Abstract
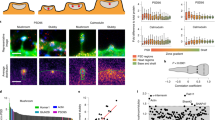
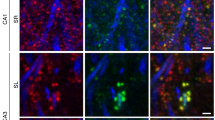
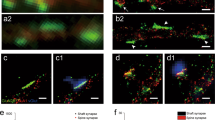
The dynamics of postsynaptic density (PSD) formation and remodeling were investigated in live developing hippocampal tissue slices. Time lapse imaging of transfected neurons expressing GFP-tagged PSD95, a prominent PSD protein, revealed that up to 40% of PSDs in developing dendrites are structurally dynamic; they rapidly (<15 min) appear or disappear, but also grow, shrink and move within shafts and spines. New spines containing PSDs were formed by conversion of dynamic filopodia-like spine precursors in which PSDs appeared de novo, or by direct extension of spines or spine precursors carrying preformed PSDs from the shaft. PSDs are therefore highly dynamic structures that can undergo rapid structural alteration within dendrite shafts, spines and spine precursors, permitting rapid formation and remodeling of synaptic connections in developing CNS tissues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Harris, K. M. & Kater, S. B. Dendritic spines: cellular specializations imparting both stability and flexibility to synaptic function. Annu. Rev. Neurosci. 17, 341–371 (1994).
Sorra, K. E. & Harris, K. M. Overview on the structure, composition, function, development, and plasticity of hippocampal dendritic spines. Hippocampus 10, 501–511 (2000).
Kennedy, M. B. Signal-processing machines at the postsynaptic density. Science 290, 750–754 (2000).
Cho, K. O., Hunt, C. A. & Kennedy, M. B. The rat brain postsynaptic density fraction contains a homolog of the Drosophila discs-large tumor suppressor protein. Neuron 9, 929–942 (1992).
Kistner, U. et al. SAP90, a rat presynaptic protein related to the product of the Drosophila tumor suppressor gene dlg-A. J. Biol. Chem. 268, 4580–4583 (1993).
Friedman, H. V., Bresler, T., Garner, C. C. & Ziv, N. E. Assembly of new individual excitatory synapses: time course and temporal order of synaptic molecule recruitment. Neuron 27, 57–69 (2000).
Husseini, A. E.-D., Schnell, E., Chetkovich, D. M., Nicoll, R. A. & Bredt, D. S. PSD-95 involvement in maturation of excitatory synapses. Science 290, 1364–1368 (2000).
Okabe, S., Kim, H.-D., Miwa, A., Kuriu, T. & Okado, H. Continual remodeling of postsynaptic density and its regulation by synaptic activity. Nat. Neurosci. 2, 804–811 (1999).
Harris, K.M. Structure, development, and plasticity of dendritic spines. Curr. Opin. Neurobiol. 9, 343–348 (1999).
Morest, D. K. The differentiation of cerebral dendrites: a study of the post-migratory neuroblast in the medial nucleus of the trapezoid body. Z. Anat. Entwicklungsgesch. 128, 271–289 (1969).
Purpura, D. P. Dendritic differentiation in human cerebral cortex: normal and aberrant developmental patterns. Adv. Neurol. 12, 91–134 (1975).
Ziv, N. E. & Smith, S. J. Evidence for a role of dendritic filopodia in synaptogenesis and spine formation. Neuron 17, 91–102 (1996).
Dailey, M. E. & Smith, S. J. The dynamics of dendritic structure in developing hippocampal slices. J. Neurosci. 16, 2983–2994 (1996).
Dunaevsky, A., Tashiro, A., Majewska, A., Mason, C. & Yuste, R. Developmental regulation of spine motility in the mammalian central nervous system. Proc. Natl. Acad. Sci. USA 96, 13438–13443 (1999).
Parnass, Z., Tashiro, A. & Yuste, R. Analysis of spine morphological plasticity in developing hippocampal pyramidal neurons. Hippocampus 10, 561–568 (2000).
Lendvai, B., Stern, E. A., Chen, B. & Svoboda, K. Experience-dependent plasticity of dendritic spines in the developing rat barrel cortex in vivo. Nature 404, 876–881 (2000).
Maletic-Savatic, M., Malinow, R. & Svoboda, K. Rapid dendritic morphogenesis in CA1 hippocampal dendrites induced by synaptic activity. Science 283, 1923–1927 (1999).
Jontes, J. D. & Smith, S. J. Filopodia, spines, and the generation of synaptic diversity. Neuron 27, 11–14 (2000).
Wong, W. T. & Wong, R. O. L. Rapid dendritic movements during synapse formation and rearrangement. Curr. Opin. Neurobiol. 10, 118–124 (2000).
Saito, Y. et al. Developing corticorubral axons of the cat form synapses on filopodial dendritic protrusions. Neurosci. Lett. 147, 81–84 (1992).
Fiala, J. C., Feinberg, M., Popov, V. & Harris, K. M. Synaptogenesis via dendritic filopodia in developing hippocampal area CA1. J. Neurosci. 18, 8900–8911 (1998).
