Abstract
Sensory transduction in hair cells requires assembly of membrane-bound transduction channels, extracellular tip-links and intracellular adaptation motors with sufficient precision to confer nanometer displacement sensitivity. Here we present evidence based on FM1-43 fluorescence, scanning electron microscopy and RT-PCR that these three essential elements are acquired concurrently between embryonic day 16 and 17, several days after the appearance of hair bundles1, and that their acquisition coincides with the onset of mechanotransduction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Denman-Johnson, K. & Forge, A. J. Neurocytol. 28, 821–835 (1999).
Denk, W., Holt, J.R., Shepherd, G.M. & Corey D.P. Neuron 15, 1311–1321 (1995).
Holt, J.R. et al. Cell 108, 371–381 (2002).
Kros, C.J. et al. Nat. Neurosci. 5, 41–47 (2002).
Sahly, I., El-Amraoui, A., Abitbol, M., Petit, C. & Dufier, J.L. Anat. Embryol. (Berl.) 196, 159–170 (1997).
Hasson, T. et al. J. Cell Biol. 137, 1287–1307 (1997).
Kachar, B., Parakkal, M., Kurc, M., Zhao, Y. & Gillespie, P.G. Proc. Natl. Acad. Sci. USA 97, 13336–13341 (2000).
Meyers, J.R. et al. J. Neurosci. 23, 4054–4065 (2003).
Géléoc, G.S.G., Lennan, G.W.T., Richardson, G.P. & Kros, C.J. Proc. R. Soc. Lond. 264, 611–621 (1997).
Holt, J.R., Corey, D.P. & Eatock, R.A. J. Neurosci. 17, 8739–8748 (1997).
Wu, Y.C., Ricci, A.J. & Fettiplace, R. J. Neurophysiol. 82, 2171–2181 (1999).
Holt, J.R. & Corey, D.P. Proc. Natl. Acad. Sci. USA 97, 11730–11735 (2000).
Crawford, A.C., Evans, M.G. & Fettiplace, R. J. Physiol. 419, 405–434 (1989).
Ratto, G.M., Robinson, D.W., Yan, B. & McNaughton, P.A. Nature 351, 654–657 (1991).
Acknowledgements
We thank D. Abraham, J. Assad, J. Risner and E. Stauffer for comments on an earlier version of the manuscript. This work was supported by NIH grants to J.R.H. (DC05439-03) and to G.S.G.G.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
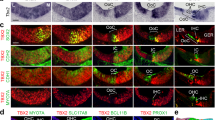
Supplementary Fig. 1.
Gentamicin blocks FM 1-43 uptake in E17 vestibular hair cells. (a) DIC image focused at the level of the hair bundles. The scale bar (10 mm) applies to all images. (b) Fluorescence image of the field corresponding to 'a' focused at the level of the cell bodies. The epithelium was exposed to 1 μM gentamicin for 5 minutes prior and during FM 1-43 treatment. (c) Fluorescence image of the same field 5 minutes after washout of gentamicin. The epithelium was then retreated with FM 1-43. (d) Histogram showing the average fluorescence of 128 gentamicin-treated cells (orange, solid bars) and 136 cells after washout of gentamicin (green, open bars). The gentamicin-treated cells had a mean brightness of 99 ± 205 a.u. After gentamicin washout the mean brightness was 1866 ± 1338 a.u. The broader distribution relative to control E17 cells was likely due to incomplete gentamicin recovery in some cells. (JPG 51 kb)
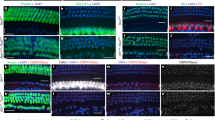
Supplementary Fig. 2.
Acquisition of channels and mechanotransduction between E16 and E17. (a) Representative fluorescence image of hair cell epithelium at E16 + 6 hours focused at the level of the cell bodies. 2 of 21 cells in this field took up FM 1-43. Cells were counted as fluorescent if their brightness was more than 2 standard deviations below the mean fluorescence of E17 cells: 472 a.u. (b) Representative fluorescence image from an epithelium at E16 + 12 hours. 9 of 19 cells took up FM 1-43. (c)Representative image from an epithelium at E16 + 18 hours. 18 of 23 cells took up FM 1-43. (d) The percent of cells that took up FM 1-43 as a function of embryonic age. The number of fluorescent cells relative to the number examined is shown for each time point. The data were fitted with a Boltzmann function that had a mid point of 16.6 days. (e) A family of transduction currents recorded at E16 + 12 hours. The current decay in response to positive and negative steps and the rebounds at the end of the steps are indicative of adaptation. Three datasets were averaged and digitally filtered at 200Hz. Stimulus protocols are shown at the bottom. (f) A family of transduction currents recorded at E16 + 18 hours. Five datasets were averaged. (JPG 50 kb)
Rights and permissions
About this article
Cite this article
Géléoc, G., Holt, J. Developmental acquisition of sensory transduction in hair cells of the mouse inner ear. Nat Neurosci 6, 1019–1020 (2003). https://doi.org/10.1038/nn1120
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1120
This article is cited by
-
Improved TMC1 gene therapy restores hearing and balance in mice with genetic inner ear disorders
Nature Communications (2019)
-
Distinct functions of TMC channels: a comparative overview
Cellular and Molecular Life Sciences (2019)
-
Different rates of endocytic activity and vesicle transport from the apical and synaptic poles of the outer hair cell
HNO (2019)
-
Computational analysis of mRNA expression profiling in the inner ear reveals candidate transcription factors associated with proliferation, differentiation, and deafness
Human Genomics (2018)
-
The microRNA-183/96/182 Cluster is Essential for Stereociliary Bundle Formation and Function of Cochlear Sensory Hair Cells
Scientific Reports (2018)