take the neuron membrane potential beyond the activation threshold of the fast Na+ current21. A striking feature of these inward currents was their kinetics (τact, 332 ± 115 ms, n = 17), and in particular, their very slow decay (τdecay, 2.7 ± 0.7 s, n = 17; Fig. 5d). Stimulation of astrocytic [Ca2+]i increases by trans-ACPD was also accompanied by slowly decaying inward currents in adjacent neurons (n = 4; Fig. 5c), confirming a causal role for astrocytic [Ca2+]i transients as the signal for the generation of the observed TC neuron inward currents.
To investigate the physiological significance of the inward currents evoked by spontaneous astrocytic activity, experiments were also done at 33°C in the presence of Mg2+. Inward currents were still observed (6 of 8 neurons; Fig. 6a) under physiological conditions. The duration of [Ca2+]i increase at 33°C was 10 ± 1 seconds (n = 46) at half-maximal amplitude, and the apparent speed of propagating astrocytic signaling was 7.6 ± 1.89 μm/s (n = 6). Inward currents under physiological conditions had an amplitude of 100 ± 30.3 pA (and therefore would still be capable of depolarizing the neuron sufficiently to elicit action potentials), and their kinetics included a τact of 145.1 ± 48 ms and a τdecay of 580 ± 356 ms (n = 12).
(a) Inward current recorded in a TC neuron in Mg2+ containing ACSF at 33°C, and percent ΔF from 3 adjacent astrocytes (astrocytes 1, 2 and 3, which were 12, 15 and 20 μm, respectively, from the tip of the recording electrode). (b) Probability profile of the experimentally observed incidence of current–[Ca2+]i in-crease activity patterns for all possible astrocyte–neuron pairs (bin width, 0.01). Data points from activity combinations exhibiting correlations are plotted in blue, showing that lower probability (≤0.6) cases are exclusively due to correlated activity, and that uncorrelated activity (black bars) has an exclusively higher probability (>0.6). (c) Theoretical probability profile of current–[Ca2+]i increase combinations generated using theoretical probability distributions. Data points corresponding to correlated activity patterns are plotted in blue. (d) The difference in experimental (exp) and theoretical probability profiles (th) shows that there are more correlated neuron–astrocyte pairs than expected by chance. (e) Ratio of the experimental to theoretical probability profile, illustrating that, for example, the incidence of correlated activity patterns probabilities less than 0.01 in astrocyte–neuron pairs is 86 times greater than expected by chance. (f–h) Ratios of experimental to theoretical probability profiles (shown for the region corresponding exclusively to correlated activity patterns) for an inward current and all possible groups of 2, 3 and 4 astrocytes (bin widths, 0.001, 0.00002 and 0.00002, respectively). (i) Recordings of spontaneous inward currents under control conditions, in the presence of 100 μM APV and following wash off of APV. (j) Number of spontaneous inward currents observed under these conditions and reversibly blocked by 100 μM APV, in 4 neurons.
Analysis of the relative times of neuronal inward currents and [Ca2+]i peaks in different astrocytes in a slice showed that the degree of correlation with astrocytic [Ca2+]i increases was much greater than would be expected by chance (Fig. 6b, blue bars). Examination of all possible pairs of a TC neuron and an astrocyte (n = 65) in a slice showed that overall there were six times more correlated pairs than expected by chance (Fig. 6b–e). Inward currents were also correlated to coordinated transients in groups of astrocytes, with analysis revealing that the incidence of correlated activity patterns between a neuron and groups of 2, 3, 4 and 5 astrocytes (n = 151, 217, 212 and 143, respectively) was 57, 600, 8333 and 160,000 times greater, respectively, than expected by chance (Fig. 6f–h). The results confirm that neuronal inward currents are correlated to astrocytic [Ca2+]i increases. The distance between the neuron and astrocytes displaying correlated activity was 34.51 ± 3.5 μm (n = 33), compared with maximum dendritic extensions for VB TC neurons of 70.2 ± 9.3 μm (n = 10).
