Abstract
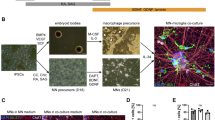
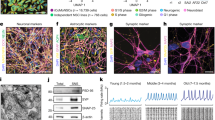
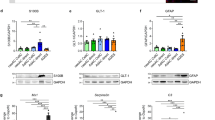
Here we report an in vitro model system for studying the molecular and cellular mechanisms that underlie the neurodegenerative disease amyotrophic lateral sclerosis (ALS). Embryonic stem cells (ESCs) derived from mice carrying normal or mutant transgenic alleles of the human SOD1 gene were used to generate motor neurons by in vitro differentiation. These motor neurons could be maintained in long-term coculture either with additional cells that arose during differentiation or with primary glial cells. Motor neurons carrying either the nonpathological human SOD1 transgene or the mutant SOD1G93A allele showed neurodegenerative properties when cocultured with SOD1G93A glial cells. Thus, our studies demonstrate that glial cells carrying a human SOD1G93A mutation have a direct, non–cell autonomous effect on motor neuron survival. More generally, our results show that ESC-based models of disease provide a powerful tool for studying the mechanisms of neural degeneration. These phenotypes displayed in culture could provide cell-based assays for the identification of new ALS drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Boillee, S., Vande Velde, C. & Cleveland, D.W. ALS: a disease of motor neurons and their nonneuronal neighbors. Neuron 52, 39–59 (2006).
Brown, R.H., Jr . Amyotrophic lateral sclerosis. Insights from genetics. Arch. Neurol. 54, 1246–1250 (1997).
Cole, N. & Siddique, T. Genetic disorders of motor neurons. Semin. Neurol. 19, 407–418 (1999).
Rosen, D.R. et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362, 59–62 (1993).
Gurney, M.E. et al. Motor neuron degeneration in mice that express a human Cu/Zn superoxide dismutase mutation. Science 264, 1772–1775 (1994).
Nagai, M. et al. Rats expressing human cytosolic copper-zinc superoxide dismutase transgenes with amyotrophic lateral sclerosis: associated mutations develop motor neuron disease. J. Neurosci. 21, 9246–9254 (2001).
Bruijn, L.I. et al. ALS-linked SOD1 mutant G85R mediates damage to astrocytes and promotes rapidly progressive disease with SOD1-containing inclusions. Neuron 18, 327–338 (1997).
Wong, P.C. et al. An adverse property of a familial ALS-linked SOD1 mutation causes motor neuron disease characterized by vacuolar degeneration of mitochondria. Neuron 14, 1105–1116 (1995).
Bruijn, L.I., Miller, T.M. & Cleveland, D.W. Unraveling the mechanisms involved in motor neuron degeneration in ALS. Annu. Rev. Neurosci. 27, 723–749 (2004).
Clement, A.M. et al. Wild-type non-neuronal cells extend survival of SOD1 mutant motor neurons in ALS mice. Science 302, 113–117 (2003).
Boillee, S. et al. Onset and progression in inherited ALS determined by motor neurons and microglia. Science 312, 1389–1392 (2006).
Wichterle, H., Lieberam, I., Porter, J.A. & Jessell, T.M. Directed differentiation of embryonic stem cells into motor neurons. Cell 110, 385–397 (2002).
Arber, S. et al. Requirement for the homeobox gene Hb9 in the consolidation of motor neuron identity. Neuron 23, 659–674 (1999).
Thaler, J. et al. Active suppression of interneuron programs within developing motor neurons revealed by analysis of homeodomain factor HB9. Neuron 23, 675–687 (1999).
Ince, P.G., Tomkins, J., Slade, J.Y., Thatcher, N.M. & Shaw, P.J. Amyotrophic lateral sclerosis associated with genetic abnormalities in the gene encoding Cu/Zn superoxide dismutase: molecular pathology of five new cases, and comparison with previous reports and 73 sporadic cases of ALS. J. Neuropathol. Exp. Neurol. 57, 895–904 (1998).
Wang, J. et al. Copper-binding–site null SOD1 causes ALS in transgenic mice: aggregates of non-native SOD1 delineate a common feature. Hum. Mol. Genet. 12, 2753–2764 (2003).
Watanabe, M. et al. Histological evidence of protein aggregation in mutant SOD1 transgenic mice and in amyotrophic lateral sclerosis neural tissues. Neurobiol. Dis. 8, 933–941 (2001).
Pasinelli, P., Houseweart, M.K., Brown, R.H., Jr . & Cleveland, D.W. Caspase-1 and 3 are sequentially activated in motor neuron death in Cu/Zn superoxide dismutase–mediated familial amyotrophic lateral sclerosis. Proc. Natl. Acad. Sci. USA 97, 13901–13906 (2000).
Raoul, C. et al. Motoneuron death triggered by a specific pathway downstream of Fas potentiation by ALS-linked SOD1 mutations. Neuron 35, 1067–1083 (2002).
Beers, D.R. et al. Wild-type microglia extend survival in PU.1 knockout mice with familial amyotrophic lateral sclerosis. Proc. Natl. Acad. Sci. USA 103, 16021–16026 (2006).
Zhou, Q. & Anderson, D.J. The bHLH transcription factors OLIG2 and OLIG1 couple neuronal and glial subtype specification. Cell 109, 61–73 (2002).
