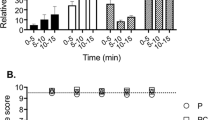
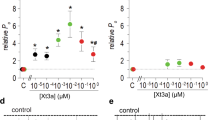
Abstract
Cone snails use venom containing a cocktail of peptides ('conopeptides') to capture their prey. Many of these peptides also target mammalian receptors, often with exquisite selectivity. Here we report the discovery of two new classes of conopeptides. One class targets α1-adrenoceptors (ρ-TIA from the fish-hunting Conus tulipa), and the second class targets the neuronal noradrenaline transporter (χ-MrIA and χ-MrIB from the mollusk-hunting C. marmoreus). ρ-TIA and χ-MrIA selectively modulate these important membrane-bound proteins. Both peptides act as reversible non-competitive inhibitors and provide alternative avenues for the identification of inhibitor drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Olivera, B. M. et al. Diversity of Conus neuropeptides. Science 249, 257–263 (1990).
Olivera, B. M., McIntosh, J. M., Cruz, L. J., Luque, F. A. & Gray, W. R. Purification and sequence of a presynaptic peptide toxin from Conus geographus venom. Biochemistry 23, 5087–5090 (1984).
Cruz, L. J. et al. Conus geographus toxins that discriminate between neuronal and muscle sodium channels. J. Biol. Chem. 260, 9280–9288 (1985).
Fainzilber, M., Kofman, O., Zlotkin, E. & Gordon, D. A new neurotoxin receptor site on sodium channels is identified by a conotoxin that affects sodium channel inactivation in molluscs and acts as an antagonist in rat brain. J. Biol. Chem. 269, 2574–2580 (1994).
McIntosh, J. M. et al. A new family of conotoxins that blocks voltage-gated sodium channels. J. Biol. Chem. 270, 16796–16802 (1995).
Craig, A. G. et al. An O-glycosylated neuroexcitatory Conus peptide. Biochemistry 37, 16019–16025 (1998).
Shon, K. J. et al. κ-Conotoxin PVIIA is a peptide inhibiting the shaker K+ channel. J. Biol. Chem. 273, 33–38 (1998).
Gray, W. R., Luque, A., Olivera, B. M., Barrett, J. & Cruz, L. J. Peptide toxins from Conus geographus venom. J. Biol. Chem. 256, 4734–4740 (1981).
Hopkins, C. et al. A new family of Conus peptides targeted to the nicotinic acetylcholine receptor. J. Biol. Chem. 270, 22361–22367 (1995).
Shon, K. J. et al. A noncompetitive peptide inhibitor of the nicotinic acetylcholine receptor from Conus purpurascens venom. Biochemistry 36, 9581–9587 (1997).
England, L. J. et al. Inactivation of a serotonin-gated ion channel by a polypeptide toxin from marine snails. Science 281, 575–578 (1998).
Mena, E. E. et al. Conantokin-G: a novel peptide antagonist to the N-methyl-D-aspartic acid (NMDA) receptor. Neurosci. Lett. 118, 241–244 (1990).
Cruz, L. J. et al. Invertebrate vasopressin/oxytocin homologs. Characterization of peptides from Conus geographus and Conus striatus venoms. J. Biol. Chem. 262, 15821–15824 (1987).
Craig, A. G. et al. Contulakin-G, an O-glycosylated invertebrate neurotensin. J. Biol. Chem. 274, 13752–13759 (1999).
Adams, D. J., Alewood, P. F., Craik, D. J., Drinkwater, R. D. & Lewis, R. J. Conotoxins and their potential pharmaceutical applications. Drug Dev. Res. 46, 219–234 (1999).
McGrath, J. C. Adrenergic and 'non-adrenergic' components in the contractile response of the vas deferens to a single indirect stimulus. J. Physiol. (Lond.) 283, 23–39 (1978).
Sneddon, P. & Westfall, D. P. Pharmacological evidence that adenosine triphosphate and noradrenaline are co-transmitters in the guinea-pig vas deferens. J. Physiol. (Lond.) 347, 561–580 (1984).
Minneman, K. P., Fox, A. W. & Abel, P. W. Occupancy of α1-adrenergic receptors and contraction of rat vas deferens. Mol. Pharmacol. 23, 359–368 (1983).
Kenakin, T. P. Pharmacologic Analysis of Drug–Receptor Interaction (Lippincott-Raven, Philadelphia, Pennsylvania, 1997).
Hwa, J., Graham, R. M. & Perez, D. M. Identification of critical determinants of α1-adrenergic receptor subtype selective agonist binding. J. Biol. Chem. 270, 23189–23195 (1995).
Trendelenburg, U., Maxwell, R. A. & Pluchino, S. Methoxamine as a tool to assess the importance of intraneuronal uptake of l-norepinephrine in the cat's nictitating membrane. J. Pharmacol. Exp. Ther. 172, 91–99 (1970).