Although the mechanism underlying spontaneous astrocyte–neuron signaling in this study is unknown, a correlation of neuronal inward currents with astrocytic [Ca2+]i increases clearly shows the involvement of astrocytic [Ca2+]i. In astrocytes displaying multiple [Ca2+]i increases (for example, astrocyte 2, Fig. 6a), not all [Ca2+]i transients resulted in neuronal inward currents. Comparison of the relative percent change in fluorescence (ΔF) of the [Ca2+]i increase correlated with the neuronal inward current with that uncorrelated to the inward currents showed that the uncorrelated [Ca2+]i transients were 38 ± 7% smaller (p < 0.005, n = 15) than the correlated events. This indicates that the astrocyte–neuron signaling process depends on the magnitude of the astrocytic [Ca2+]i increase, consistent with a Ca2+-dependent process.
As astrocytic [Ca2+]i transients are blocked by CPA, it might also be expected that astrocytic intracellular store depletion by CPA would also block the appearance of neuronal inward currents recorded from TC neurons. In the presence of CPA, the amplitude and frequency of inward currents were reduced relative to control and wash conditions, and τdecay was faster (amplitude, control, 153 ± 29 pA, n = 15; CPA, 101 ± 12 pA, n = 10; wash, 176 ± 32 pA, n = 14; frequency, control, 0.005 Hz; CPA, 0.0034 Hz; wash, 0.0054 Hz; τdecay, control, 260 ± 61 ms; CPA, 165 ± 19 ms; wash, 242 ± 40 ms). Inward currents are therefore not abolished by CPA treatment, though the observed effects are consistent with a reduction in the astrocyte activity correlated to the inward current component.
Glutamate is released from astrocytes following stimuli that increase [Ca2+]i11,12. The slow kinetics of the neuronal currents observed during spontaneous astrocytic waves suggested an involvement of the NR2D subtype22,23 of NMDA receptors, which gives rise to particularly long-lasting neuronal currents and in the VB shows high expression at postnatal day 10 but disappears by day 21 (ref. 24). Indeed, spontaneously occurring slowly decaying inward currents were reversibly abolished by the NMDA receptor antagonist d,l-2-amino-5-phosphonopentanoic acid (APV; 100 μM; n = 4/4; Fig. 6i and j), indicating that they were NMDA receptor-mediated currents. The effect of APV could not be explained by an effect on astrocytic [Ca2+]i transients, as the number of active astrocytes and the magnitude of astrocytic [Ca2+]i transients were unaffected by APV (91 ± 9% and 119 ± 22.6% of control, respectively, n = 4 slices), in agreement with the lack of effect of kynurenic acid (Fig. 2).
NMDA receptor activation would be expected to lead to neuronal Ca2+ entry, and indeed, in TC neurons (n = 3/3) patch-clamped with electrodes containing Fluo-4 (200 μM), we observed [Ca2+]i increases that followed spontaneous [Ca2+]i transients in adjacent astrocytes (Fig. 7a and b), indicating that as well as electrical excitability, astrocyte-driven signaling to neurons results in neuronal biochemical changes.
(a) Fluorescent image of a TC neuron (n) filled with Fluo-4 via the patch pipette and adjacent astrocyte (arrow). Images below show pseudocolor images representing percent ΔF transients in the astrocyte followed by increases in the neuron soma at indicated times. Scale bar, 10 μm. (b) Percent ΔF versus time for the astrocyte–neuron pair in (a), and the current recorded from the neuron soma, showing the relative timings of the astrocytic [Ca2+]i increases and the neuronal inward current and [Ca2+]i increase (0 mM Mg2+). (c) Developmental time course of spontaneous astrocytic [Ca2+]i activity. The number of active astrocytes are plotted against the postnatal age (n = 4–12 slices), *p < 0.05, **p < 0.005.