Gähwiler, B. H., Capogna, M., Debanne, D., McKinney, R. A. & Thompson, S. M. Organotypic slice cultures: a technique has come of age. Trends Neurosci. 20, 471–477 (1997).
Frotscher, M., Zafirov, S. & Heimrich, B. Development of identified neuronal types and of specific synaptic connections in slice cultures of rat hippocampus. Prog. Neurobiol. 45, 143–164 (1995).
Arnold, D. B. & Clapham, D. E. Molecular determinants for subcellular localization of PSD95 with an interacting K+ channel. Neuron 23, 149–157 (1999).
DeCamilli, P., Cameron, R. & Greengard, P. Synapsin I (protein I), a nerve terminal specific phosphoprotein: I. Its general distribution in synapses of the central and peripheral nervous system demonstrated by immunofluorescence in frozen and plastic sections. J. Cell. Biol. 96, 1337–1354 (1983).
Boyer, C., Schikorski, T. & Stevens, C. F. Comparison of hippocampal dendritic spines in culture and in brain. J. Neurosci. 18, 5294–5300 (1998).
Rao, A., Kim, E., Sheng, M. & Craig, A. M. Heterogeneity in the molecular composition of excitatory postsynaptic sites during development of hippocampal neurons in culture. J. Neurosci. 18, 1217–1229 (1998).
Hinds, J. W. & Hinds, P. L. Synapse formation in the mouse olfactory bulb. II. Morphogenesis. J. Comp. Neurol. 169, 41–61 (1976).
Blue, M. E. & Parnavelas, J. G. The formation and maturation of synapses in the visual cortex of the rat. I. Qualitative analysis. J. Neurocytol. 12, 599–616 (1983).
Halpain, S. Actin and the agile spine: how and why do dendritic spines dance? Trends Neurosci. 23, 141–146 (2000).
Matus, A. Actin-based plasticity in dendritic spines. Science 290, 754–758 (2000).
Walikonis, R. S. et al. Identification of proteins in the postsynaptic density fraction by mass spectrometry. J. Neurosci. 20, 4069–4080 (2000).
Naisbitt, S. et al. Interaction of the postsynaptic density-95/Guanylate kinase domain-associated protein complex with a light chain of myosin-V and dynein. J. Neurosci. 20, 4524–4534 (2000).
Passafaro, M., Sala, C., Niethammer, M. & Sheng, M. Microtubule binding by CRIPT and its potential role in the synaptic clustering of PSD-95. Nat. Neurosci. 2, 1063–1069 (1999).
Rao, A. & Craig, A. M. Signaling between the actin cytoskeleton and the postsynaptic density of dendritic spines. Hippocampus 10, 527–541 (2000).
Smith, S. J. & Jahr, C. E. in The Nerve Growth Cone (eds. Letourneau, P. C., Kater, S. B. & Macagno, E. R.) 19–26 (Raven, New York, 1992).
Korkotian, E. & Segal, M. Bidirectional regulation of dendritic spine dimensions by glutamate receptors. Neuroreport 10, 2875–2877 (1999).
Engert, F. & Bonhoeffer, T. Dendritic spine changes associated with hippocampal long-term synaptic plasticity. Nature 399, 66–70 (1999).
Krucker, T., Siggins, G. R. & Halpain, S. Dynamic actin filaments are required for stable long-term potentiation (LTP) in area CA1 of the hippocampus. Proc. Natl. Acad. Sci. USA 97, 6856–6861 (2000).
Luscher, C., Nicoll, R. A., Malenka, R. C. & Muller, D. Synaptic plasticity and dynamic modulation of the postsynaptic membrane. Nat. Neurosci. 3, 545–550 (2000).
Ahmari, S., Buchanan, J. & Smith, S. J. Assembly of presynaptic active zones from cytoplasmic transport packets. Nat. Neurosci. 3, 445–451 (2000).
Cotman, C., Taylor, D. & Lynch, G. Ultrastructural changes in synapses in the dentate gyrus of the rat during development. Brain Res. 63, 205–213 (1973).
Pokorny, J. & Yamamoto, T. Postnatal ontogenesis of hippocampal CA1 area in rats. II. Development of ultrastructure in stratum lacunosum and moleculare. Brain Res. Bull. 7, 121–130 (1981).
Steward, O. et al. Protein synthesis and processing in cytoplasmic microdomains beneath postsynaptic sites on CNS neurons. A mechanism for establishing and maintaining a mosaic postsynaptic receptive surface. Mol. Neurobiol. 2, 227–261 (1988).
Harris, K. M., Jensen, F. E. & Tsao, B. Y. in The Hippocampus: New Vistas (eds. Chan-Palay, V. & Kohler, C.) 33–52 (Alan Liss, New York, 1989).
Wong, W. T. & Wong, R. O. L. Changing specificity of neurotransmitter regulation of rapid dendritic remodeling during synaptogenesis. Nat. Neurosci. 4, 351–352 (2001).
Stoppini, L., Buchs, P.-A. & Muller, D. A simple method for organotypic cultures of nervous tissue. J. Neurosci. Methods 37, 173–182 (1991).
