Abstract
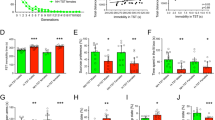
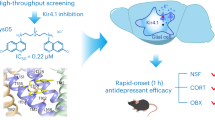
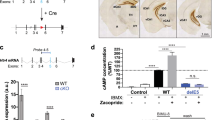
Depression is a devastating illness with a lifetime prevalence of up to 20%. The neurotransmitter serotonin or 5-hydroxytryptamine (5-HT) is involved in the pathophysiology of depression and in the effects of antidepressant treatments. However, molecular alterations that underlie the pathology or treatment of depression are still poorly understood. The TREK-1 protein is a background K+ channel regulated by various neurotransmitters including 5-HT. In mice, the deletion of its gene (Kcnk2, also called TREK-1) led to animals with an increased efficacy of 5-HT neurotransmission and a resistance to depression in five different models and a substantially reduced elevation of corticosterone levels under stress. TREK-1–deficient (Kcnk2−/−) mice showed behavior similar to that of naive animals treated with classical antidepressants such as fluoxetine. Our results indicate that alterations in the functioning, regulation or both of the TREK-1 channel may alter mood, and that this particular K+ channel may be a potential target for new antidepressants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lapin, I. & Oxenkrug, G. Intensification of the central serotoninergic processes as a possible determinant of the thymoleptic effect. Lancet 1, 132–136 (1969).
Coppen, A. The biochemistry of affective disorders. Br. J. Psychiatry 113, 1237–1264 (1967).
Delgado, P. et al. Tryptophan-depletion challenge in depressed patients treated with desipramine or fluoxetine: implications for the role of serotonin in the mechanism of antidepressant action. Biol. Psychiatry 46, 212–220 (1999).
Nestler, E.J. et al. Neurobiology of depression. Neuron 34, 13–25 (2002).
Maes, M. & Meltzer, H. The serotonin hypothesis of major depression. in Psychopharmacology The Fourth Generation of Progress (eds. Bloom, F.E. & Kupfer, D.J.) 933–944 (Raven, New York, 1995).
Owens, M. Molecular and cellular mechanisms of antidepressant drugs. Depress. Anxiety 4, 153–159 (1996).
Haddjeri, N., Blier, P. & de Montigny, C. Long-term antidepressant treatments result in a tonic activation of forebrain 5–HT1A receptors. J. Neurosci. 18, 10150–10156 (1998).
Mayorga, A. et al. Antidepressant-like behavioral effects in 5-hydroxytryptamine(1A) and 5-hydroxytryptamine(1B) receptor mutant mice. J. Pharmacol. Exp. Ther. 298, 1101–1107 (2001).
Fink, M. et al. Cloning, functional expression and brain localization of a novel unconventional outward rectifier K+ channel. EMBO J. 15, 6854–6862 (1996).
Lesage, F. & Lazdunski, M. Molecular and functional properties of two-pore-domain potassium channels. Am. J. Physiol. Renal Physiol. 279, F793–F801 (2000).
Patel, A.J. & Honore, E. Properties and modulation of mammalian 2P domain K+ channels. Trends Neurosci. 24, 339–346 (2001).
Patel, A.J. et al. A mammalian two pore domain mechano-gated S-like K+ channel. EMBO J. 17, 4283–4290 (1998).
Chemin, J. et al. Mechanisms underlying excitatory effects of group I metabotropic glutamate receptors via inhibition of 2P domain K+ channels. EMBO J. 22, 5403–5411 (2003).
Siegelbaum, S.A., Camardo, J.S. & Kandel, E.R. Serotonin and cyclic AMP close single K+ channels in Aplysia sensory neurones. Nature 299, 413–417 (1982).
Kandel, E.R. & Schwartz, J.H. Molecular biology of learning: modulation of transmitter release. Science 218, 433–443 (1982).
Talley, E.M., Solorzano, G., Lei, Q., Kim, D. & Bayliss, D.A. CNS distribution of members of the two-pore-domain (KCNK) potassium channel family. J. Neurosci. 21, 7491–7505 (2001).
Maingret, F. et al. TREK-1 is a heat-activated background K(+) channel. EMBO J. 19, 2483–2491 (2000).
Heurteaux, C. et al. TREK-1, a K(+) channel involved in neuroprotection and general anesthesia. EMBO J. 23, 2684–2695 (2004).
Porsolt, R.D., Le Pichon, M. & Jalfre, M. Depression: a new animal model sensitive to antidepressant treatments. Nature 266, 730–732 (1977).
