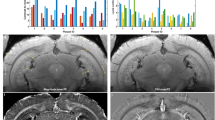
Abstract
Formation of senile plaques composed of amyloid β peptide, a pathological hallmark of Alzheimer disease, in human brains precedes disease onset by many years. Noninvasive detection of such plaques could be critical in presymptomatic diagnosis and could contribute to early preventive treatment strategies. Using amyloid precursor protein (APP) transgenic mice as a model of amyloid β amyloidosis, we demonstrate here that an intravenously administered 19F-containing amyloidophilic compound labels brain plaques and allows them to be visualized in living mice by magnetic resonance imaging (MRI) using 19F and 1H. Our findings provide a new direction for specific noninvasive amyloid imaging without the danger of exposure to radiation. This approach could be used in longitudinal studies in mouse models of Alzheimer disease to search for biomarkers associated with amyloid β pathology as well as to track disease course after treatment with candidate medications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Selkoe, D.J. Alzheimer's disease: a central role for amyloid. J. Neuropathol. Exp. Neurol. 53, 438–447 (1994).
Goedert, M., Trojanowski, J.Q. & Lee, V.M.-Y. The Molecular and Genetic Basis of Neurological Diseases 2nd edn. (Butterworth, Boston, 1997).
Hardy, J. & Selkoe, D.J. The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science 297, 353–356 (2002).
Gomez-Isla, T. et al. Profound loss of layer II entorhinal cortex neurons occur in very mild Alzheimer's disease. J. Neurosci. 16, 4491–4500 (1996).
Mathis, C.A. et al. A lipophilic thioflavin-T derivatives for position emission tomography (PET) imaging of amyloid in brain. Bioorg. Med. Chem. 12, 295–298 (2002).
Kung, M-P. et al. IMPY: an improved thioflavin-T derivative for in vivo labeling of β-amyloid plaques. Brain Res. 956, 202–210 (2002).
Klunk, W.E. et al. Imaging brain amyloid in Alzheimer's disease with Pittsburgh compound-B. Ann. Neurol. 55, 306–319 (2004).
Okamura, N. et al. Styrylbenzoxazol derivatives for in vivo imaging of amyloid plaques in the brain. J. Neurosci. 24, 2535–2541 (2004).
Shoghi-Jadid, K. et al. Localization of neurofibrillary tangles and beta-amyloid plaques in the brains of living patients with Alzheimer disease. Am. J. Geriatr. Psychiatry 10, 24–35 (2002).
Sair, H.I., Doraiswamy, P.M. & Petrella, J.R. In vivo amyloid imaging in Alzheimer's disease. Neuroradiology 46, 93–104 (2004).
Skovronsky, D.M. et al. In vivo detection of amyloid plaques in a mouse model of Alzheimer's disease. Proc. Natl. Acad. Sci. USA 97, 7609–7614 (2000).
Nordberg, A. PET imaging of amyloid in Alzheimer's disease. Lancet Neurol. 3, 519–527 (2004).
Bacskai, B.J. et al. Four-dimensional multiphoton imaging of brain entry, amyloid binding, and clearance of an amyloid-β ligand in transgenic mice. Proc. Natl. Acad. Sci. USA 100, 12462–12467 (2003).
Bacskai, B.J. et al. Imaging of amyloid β deposits in brains of living mice permits direct observation of clearance of plaques with immunotherapy. Nat. Med. 7, 369–372 (2001).
Benveniste, H., Einstein, G., Kim, K.R., Hulette, C. & Johnson, G.A. Detection of neuritic plaques in Alzheimer's disease by magnetic resonance microscopy. Proc. Natl. Acad. Sci. USA 96, 14079–14084 (1999).
Poduslo, J.F. et al. Molecular targeting of Alzheimer's disease amyloid plaques for contrast-enhanced magnetic resonance imaging. Neurobiol. Aging 11, 315–329 (2002).
Wadghiri, Y.Z. et al. Detection of Alzheimer's amyloid in transgenic mice using magnetic resonance microimaging. Magnet. Reson. Med. 50, 293–302 (2003).