Bignami, A. & Dahl, D. Astrocyte-specific protein and neuroglial differentiation. An immunofluorescence study with antibodies to the glial fibrillary acidic protein. J. Comp. Neurol. 153, 27–38 (1974).
Banker, G. & Goslin, K. Culturing Nerve Cells 2nd ed. (MIT Press, Cambridge, Massachusetts, 1998).
Ullian, E.M., Harris, B.T., Wu, A., Chan, J.R. & Barres, B.A. Schwann cells and astrocytes induce synapse formation by spinal motor neurons in culture. Mol. Cell. Neurosci. 25, 241–251 (2004).
Allen, N.J. & Barres, B.A. Signaling between glia and neurons: focus on synaptic plasticity. Curr. Opin. Neurobiol. 15, 542–548 (2005).
Ullian, E.M., Christopherson, K.S. & Barres, B.A. Role for glia in synaptogenesis. Glia 47, 209–216 (2004).
Evans, M.J. & Kaufman, M.H. Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 (1981).
Martin, G.R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl. Acad. Sci. USA 78, 7634–7638 1981).
Lerou, P.H. & Daley, G.Q. Therapeutic potential of embryonic stem cells. Blood Rev. 19, 321–331 (2005).
Ben-Nun, I.F. & Benvenisty, N. Human embryonic stem cells as a cellular model for human disorders. Mol. Cell. Endocrinol. 252, 154–159 (2006).
Nagai, M. et al. Astrocytes expressing ALS-linked mutated SOD1 release factors selectively toxic to motor neurons. Nat. Neurosci. advance online publication, 15 April 2007 (doi:10.1038/nn1876).
Hogan, B., Beddington, R., Costantini, F. & Lacey, E. Manipulating the Mouse Embryo: A Laboratory Manual 2nd edn. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1994).
Acknowledgements
We thank J.W. Lichtman and J.R. Sanes for helpful discussions and use of confocal microscopy facilities at The Center for Brain Science, Harvard University. We are grateful to H. Akutsu for assistance in the derivation of mouse ESC lines, G. Birkhoff for preparing figures and B. Tilton for assistance with flow cytometry. We thank S. Przedborski and other members of the Columbia University research groups for communicating their results to us prior to publication, and for their criticals comments in our manuscripts. We thank Project ALS for supporting contact and cooperation between the research groups at Columbia and Harvard and for donating transgenic SOD1G93A mice. We thank T.M. Jessell and H. Wichterle for providing Hb9 antisera. This work was supported by the Stowers Medical Institute and Harvard Stem Cell Institute and US National Institutes of Health R01 HD046732-01A1 to K. Eggan and by the ALS Association to T. Maniatis. M.A. Carrasco is a Milton Safenowitz Post-Doctoral Fellow of the ALS Association and K. Eggan is a MacArthur Fellow of the John D. and Catherine T. MacArthur Foundation.
Author information
Authors and Affiliations
Contributions
F.P.D. derived the SOD1G93A, SOD1 and HB9::GFP mouse stem cell lines and performed both the differentiation of ESCs into motor neurons and the initial characterization of these neurons, including immunocytochemistry and initial neuronal counting experiments. M.A.C. performed morphometric analysis of SOD1 inclusions, ES-derived motor neuron coculture experiments with glia cells, and the associated characterization, survival assays and immunocytochemistry. M.S. contributed to the isolation of primary glia and glial genotyping. All authors contributed to experimental design and the writing of this paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Percentage of differentiating EBs cells that express GFP. (PDF 9308 kb)
Supplementary Fig. 2
Expression of neuronal marker Isl1 in cells derived from SOD1G93A mouse ES cell lines. (PDF 19109 kb)
Supplementary Fig. 3
Quantitative and qualitative analysis of SOD1 protein inclusions in ESC-derived motor neurons. (PDF 14249 kb)
Supplementary Fig. 4
Activation of caspase-3 and cytoplasmic localization of cytochrome C in SOD1G93A motor neurons 14 d after EBs dissociation. (PDF 14455 kb)
Supplementary Fig. 5
Characterization of primary glial monolayers derived from SOD1 and SOD1G93A mice. (PDF 14632 kb)
Supplementary Fig. 6
Table describes primary antibodies used. (PDF 16 kb)
Rights and permissions
About this article
Cite this article
Di Giorgio, F., Carrasco, M., Siao, M. et al. Non–cell autonomous effect of glia on motor neurons in an embryonic stem cell–based ALS model. Nat Neurosci 10, 608–614 (2007). https://doi.org/10.1038/nn1885
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1885
This article is cited by
-
Functional roles of reactive astrocytes in neuroinflammation and neurodegeneration
Nature Reviews Neurology (2023)
-
In Vitro Models of Amyotrophic Lateral Sclerosis
Cellular and Molecular Neurobiology (2023)
-
Organ-On-A-Chip Technology: An In-depth Review of Recent Advancements and Future of Whole Body-on-chip
BioChip Journal (2023)
-
Remodeling of astrocyte secretome in amyotrophic lateral sclerosis: uncovering novel targets to combat astrocyte-mediated toxicity
Translational Neurodegeneration (2022)
-
Serum biomarkers of neuroinflammation and blood-brain barrier leakage in amyotrophic lateral sclerosis
BMC Neurology (2022)