Recordings made at different postnatal days showed that the occurrence of spontaneous astrocytic [Ca2+]i transients exhibited a clear developmental profile (Fig. 7c). The number of spontaneously active astrocytes remained steady until postnatal day 9 (24 ± 3 active cells/area, n = 12 slices), and then declined to 13 ± 2 at day 11 (n = 6, p < 0.05, compared to day 9) and 7 ± 3 by day 14 (n = 4, p < 0.005, compared to day 9). In slices older than 14 days, the number of spontaneous astrocytes was 6 ± 1 (n = 7, p < 0.0005). As the observed reduction in spontaneous [Ca2+]i transients might have reflected a decreased dye loading with age25, trans-ACPD (100 μM) was added to the perfusate to elicit astrocytic [Ca2+]i increases10,26 and so reveal the number of loaded astrocytes at different postnatal ages. The number of astrocytes activated by trans-ACPD was 52 ± 5 per area at day 9 (n = 3) and 42 ± 2 at day 14 (n = 4, p = 0.051). Therefore, whereas 19% fewer astrocytes were loaded at day 14 compared to day 9, the number of spontaneously active astrocytes was reduced by 71%, demonstrating that the observed developmental decrease in the number of astrocytes exhibiting intrinsic [Ca2+]i transients was not due to a loading artifact.
Discussion
This study shows that astrocytes in situ display spontaneous [Ca2+]i oscillations and cause NMDA-receptor-mediated neuronal excitation. This result challenges the established view that astrocytes are purely reactive elements by demonstrating that they can be a source for the generation of neuronal activity in the mammalian CNS.
Spontaneous astrocytic [Ca2+]i oscillations in situ have not previously been described and so represent a new form of astrocytic activity. The oscillations are spontaneous in nature in that they are independent of synaptic activity and are not affected by antagonists of thalamic neurotransmitters. The oscillations are abolished by intracellular Ca2+ store depletors and so are dependent on intracellular Ca2+ release.
Spontaneous activity was seen to propagate as waves within single astrocytes and between neighboring astrocytes, a property of astrocytic [Ca2+]i signaling that is well described in culture1,2,3. Such signal propagation was directly observed in situ between pairs of astrocytes, whereas analysis showed that groups of up to five astrocytes could be involved in correlated activity, thus indicating signal propagation within these groups. The sequence of propagation within these groups, however, was not as clear as might be expected from studies in culture, presumably due to the greater complexity of the spatial arrangement in the three-dimensional slice compared to monolayer culture.
Evoked rises in astrocytic [Ca2+]i result in the release of neurotransmitter11,12,13. This agrees with our observations that large inward currents seen in TC neurons were preceded by spontaneous [Ca2+]i increases in adjacent astrocytes, and the finding that evoked [Ca2+]i increases in astrocytes also resulted in TC neuron inward currents supports a causal link between the spontaneous astrocytic [Ca2+]i transients and TC neuron activation.
The neuronal currents were blocked by APV, most likely because of astrocytic glutamate release following an increase in astrocytic [Ca2+]i. However, our experiments do not rule out the possibility that the astrocytes are releasing another factor that activates presynaptic afferents to release glutamate onto the TC neuron27. Although the absence of an AMPA component could argue against such a synaptic effect28, it is known that AMPA receptor expression is low in the thalamus during the developmental period examined in this study and that EPSPs are mostly generated via NMDA receptor activation29. The long NMDA responses could reflect the activation of the NR2D subtype of NMDA receptors22,23; as the location of these receptors is believed to be mainly extrasynaptic23, this would then point to an extrasynaptic effect of the astrocyte-released glutamate30. It is also possible that the observed long duration of the NMDA responses are not due to the NMDA receptor properties but to the slow release of glutamate from astrocytes elicited by the long-lasting [Ca2+]i increases. Whatever the mechanism of glutamate release, the causative role of the astrocytic [Ca2+]i increase in eliciting excitatory currents in TC neurons is unchallenged. CPA failed to block inward currents in TC neurons, as might be expected from an effect on blocking astrocytic [Ca2+]i release. As CPA can cause an increase in miniature excitatory postsynaptic current frequency by a mechanism involving the activation of store-operated calcium channels31, it is possible that a similar mechanism could be occurring in presynaptic terminals in the VB, or indeed in the astrocytes themselves.