Zhang, J. et al. Detection of amyloid plaques in mouse models of Alzheimer's disease by magnetic resonance imaging. Magnet. Reson. Med. 51, 452–457 (2004).
Helpern, J.A. et al. MRI assessment of neuropathology in a transgenic mouse model of Alzheimer's disease. Magnet. Reson. Med. 51, 794–798 (2004).
Lee, S.P. et al. Visualization of β-amyloid plaques in a transgenic mouse model of Alzheimer's disease using MR microscopy without contrast reagents. Magnet. Reson. In Med. 52, 538–544 (2004).
Jack, C.R. Jr. et al. In vivo visualization of Alzheimer's amyloid plaques by magnetic resonance imaging in transgenic mice without a contrast agent. Magnet. Reson. In Med. 52, 1263–1271 (2004).
Sato, K., Higuchi, M., Iwata, N., Saido, T.C. & Sasamoto, K. Fluoro-substituted and 13C-labeled styrylbenzene derivatives for detecting brain amyloid plaques. Eur. J. Med. Chem. 39, 573–578 (2004).
Hsiao, K. et al. Correlative memory deficits, Aβ elevation, and amyloid plaques in transgenic mice. Science 274, 99–102 (1996).
Zhuang, Z-P. et al. Radioiodinated styrylbenzenes and thioflavins as probes for amyloid aggregates. J. Med. Chem. 44, 1905–1914 (2001).
Lorenzo, A. & Yankner, B.A. β-amyloid neurotoxicity requires fibril formation and is inhibited by Congo Red. Proc. Natl. Acad. Sci. USA 91, 12243–12247 (1994).
Saido, T.C. Aβ Metabolism and Alzheimer's Disease (Landes Bioscience, Georgetown, Texas, USA, 2003).
The Ronald and Nancy Reagan Research Institute of the Alzheimer's Association and the NIA Working Group. Consensus report of the Working Group on: 'Molecular and Biochemical Markers of Alzheimer's Disease. Neurobiol. Aging 19, 109–116 (1998).
Saido, T.C. et al. Dominant and differential deposition of distinct beta-amyloid peptide species, AβN3(pE), in senile plaques. Neuron 14, 457–466 (1995).
Hennig, J., Nauerth, A. & Friedburg, H. RARE imaging: a fast imaging method for clinical MR. Magn. Reson. Med. 3, 823–833 (1986).
Acknowledgements
We thank M. Sekiguchi for technical assistance; K H. Ashe, University of Minnesota, for providing Tg2576; C. Itakura and K. Tanaka, RIKEN Brain Science Institute, for administrative support; and T. Iwatsubo, University of Tokyo, for helpful advice. This research was supported by research grants from the RIKEN Brain Science Institute and from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
In vivo 3D RARE 19F magnetic resonance image of a 20-month-old transgenic mouse brain; 60-min scan. (PDF 35 kb)
Supplementary Fig. 2
Magnetic resonance detection of aging-dependent amyloid β deposition in APP transgenic mice. (PDF 59 kb)
Rights and permissions
About this article
Cite this article
Higuchi, M., Iwata, N., Matsuba, Y. et al. 19F and 1H MRI detection of amyloid β plaques in vivo. Nat Neurosci 8, 527–533 (2005). https://doi.org/10.1038/nn1422
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1422
This article is cited by
-
Biodegradable polyphosphoester micelles act as both background-free 31P magnetic resonance imaging agents and drug nanocarriers
Nature Communications (2023)
-
Multiscale optical and optoacoustic imaging of amyloid-β deposits in mice
Nature Biomedical Engineering (2022)
-
Network activity changes in the pathophysiology of Alzheimer’s disease: the role of aging and early entorhinal cortex dysfunction
Metabolic Brain Disease (2022)
-
Evaluation of Styrylbenzene analog- FSB and its affinity to bind parenchymal plaques and tangles in patients of Alzheimer’s disease
Metabolic Brain Disease (2022)
-
Turn-on chemiluminescence probes and dual-amplification of signal for detection of amyloid beta species in vivo
Nature Communications (2020)