Northover, B. J. A comparison of the electrophysiological actions of phentolamine with those of some other antiarrhythmic drugs on tissues isolated from the rat heart. Br. J. Pharmacol. 80, 85–93 (1983).
Pérez, O., Valenzuela, C., Delpón, E. & Tamargo, J. Class I and III antiarrhythmic actions of prazosin in guinea-pig papillary muscles. Br. J. Pharmacol. 111, 717–722 (1994).
Przywara, D. A. & Dambach, G. E. Direct actions of cocaine on cardiac cellular electrical activity. Circ. Res. 65, 185–192 (1989).
Cusack, B., Nelson, A. & Richelson, E. Binding of antidepressants to human brain receptors: focus on newer generation compounds. Psychopharmacology (Berl.) 114, 559–565 (1994).
McIntosh, J. M. et al. Isolation and characterization of a novel Conus peptide with apparent antinociceptive activity. J. Biol. Chem. 275, 32391–32397 (2000).
Fürst, S. Transmitters involved in antinociception in the spinal cord. Brain Res. Bull. 48, 129–141 (1999).
Hu, S. H. et al. The 1.1 Å resolution crystal structure of [Tyr15]-EpI, a novel α-conotoxin from Conus episcopatus, solved by direct methods. Biochemistry 37, 11425–11433 (1998).
Hu, S. H. et al. The 1.1 Å crystal structure of the neuronal acetylcholine receptor antagonist, α-conotoxin PnIA from Conus pennaceus. Structure 4, 417–423 (1996).
Hu, S. H., Gehrmann, J., Alewood, P. F., Craik, D. J. & Martin, J. L. Crystal structure at 1.1 Å resolution of α-conotoxin PnIB: comparison with α-conotoxins PnIA and GI. Biochemistry 36, 11323–11330 (1997).
Balaji, R. A. et al. λ-Conotoxins, a new family of conotoxins with unique disulfide pattern and protein folding. Isolation and characterization from the venom of Conus marmoreus. J. Biol. Chem. 275, 39516–39522 (2000).
Massot, O. et al. 5-hydroxytryptamine-moduline, a new endogenous cerebral peptide, controls the serotonergic activity via its specific interaction with 5-hydroxytryptamine1B/1D receptors. Mol. Pharmacol. 50, 752–762 (1996).
Olivera, B. M. & Cruz, L. J. Conotoxins, in retrospect. Toxicon 39, 7–14 (2001).
Loughnan, M. et al. α-Conotoxin EpI, a novel sulfated peptide from Conus episcopatus that selectively targets neuronal nicotinic acetylcholine receptors. J. Biol. Chem. 273, 15667–15674 (1998).
Brüss, M., Pörzgen, P., Bryan-Lluka, L. J. & Bönisch, H. The rat norepinephrine transporter: molecular cloning from PC12 cells and functional expression. Brain Res. Mol. 52, 257–262 (1997).
Percy, E. et al. Catechol-O-methyltransferase activity in CHO cells expressing norepinephrine transporter. Br. J. Pharmacol. 128, 774–780 (1999).
Nielsen, K. J. et al. Effects of chirality at Tyr13 on the structure–activity relationships of ω-conotoxins from Conus magus. Biochemistry 38, 6741–6751 (1999).
Fainzilber, M. et al. New mollusc-specific α-conotoxins block Aplysia neuronal acetylcholine receptors. Biochemistry 33, 9523–9529 (1994).
Acknowledgements
We thank R. Graham (Sydney) and D. Kaye (Melbourne) for assistance and advice and for providing the α1B-adrenoceptor (R.G.) and human NET (D.K.) clones. L. Bryan-Lluka (Brisbane) provided the rat NET clone. A. Jones, N. Daly, K. Nielsen and T. Bond (I.M.B.) provided assistance. This work was supported by grants from AusIndustry and the National Health and Medical Research Council, Australia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharpe, I., Gehrmann, J., Loughnan, M. et al. Two new classes of conopeptides inhibit the α1-adrenoceptor and noradrenaline transporter. Nat Neurosci 4, 902–907 (2001). https://doi.org/10.1038/nn0901-902
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn0901-902
This article is cited by
-
Enhancing the Stability of Tumor Homing LyP-1 Peptide Using Cyclization and Retro Grafting Strategies
International Journal of Peptide Research and Therapeutics (2023)
-
The α1-adrenoceptor inhibitor ρ-TIA facilitates net hunting in piscivorous Conus tulipa
Scientific Reports (2019)
-
Ginsentides: Cysteine and Glycine-rich Peptides from the Ginseng Family with Unusual Disulfide Connectivity
Scientific Reports (2018)
-
Ancestral protein resurrection and engineering opportunities of the mamba aminergic toxins
Scientific Reports (2017)
-
High-throughput identification of novel conotoxins from the Chinese tubular cone snail (Conus betulinus) by multi-transcriptome sequencing
GigaScience (2016)