The observation that spontaneous astrocytic activity is developmentally regulated raises issues about the participation of spontaneous astrocyte–neuron signaling in development. Spontaneous neuronal activity, which can be manifested as single neuron activity32 or correlated activity in neuronal domains25,33, is a well-described feature of the developing CNS that is essential for strengthening of synaptic connections and the establishment of neuronal pathways25,34,35,36. The spontaneous astrocyte–neuron signaling described in this study represents a previously unrecognized form of TTX-insensitive, developmentally regulated spontaneous activity.
The period over which the spontaneous astrocytic [Ca2+]i oscillations are observed is an important time in rat VB development, as TC neurons are elaborating distal dendrites onto which cortico-thalamic afferents synapse to complete the thalamocortical loop37,38,39. Indeed, the involvement of NMDA receptor activation points to a possible role in dendritic arborization40 or synaptic strengthening41, whereas the concomitant Ca2+ entry in TC neurons could be a signal for many neuronal biochemical processes42,43.
In this study we show that small groups of astrocytes display correlated activity and lead to neuronal excitation. This suggests that a small and fairly specific cluster of neurons would be excited by an active astrocytic group at any one time. This property, and the potential for the NMDA-induced depolarizations to elicit action potentials and therefore long-range signaling, endow this form of astrocyte–neuron signaling with properties required for a role in the topographic mapping of the thalamocortical loop.
Whereas glia are known to provide structural and trophic support for neurons during development and migration44,45,46, an active role for astrocytes in generating neuronal activity would be a significant extension for glial participation in brain development. Despite our observed developmental profile, it is possible that spontaneous astrocytic activity leading to TC neuron excitation persists to some extent in adulthood. If this were the case, the findings described here would have even more profound implications for the involvement of astrocytes in brain function.
Methods
Slice preparation and incubation.
Slices of neonate rat VB thalamus were prepared as described previously21 from 5–17 day old rats. After a recovery period of 1 h, slices were loaded with Fluo-3 or Fluo-4 AM (Molecular Probes, Eugene, Oregon) by incubating for 40–60 minutes at 28°C with 5 μM of the indicator dye and 0.01% pluronic acid. Under these conditions, astrocytes were preferentially loaded with the fluorescent indicator, whereas loading of neurons was rarely observed. All experiments were done in standard artificial cerebrospinal fluid (ACSF) composed of 120 mM NaCl, 16 mM NaHCO3, 2 mM KCl, 1.25 mM KH2PO4, 1 mM MgSO4 and 2 mM CaCl2 (ref. 47). TTX (1 μM) was also present in all experiments except the control group for data in Fig. 2. For Ca2+-free ACSF, Ca2+ was replaced with 2 mM EGTA. Experiments were conducted at room temperature (20–24°C) except those described in Fig. 6. All experimental solutions contained 1 mM Mg2+ except those used for Figs. 5, 6i, 7a and 7b. Chemicals were obtained from Sigma (St. Louis, Missouri), except d,l-APV and trans-ACPD (Tocris, Bristol, UK). For the pharmacology experiments in Fig. 2, following a control recording period in normal ACSF, pharmacological agents were included in the perfusing ACSF and washed over the preparation at the beginning of the experimental period. The number of active astrocytes seen during this experimental period was then expressed as a percentage of the number seen during the control period. Data are displayed as mean ± s.e.m. Significance was calculated using a two-tailed Student's t-test.
All animal experiments were approved by the Ethical Review Committee of Cardiff University, and conducted according to local, national and EU guidelines. Care was taken to minimize suffering and number of animals used in each experiment.
Confocal fluorescence imaging.