Nestler, E.J. et al. Preclinical models: status of basic research in depression. Biol. Psychiatry 52, 503–528 (2002).
Cryan, J.F., Page, M.E. & Lucki, I. Noradrenergic lesions differentially alter the antidepressant-like effects of reboxetine in a modified forced swim test. Eur. J. Pharmacol. 436, 197–205 (2002).
Steru, L., Chermat, R., Thierry, B. & Simon, P. The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology (Berl.) 85, 367–370 (1985).
Ripoll, N., David, D., Dailly, E., Hascoet, M. & Bourin, M. Antidepressant-like effects in various mice strains in the tail suspension. Behav. Brain Res. 143, 193–200 (2003).
Cryan, J. & Holmes, A. The ascent of mouse: advances in modelling human depression and anxiety. Nat. Rev. Drug Discov. 4, 775–790 (2005).
Kameyama, T., Nagasaka, M. & Yamada, K. Effects of antidepressant drugs on a quickly-learned conditioned-suppression response in mice. Neuropharmacology 24, 285–290 (1985).
Maier, S. Learned helplessness and animal models of depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 8, 435–446 (1984).
Caldarone, B., George, T., Zachariou, V. & Picciotto, M. Gender differences in learned helplessness behavior are influenced by genetic background. Pharmacol. Biochem. Behav. 66, 811–817 (2000).
Caldarone, B. et al. Sex differences in response to oral amitriptyline in three animal models. Psychopharmacology (Berl.) 170, 94–101 (2003).
Fink, M. et al. A neuronal two P domain K+ channel stimulated by arachidonic acid and polyunsaturated fatty acids. EMBO J. 17, 3297–3308 (1998).
Reyes, R. et al. Immunolocalization of the arachidonic acid and mechanosensitive baseline TRAAK potassium channel in the nervous system. Neuroscience 95, 893–901 (2000).
Maingret, F., Fosset, M., Lesage, F., Lazdunski, M. & Honore, E. TRAAK is a mammalian neuronal mechano-gated K+ channel. J. Biol. Chem. 274, 1381–1387 (1999).
Sheline, Y.I., Wang, P.W., Gado, M.H., Csernansky, J.G. & Vannier, M.W. Hippocampal atrophy in recurrent major depression. Proc. Natl. Acad. Sci. USA 93, 3908–3913 (1996).
Heuser, I.J. et al. Pituitary-adrenal-system regulation and psychopathology during amitriptyline treatment in elderly depressed patients and normal comparison subjects. Am. J. Psychiatry 153, 93–99 (1996).
Hermann, G. et al. Stress-induced changes attributable to the sympathetic nervous system. J. Neuroimmunol. 53, 173–180 (1994).
Santarelli, L. et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 301, 805–809 (2003).
Malberg, J. Implications of adult hippocampal neurogenesis in antidepressant action. J. Psychiatry Neurosci. 29, 196–205 (2004).
Couillard-Despres, S. et al. Doublecortin expression levels in adult brain reflect neurogenesis. Eur. J. Neurosci. 21, 1–14 (2005).
Kennard, L.E. et al. Inhibition of the human two-pore domain potassium channel, TREK-1, by fluoxetine and its metabolite norfluoxetine. Br. J. Pharmacol. 144, 821–829 (2005).
Karson, C.N. et al. Human brain fluoxetine concentrations. J. Neuropsychiatry Clin. Neurosci. 5, 322–329 (1993).
Komoroski, R.A. et al. In vivo 19F spin relaxation and localized spectroscopy of fluoxetine in human brain. Magn. Reson. Med. 31, 204–211 (1994).
Bolo, N.R. et al. Brain pharmacokinetics and tissue distribution in vivo of fluvoxamine and fluoxetine by fluorine magnetic resonance spectroscopy. Neuropsychopharmacology 23, 428–438 (2000).
Redrobe, J., Dumont, Y., Fournier, A., Baker, G. & Quirion, R. Role of serotonin (5-HT) in the antidepressant-like properties of neuropeptide Y (NPY) in the mouse forced swim test. Peptides 26, 1394–1400 (2005).
Blier, P. & de Montigny, C. Serotonin and drug-induced therapeutic responses in major depression, obsessive-compulsive and panic disorders. Neuropsychopharmacology 21, 91S–98S (1999).
Descarries, L., Watkins, K., Garcia, S. & Beaudet, A. The serotonin neurons in nucleus raphe dorsalis of adult rat: a light and electron microscope radioautographic study. J. Comp. Neurol. 207, 239–254 (1982).