Lo, D. C., McAllister, A. K. & Katz, L. C. Neuronal transfection in brain slices using particle-mediated gene transfer. Neuron 13, 1263–1268 (1994).
Dailey, M. E. in Imaging Neurons: A Laboratory Manual (eds. Yuste, R., Lanni, F. & Konnerth, A.) 10.1–10.7 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1999).
Acknowledgements
We thank D. Bredt (UCSF) for the PSD95-GFP construct, S. Lee for help with data analysis, J. Rathner for reading the manuscript, and L. Katz, D. Lo and P. Bridgman for discussions on use of the gene gun. This work was supported by grants from the National Institutes of Health (NS37159 to M.E.D.; DC02961 to S.H.G.), Whitehall Foundation (98-6 to M.E.D.), and University of Iowa Biosciences Initiative (M.E.D. and S.H.G.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Fig. 1.
Quantitative analysis of a neuron co-transfected with PSD95-GFP and DsRed. These analyses demonstrate the minimal effect of z-movement on the size and intensity of PSD95-GFP hotspots during a typical time lapse imaging session, and also show that free DsRed is not significantly 'clumpy.' GFP-PSD95 and DsRed were expressed in single hippocampal pyramidal neurons. Images of GFP-PSD95 and DsRed were captured simultaneously in two different fluorescent channels using a single excitation light source (Argon laser). Twenty optical sections were collected at each time point. The z-step size was 0.9 mm. (a) Selected confocal images of GFP-PSD95 for 8 time points spanning nearly 1 h. Only 10 of the original 20 focal planes are shown. The columns represent focal planes 6 through 15 (f6-f15) through a portion of a dendrite containing PSD95-GFP hotspots. A hotspot within a spine (arrow in f13 at t = 0') is most clearly in focus in different image planes at different time points, indicating z-movement relative to the z-stack of images. In some instances, large focus jumps were intentionally introduced by manually turning the focus knob a random amount between time points. The arrows indicate the optimal (center) image of the spine hotspot at each time point. (b) Analysis of fluorescent intensities in corresponding GFP-PSD95 and DsRed projection images. The projection images represent maximum brightness projections of all captured focal planes (f1-f20). Plots of fluorescent intensity along a line (green and red lines overlaying the projection images) are shown for GFP-PSD95 and DsRed. To account for differences in the level of protein expression and imaging parameters, the PSD95-GFP and DsRed intensity plots were normalized with respect to each other by matching the peak fluorescent intensity values in the shaft region as well as the low intensity values of the non-fluorescent background (as in Fig. 1d). 'Difference' images were computed by mathematically subtracting the DsRed fluorescence signal from the GFP-PSD95 signal. The shaft GFP fluorescence is negated by the DsRed fluorescence (arrowhead), whereas the two PSD95 hotspots (two arrows) are significantly brighter and thus readily apparent in the difference plots at each time point. (JPG 104 kb)
41593_2001_BFnn717_MOESM2_ESM.jpg
(c) Peak intensity values at each time point for the shaft and two PSD95 hotspots shown in panel (b). Note that intensities of the shaft and hotspot 1 show little variation over the time period observed, even though the sample drifted wildly within the z-stack of images. Hotspot 2 shows more variation in the fluorescent intensity of GFP-PSD95, but the 'difference' intensity plot that was normalized against DsRed (and thus negates any effect of focus change) closely mimics changes in the raw GFP-PSD95 intensity. These data indicate that z-movement cannot account for the significant changes in hotspot features we present elsewhere in the manuscript. (d) Evidence that the pattern of DsRed expression is not 'clumpy,' and thus DsRed serves as a good volume indicator. Line plots of GFP-PSD95 (green) and DsRed (red) fluorescence intensity in a region of the dendrite shaft devoid of GFP-PSD95 hotspots are shown for the projection images (left) and for a single focal plane through the center of the shaft (right). Note that standard deviations of the pixel intensity values are not significantly different for GFP and DsRed, indicating that the DsRed is no more 'clumpy' than GFP-PSD95 in regions devoid of GFP-PSD95 hotspots. (JPG 38 kb)
Rights and permissions
About this article
Cite this article
Marrs, G., Green, S. & Dailey, M. Rapid formation and remodeling of postsynaptic densities in developing dendrites. Nat Neurosci 4, 1006–1013 (2001). https://doi.org/10.1038/nn717
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn717
This article is cited by
-
Regulation of local alternating electric fields on synaptic plasticity in brain tissue
Biomedical Engineering Letters (2023)
-
BDNF signaling during the lifetime of dendritic spines
Cell and Tissue Research (2020)
-
Structural and molecular correlates of cognitive aging in the rat
Scientific Reports (2019)
-
The Exocyst Component Exo70 Modulates Dendrite Arbor Formation, Synapse Density, and Spine Maturation in Primary Hippocampal Neurons
Molecular Neurobiology (2019)
-
Quantifying barcodes of dendritic spines using entropy-based metrics
Scientific Reports (2015)