The recording chamber and manipulators were mounted on a moveable top plate platform (MP MTP-01, Burleigh, UK). Fluorescence was measured using a Noran Odyssey confocal microscope (Noran, UK). Averages of 8 whole field images were routinely acquired every 0.8–5 s. Acquisition and image analysis were done using Noran Intervision software. Fluorescence values over time for specific regions of interest were exported into Sigmaplot (Jandel, Chicago, Illinois) for further analysis and construction of plot of percent ΔF. Displayed monochrome images showing slice and cellular morphology were produced by averaging in Intervision; contrast and brightness were also adjusted to enhance morphological information. Images of percent ΔF were produced by dividing images during Ca2+ changes by baseline images and multiplying by the appropriate scaling factor using the Intervision 2D-analysis Math Function. Linear pseudocolor maps were then assigned, and images were exported to Adobe Photoshop (Adobe, San Jose, California) and merged with monochrome background images for display purposes. For stimulation of astrocytic Ca2+ increases in astrocytes, patch pipettes were filled with 100 μM trans-ACPD dissolved in ACSF. Pipettes were positioned above visually identified astrocytes in the slice and pressure applied to the attached tube.
Patch clamping.
Patch-clamp recordings were made using sylgarded pipettes (2–4 MΩ) containing an internal solution composed of 120 mM KMeSO4, 10 mM HEPES, 10 mM EGTA, 4 mM Na2ATP and 0.5 mM GTP. Typical input resistances of patched TC neurons were over 200 MΩ (238 ± 26 MΩ, n = 16). Biocytin (1%) and/or lucifer yellow (1%) or tetrapotassium Fluo-4 (200 μM) were included in the internal solution as indicated. Recording was started after 20 min of attaining whole cell patch to allow intracellular diffusion of pipette contents. In the recordings conducted at room temperature (Fig. 4), Mg2+ was replaced by Ca2+ in the ACSF. To study spontaneous astrocyte–neuron signaling under physiological conditions, neurons were held at –50 mV (the resting membrane potential of VB TC neurons during this developmental period48) and experiments were conducted at 33°C with standard 1 mM Mg2+ and 2 mM Ca2+ ACSF (as used in all other experiments in this study). Currents were recorded using an Axopatch 200B amplifier, and acquired and analyzed using PClamp8 software (Axon Instruments, Foster City, California).
Immunohistochemistry.
Slices were fixed in 2% paraformaldahyde for 30 min, then washed with 0.1% phosphate buffered saline (PBS). For biocytin-filled neuron visualization the avidin-biotin-peroxidase complex (ABC) method was used (ABC elite kit, Vector labs, Burlingame, California). After blocking endogenous peroxidase with 0.3% H2O2 in 0.4% Triton-X-100 and 0.1% PBS, slices were incubated in ABC for 3.5 h followed by 1 min development in very intense purple (VIP) substrate (Vector labs). For vimentin staining, non-specific binding was blocked with 10% horse serum for 30 min. Slices were incubated with anti-vimentin antibody (Chemicon, Temecula, California) at 1:4 dilution in 0.4% Triton-X-100, 1% horse serum in 0.1% PBS for 12 h at 4°C, and then 2 h at room temperature. Following a 1-h wash in PBS, the slices were incubated with the biotinylated secondary antibody (anti-mouse, Vector labs) for 3.5 h. After a 3-h incubation in ABC and a 12-h wash in 0.1% PBS, vimentin staining was visualized with 1 min DAB+ (Dako Labs, Ely, UK) substrate.
Correlation and probability analysis.
Cross-correlation plots were generated in Sigmaplot. A transform was written implementing a standard algorithm for generating discrete correlation data, taking advantage of the in-built fast Fourier transform (FFT) and inverse FFT algorithms. The transform was applied to normalized fluorescence traces to produce the final cross-correlation data.
On the basis of the average half-maximal duration of [Ca2+]i transients and their average speed of propagation, coincidence was defined as [Ca2+]i peaks being within 10 s of each other. We considered the occurrence of [Ca2+]i peaks on a predefined time interval T (the total recording time) that was subdivided into N bins (time windows) of size ΔT (= T/N) = 10 s. With this value of ΔT, no two peaks in the same trace occurred in the same bin. Each peak was assumed to occur randomly and independently from all others with uniform distribution over T (that is, over the N bins) in each cell. Theoretically, we then defined m (>0) peaks occurring in k different cells to be coincident if they all occurred within the same bin. (In practice, time bins were dynamically adjusted with reference to peaks in other traces to ensure that all possible coincidences were accounted for).
