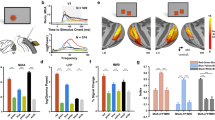
Abstract
When two isoluminant colors alternate at frequencies of 25 Hz or higher, observers perceive only one fused color. Chromatic flicker beyond the fusion frequency induces flicker adaptation in human observers and stimulates monkey V1 neurons. Here we use functional magnetic resonance imaging (fMRI) to show that many human visual cortical areas, with the exception of VO, can distinguish between fused chromatic flicker and its matched nonflickering control. This result supports the existence of significant intracortical temporal filtering of high-frequency chromatic information. The result also suggests that a considerable difference in cortical activation in many visual cortical areas does not necessarily lead to different conscious experiences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
De Lange, H. Research into the dynamic nature of the human fovea-cortex systems with intermittent and modulated light. II. Phase shift in brightness and delay in color perception. J. Opt. Soc. Am. 48, 784–789 (1958).
Brown, J.L. in Vision and Visual Perception (ed. Graham, C. H.) (John Wiley and Sons, Inc., New York, 1965).
Matin, L. Critical duration, the differential luminance threshold, critical flicker frequency, and visual adaptation: a theoretical treatment. J. Opt. Soc. Am. 58, 404–415 (1968).
Kelly, D.H. Theory of flicker and transient responses. I. Uniform fields. J. Opt. Soc. Am. 61, 537–546 (1971).
Gur, M. & Snodderly, D.M. A dissociation between brain activity and perception: chromatically opponent cortical neurons signal chromatic flicker that is not perceived. Vision Res. 37, 377–382 (1997).
Shady, S., MacLeod, D.I. & Fisher, H.S. Adaptation from invisible flicker. Proc. Natl. Acad. Sci. USA 101, 5170–5173 (2004).
Vul, E. & Macleod, D.I. Contingent aftereffects distinguish conscious and preconscious color processing. Nat. Neurosci. 9, 873–874 (2006).
Williams, P.E., Mechler, F., Gordon, J., Shapley, R. & Hawken, M.J. Entrainment to video displays in primary visual cortex of macaque and humans. J. Neurosci. 24, 8278–8288 (2004).
Engel, S., Zhang, X. & Wandell, B. Color tuning in human visual cortex measured with functional magnetic resonance imaging. Nature 388, 68–71 (1997).
Liu, J. & Wandell, B.A. Specializations for chromatic and temporal signals in human visual cortex. J. Neurosci. 25, 3459–3468 (2005).
Carmel, D., Lavie, N. & Rees, G. Conscious awareness of flicker in humans involves frontal and parietal cortex. Curr. Biol. 16, 907–911 (2006).
Hadjikhani, N., Liu, A.K., Dale, A.M., Cavanagh, P. & Tootell, R.B. Retinotopy and color sensitivity in human visual cortical area V8. Nat. Neurosci. 1, 235–241 (1998).
Engel, S.A., Glover, G.H. & Wandell, B.A. Retinotopic organization in human visual cortex and the spatial precision of functional MRI. Cereb. Cortex 7, 181–192 (1997).
Brewer, A.A., Liu, J., Wade, A.R. & Wandell, B.A. Visual field maps and stimulus selectivity in human ventral occipital cortex. Nat. Neurosci. 8, 1102–1109 (2005).
McKeefry, D.J. & Zeki, S. The position and topography of the human color centre as revealed by functional magnetic resonance imaging. Brain 120, 2229–2242 (1997).
Logothetis, N.K., Pauls, J., Augath, M., Trinath, T. & Oeltermann, A. Neurophysiological investigation of the basis of the fMRI signal. Nature 412, 150–157 (2001).
Pearlman, A.L., Birch, J. & Meadows, J.C. Cerebral color blindness: an acquired defect in hue discrimination. Ann. Neurol. 5, 253–261 (1979).
Damasio, A., Yamada, T., Damasio, H., Corbett, J. & McKee, J. Central achromatopsia: behavioral, anatomic, and physiologic aspects. Neurology 30, 1064–1071 (1980).