Gross, C. et al. Serotonin1A receptor acts during development to establish normal anxiety-like behaviour in the adult. Nature 416, 396–400 (2002).
Xu, X., Pan, Y. & Wang, X. mRNA expression of the lipid and mechano-gated 2P domain K+ channels. J. Neurogenet. 16, 263–269 (2002).
Caccia, S., Cappi, M., Fracasso, C. & Garattini, S. Influence of dose and route of administration on the kinetics of fluoxetine and its metabolite norfluoxetine in the rat. Psychopharmacology (Berl.) 100, 509–514 (1990).
Gingrich, J.A. & Hen, R. Dissecting the role of the serotonin system in neuropsychiatric disorders using knockout mice. Psychopharmacology (Berl.) 155, 1–10 (2001).
Gobbi, G., Murphy, D.L., Lesch, K. & Blier, P. Modifications of the serotonergic system in mice lacking serotonin transporters: an in vivo electrophysiological study. J. Pharmacol. Exp. Ther. 296, 987–995 (2001).
Kandel, E.R. & Spencer, W.A. Electrophysiology of hippocampal neurons. II. After-potentials and repetitive firing. J. Neurophysiol. 24, 243–259 (1961).
Acknowledgements
This work was supported by the CNRS and the Institut Paul Hamel. G.L. received a Canadian Institute for Health Research/Wyeth Fellowship, X.-D.P. received a Québec Merit Fellowship, G.D. was a National Researcher of the Fonds de la Recherche en Santé du Québec and S.T. received a Deutsche Forschungsgemeinschaft Fellowship. We are very grateful to Y. Jacomet for the MRS analysis of brain fluoxetine concentrations, to F. Lesage (CNRS Unité Mixte de Recherche, UMR 6097) for the TREK-1 antibody and to J. Costentin for discussions, to C. Gandin and M. Jodar for their technical help and to Y. Benhamou and L. Martin for secretarial assistance.
Author information
Authors and Affiliations
Contributions
C.H. behavioral experiments, localization studies, data analysis and writing. G.L. in vivo electrophysiology and data analysis. N.G. production of knockout mice. M.E.Y. behavioral experiments. S.T. in vitro electrophysiology of antidepressants. X.-D.P. in vivo electrophysiology. F.N. behavioral experiments. N.B. localization studies and data analysis. C.W. localization studies and statistics. M.B. localization studies and biochemical experiments. G.G. in vivo electrophysiology. J.-M.V. behavioral studies. G.D. in vivo electrophysiological experiments, data analysis and writing. M.L. overall organization of the research work between the different authors and different teams, and writing.
Note: Supplementary information is available on the Nature Neuroscience website.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
TREK-1 immunolocalization throughout the network of limbic, striatal and prefrontal cortical neuronal circuits. (PDF 1149 kb)
Supplementary Fig. 2
TRAAK expression in 5-HT neurons of the dorsal raphe nucleus. (PDF 1975 kb)
Supplementary Fig. 3
5-HT selective reuptake inhibitors (SSRIs) inhibit human TREK-1 but not TRAAK currents. (PDF 280 kb)
Supplementary Fig. 4
Mean whole-brain fluoxetine concentrations after acute and chronic fluoxetine treatment. (PDF 28 kb)
Rights and permissions
About this article
Cite this article
Heurteaux, C., Lucas, G., Guy, N. et al. Deletion of the background potassium channel TREK-1 results in a depression-resistant phenotype. Nat Neurosci 9, 1134–1141 (2006). https://doi.org/10.1038/nn1749
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1749
This article is cited by
-
Ion Channel Dysregulation Following Intracerebral Hemorrhage
Neuroscience Bulletin (2023)
-
Blocking Two-Pore Domain Potassium Channel TREK-1 Inhibits the Activation of A1-Like Reactive Astrocyte Through the NF-κB Signaling Pathway in a Rat Model of Major Depressive Disorder
Neurochemical Research (2023)
-
Microglia as a Game Changer in Epilepsy Comorbid Depression
Molecular Neurobiology (2023)
-
The Two-Pore Domain Potassium Channel TREK-1 Promotes Blood–Brain Barrier Breakdown and Exacerbates Neuronal Death After Focal Cerebral Ischemia in Mice
Molecular Neurobiology (2022)
-
Antidepressant activity of pharmacological and genetic deactivation of the small-conductance calcium-activated potassium channel subtype-3
Psychopharmacology (2022)