The probability that there are m1 coincident peaks between a pair of cells that exhibit n1 and n2 peaks is given by the following equation.

Here, (n/m) are the binomial coefficients.
If we consider a third trace (cell) exhibiting n3 peaks, the probability that there are m2 coincident peaks between the three traces is

where P(m1;m1,n3,N) and P(m1;n1,n2,N) are of the same form given in Eq. 1.
In general, if there are k traces (cells) exhibiting n1...nk peaks, the probability that exactly m coincident peaks occur among them by chance is given by the following equation.

These probabilities form a discrete distribution of m.

Thus, for a given number of experimental observations of an activity-grouping of distinct (n1...nk, N) type, this distribution shows how often certain values of m would occur by chance. The distribution also clearly indicates how often we would expect to see activity patterns with particular probabilities. Thus, by considering all the different activity-grouping types encountered for a given value of k (the number of cells), a theoretical 'probability profile' was generated that predicted how often one would expect to encounter activity patterns with certain probabilities. Because correlated activity patterns occur exclusively with low probability, comparison of the theoretical and experimental probability profiles enabled us to ascertain whether or not coincident [Ca2+]i peaks between astrocytes occurred solely by chance. This method was also used to assess coincidences between [Ca2+]i transients in astrocytes and inward currents in neurons.
References
Cornell-Bell, A. H., Finkbeiner, S. M., Cooper, M. S. & Smith, S. J. Glutamate induces calcium waves in cultured astrocytes: long range glial signaling. Science 247, 470–473 (1990).
Charles, A. C., Merril, J. E., Ditksen, E. R. & Sanderson, M. J. Intercellular signaling in glial cells: calcium waves and oscillations in response to mechanical stimulation and glutamate. Neuron 6, 983–992 (1991).
Dani, J. W., Chernavsky, A. & Smith, S. J. Neuronal activity triggers calcium waves in hippocampal astrocytic networks. Neuron 8, 429–440 (1992).
Araque, A., Parpura, V., Sanzgiri, R. P. & Haydon, P. G. Glutamate dependent astrocyte modulation of synaptic transmission between cultured hippocampal neurones. Eur. J. Neurosci. 10, 2129–2142 (1998).
Hassinger, T. D.,. et al. Evidence for glutamate-mediated activation of hippocampal neurons by glial calcium waves. J. Neurobiol. 28, 159–170 (1995).
Charles, A. C. Glia–neuron intercellular Ca2+ signaling. Dev. Neurosci. 16, 196–206 (1994).
Nedergaard, M. Direct signaling from astrocytes to neurons in cultures of mammalian brain cells. Science 263, 1768–1771 (1994).
Newman, E. A. & Zahs, K. R. Modulation of neuronal activity by glial cells in the retina. J. Neurosci. 18, 4022–4028 (1998).
Kang, J., Jiang, L., Goldman, S. A. & Nedergaard, M. Astrocyte-mediated potentiation of inhibitory synaptic transmission. Nat. Neurosci. 1, 683–692 (1998).
Pasti, L., Volterra, A., Pozzan, T. & Carmignoto, G. Intracellular calcium oscillations in astrocytes: a highly plastic, bi-directional form of communication between neurons and astrocytes in situ. J. Neurosci, 17, 7817–7830 (1997).
Parpura, V., Basarsky, T. A., Liu, F., Jeftinija, K. & Haydon P. G. Glutamate-mediated astrocyte–neuron signaling. Nature 369, 744–747 (1994).
Bezzi, P. et al. Prostaglandins stimulate calcium dependent glutamate release in astrocytes. Nature 391, 281–285 (1998).
Innocenti, B., Parpura, V. & Haydon P. G. Imaging extracellular waves of glutamate during calcium signaling in cultured astrocytes. J. Neurosci. 20, 1800–1808 (2000).