Zeki, S. A century of cerebral achromatopsia. Brain 113, 1721–1777 (1990).
Howard, R.J. et al. The functional anatomy of imagining and perceiving color. Neuroreport 9, 1019–1023 (1998).
Steven, M.S., Hansen, P.C. & Blakemore, C. Activation of color-selective areas of the visual cortex in a blind synesthete. Cortex 42, 304–308 (2006).
Efron, B. & Tibshirani, R. An Introduction to the Bootstrap (Chapman & Hall, New York, 1993).
Davison, A.C. & Hinkley, D.V. Bootstrap Methods and their Application (Cambridge University Press, Cambridge; New York, 1997).
Crick, F. & Koch, C. Are we aware of neural activity in primary visual cortex? Nature 375, 121–123 (1995).
He, S., Cavanagh, P. & Intriligator, J. Attentional resolution and the locus of visual awareness. Nature 383, 334–337 (1996).
He, S. & MacLeod, D.I. Orientation-selective adaptation and tilt after-effect from invisible patterns. Nature 411, 473–476 (2001).
Blake, R. & He, S. in Fitting the Mind to the World: Adaptation and After-effects in High-level Vision (eds. Clifford, C.W.G. & Rhodes, G.) (Oxford University Press, New York, 2005).
Lamme, V.A. & Roelfsema, P.R. The distinct modes of vision offered by feedforward and recurrent processing. Trends Neurosci. 23, 571–579 (2000).
Lamme, V.A. Blindsight: the role of feedforward and feedback corticocortical connections. Acta Psychol. (Amst.) 107, 209–228 (2001).
Pascual-Leone, A. & Walsh, V. Fast backprojections from the motion to the primary visual area necessary for visual awareness. Science 292, 510–512 (2001).
Ro, T., Breitmeyer, B., Burton, P., Singhal, N.S. & Lane, D. Feedback contributions to visual awareness in human occipital cortex. Curr. Biol. 13, 1038–1041 (2003).
Juan, C.H., Campana, G. & Walsh, V. Cortical interactions in vision and awareness: hierarchies in reverse. Prog. Brain Res. 144, 117–130 (2004).
Acknowledgements
We thank F. Fang for technical assistance, D. MacLeod for comments on an early version of the manuscript, and P. Costello and R. Shannon for their help with the manuscript. This research was supported by the James S. McDonnell foundation, the US National Institutes of Health (EY-015261-01), the 973 program (2005CB522800) and the Knowledge Innovation Project of the Chinese Academy of Sciences. The 3T scanner at the University of Minnesota is supported by Biotechnology Research Resource (BTRR) grant P41 008079 and by the Mental Illness and Neuroscience Discovery (MIND) Institute.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Fig. 1
BOLD response as a function of flicker temporal frequency and contrast. (PDF 87 kb)
Supplementary Table 1
Talairach coordinates of ROIs (hV4 and VO). (PDF 59 kb)
Supplementary Table 2
Behavioral results in the 4AFC stimulus discrimination task. (PDF 61 kb)
Rights and permissions
About this article
Cite this article
Jiang, Y., Zhou, K. & He, S. Human visual cortex responds to invisible chromatic flicker. Nat Neurosci 10, 657–662 (2007). https://doi.org/10.1038/nn1879
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1879
This article is cited by
-
Research on stereoscopic visual masking in binocular combination and unconscious rivalry
Multimedia Tools and Applications (2023)
-
Informative neural representations of unseen contents during higher-order processing in human brains and deep artificial networks
Nature Human Behaviour (2022)
-
LIPO: Indoor position and orientation estimation via superposed reflected light
Personal and Ubiquitous Computing (2022)
-
Does consciousness overflow cognitive access? Novel insights from the new phenomenon of attribute amnesia
Science China Life Sciences (2021)
-
Neural correlates of consciousness: progress and problems
Nature Reviews Neuroscience (2016)