Araque, A., Li, N., Doyle, R. T. & Haydon, P. G. Snare protein dependent glutamate release from astrocytes. J. Neurosci. 20, 666–673 (2000).
Araque, A., Parpura, V., Sanzgiri, R. P. & Haydon, P. G. Tripartite synapse: glia the unacknowledged partner. Trends Neurosci. 22, 208–215 (1999).
Carmignoto, G. Reciprocal communication systems between astrocytes and neurons. Prog. Neurobiol. 62, 561–581 (2000).
Schnitzer, J., Franke, W. W. & Schachner, M. Immunocytochemical demonstration of vimentin in astrocytes and ependymal cells of developing and adult mouse nervous system. J. Cell. Biol. 90, 435–447 (1981).
Giaume, C. & Venance, L. Intercellular calcium signaling and gap junctional communication in astrocytes. Glia 24, 50–64 (1998).
Finkbeiner, S. M. Glial calcium. Glia 9, 83–104 (1993).
Guthrie, P. B. et al. ATP released from astrocytes mediates glial calcium waves. J. Neurosci. 19, 520–528 (1999).
Parri, H. R. & Crunelli, V. Sodium current in thalamocortical neurons: role of a non-inactivating component in tonic and burst firing. J. Neurosci. 18, 854–867 (1998).
Monyer, H., Burnashev, N., Laurie, D. J., Sakmann, B. & Seeburg, P. H. Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron 12, 529–540 (1994).
Wyllie, D. J. A., Behe, B. & Colquhoun D. Single-channel activation and concentration jumps: comparison of recombinant NR1a/NR2A and NR1a/NR2D NMDA receptors. J. Physiol. (Lond.) 510, 1–18 (1998).
Wenzel, A., Villa, M., Mohler, H. & Benke, D. Developmental and regional expression of NMDA receptor subtypes containing the NR2D subunit in rat brain. J. Neurochem. 66, 1240–1247 (1996).
Yuste, R., Peinado, A. & Katz, L. C. Neuronal domains in developing neocortex. Science 257, 665–669 (1992).
Porter, J. T. & Mccarthy, K. D. GFAP positive hippocampal astrocytes in situ respond to glutamatergic neuroligands with increases in [Ca]i . Glia 13, 101–112 (1995).
Araque, A., Sanzgiri, R. P., Parpura, V. & Haydon, P. G. Calcium elevation in astrocytes causes an NMDA receptor dependent increase in the frequency of miniature synaptic currents in cultured hippocampal neurons. J. Neurosci. 18, 6822–6829 (1998).
Spreafico, R. et al. Distribution of AMPA selective glutamate receptors in the thalamus of adult rats and during postnatal development. A light and ultrastructural immunocytochemical study. Brain Res. Dev. Brain Res. 82, 231–244 (1994).
Golshani, P., Warren, R. A. & Jones, E. G. Progression of change in NMDA, non-NMDA and metabotropic glutamate receptor function at the developing corticothalamic synapse. J. Neurophysiol. 80, 143–154 (1998).
Misra, S. C., Brickley, S. G., Wyllie, D. J. & Cull-Candy, S. G. Slow deactivation kinetics of NMDA receptors containing NR1 and NR2D subunits in rat cerebellar purkinje cells. J. Physiol. (Lond.) 525, 299–305 (2000).
Emptage, N. J, Reid., C. A. & Fine, A. Calcium stores in hippocampal synaptic boutons mediate short-term plasticity, store operated calcium entry and spontaneous transmitter release. Neuron 29, 197–208 (20 01).
Gu, X. & Spitzer, N. C. Distinct aspects of neuronal differentiation encoded by frequency of spontaneous Ca2+ transients. Nature 375, 784–787 (1995).
Yuste, R., Nelson, D. A., Rubin, W. W. & Katz, L. C. Neuronal domains in developing neocortex: mechanisms of coactivation. Neuron 14, 7–17 (1995).
Goodman, C. S. & Shatz, C. J. Developmental mechanisms that generate precise patterns of neuronal connectivity. Cell 72S, 77–98 (1993).
Katz, L. C. & Shatz, C. J. Synaptic activity and the construction of cortical circuits. Science 274, 1133–1138 (1996).
Catalano, S. M. & Shatz C. J. Activity-dependent cortical target selection by thalamic axons. Science 281, 559–562 (1998).
Miller, B., Chou, L. & Finlay, B. L. The early development of thalamocortical and corticothalamic projections. J. Comp. Neurol. 335, 16–41 (1993).
Liu, X. B., Honda, C. N. & Jones, E. G. Distribution of four types of synapse on physiologically identified relay neurons in the ventral posterior thalamic nucleus of the cat. J. Comp. Neurol. 352, 69–91 (1995).
Zantua, J. B., Wasserstrom, S. P., Arends, J. J. A., Jacquin, M. F. & Woolsey, T. A. Postnatal development of mouse “whisker” thalamus: ventroposterior medial nucleus (VPM), barreloids, and their thalamocortical relay neurons. Somatosens. Motor Res. 13, 307–322 (1996).
Rajan, I. & Cline, H. T. Glutamate receptor activity is required for normal development of tectal cell dendrites in vivo. J. Neurosci. 18, 7836–7846 (1998).
Collingridge, G. C. & Bliss, T.V. Memories of NMDA receptors and LTP. Trends Neurosci. 18, 54–56 (1995).
Berridge, M. J. Neuronal calcium signaling. Neuron 21, 13–26 (1998).
Perkel, D. J., Petrozzino, J. J., Nicoll, R. A. & Connor, J. A. The role of Ca2+ entry via synaptically activated NMDA receptors in the induction of long term potentiation. Neuron 11, 817–823 (1993).
Silver, J., Lopez, S. E., Wahlsten, D. & Coughlin, J. Axonal guidance during development of the great cerebral commisures, descriptive and experimental studies in vivo on the role of preformed glial pathways. J. Comp. Neurol. 210, 10–29 (1982).
Rakic, P., Cameron, R. S. & Komuro, H. Recognition adhesion, transmembrane signaling and cell motility in guided neuronal migration. Curr. Opin. Neurobiol. 4, 63–69 (1994).
Lois, C., Garcia-Verdugo, J.-M. & Alvarez-Buylla, A. Chain migration of neuronal precursors. Science 271, 978–981 (1996).
Turner, J. P., Leresche, N., Guyon, A., Soltesz, I. & Crunelli, V. Sensory input and burst firing output of rat and cat thalamocortical cells: the role of NMDA and non-NMDA receptors. J. Physiol. (Lond.) 480, 281–295 (1994).
Perez Velazquez, J. L. & Carlen, P. L. Development of firing patterns and electrical properties in neurons of the rat ventrobasal thalamus. Dev. Brain. Res. 91, 164–170 (1996).
Acknowledgements
The authors wish to thank S.W. Hughes and T.I. Tóth for data analysis and the development of specialized statistical analysis software, and V.H. Perry for assistance with the immunocytochemistry analysis. The work was supported by the Wellcome Trust (grant 37089–98).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Parri, H., Gould, T. & Crunelli, V. Spontaneous astrocytic Ca2+ oscillations in situ drive NMDAR-mediated neuronal excitation. Nat Neurosci 4, 803–812 (2001). https://doi.org/10.1038/90507
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/90507
This article is cited by
-
Analog neuromorphic circuit for spontaneous Ca2+ oscillations
Scientific Reports (2023)
-
Loss of neuron network coherence induced by virus-infected astrocytes: a model study
Scientific Reports (2023)
-
Dysfunctional serotonergic neuron-astrocyte signaling in depressive-like states
Molecular Psychiatry (2023)
-
Dysregulation of astrocytic Ca2+ signaling and gliotransmitter release in mouse models of α-synucleinopathies
Acta Neuropathologica (2023)
-
Dynamical analysis of astrocyte-induced neuronal hyper-excitation
Nonlinear Dynamics (2023)