Abstract
The ability to discern temporally pertinent environmental events is essential for the generation of adaptive behavior in conventional tasks, and our overall survival. Cannabinoids are thought to disrupt temporally controlled behaviors by interfering with dedicated brain timing networks. Cannabinoids also increase dopamine release within the mesolimbic system, a neural pathway generally implicated in timing behavior. Timing can be assessed using fixed-interval (FI) schedules, which reinforce behavior on the basis of time. To date, it remains unknown how cannabinoids modulate dopamine release when responding under FI conditions, and for that matter, how subsecond dopamine release is related to time in these tasks. In the present study, we hypothesized that cannabinoids would accelerate timing behavior in an FI task while concurrently augmenting a temporally relevant pattern of dopamine release. To assess this possibility, we measured subsecond dopamine concentrations in the nucleus accumbens while mice responded for food under the influence of the cannabinoid agonist WIN 55 212-2 in an FI task. Our data reveal that accumbal dopamine concentrations decrease proportionally to interval duration—suggesting that dopamine encodes time in FI tasks. We further demonstrate that WIN 55 212-2 dose-dependently increases dopamine release and accelerates a temporal behavioral response pattern in a CB1 receptor-dependent manner—suggesting that cannabinoid receptor activation modifies timing behavior, in part, by augmenting time-engendered patterns of dopamine release. Additional investigation uncovered a specific role for endogenous cannabinoid tone in timing behavior, as elevations in 2-arachidonoylglycerol, but not anandamide, significantly accelerated the temporal response pattern in a manner akin to WIN 55 212-2.
Similar content being viewed by others
INTRODUCTION
Marijuana use is hallmarked by distorted perception of time (Moreau de Tours, 1845; Tart, 1970). Time is said to slow down: ‘the minutes become hours, the hours become days; soon any precise idea of duration escapes us’ (Moreau de Tours, 1845). Experimental studies suggest that cannabinoids slow down time by speeding up an internal clock (Hicks et al, 1984; Tinklenberg et al, 1976), so that an accelerated internal pace gives rise to the subjective impression that time is slowing (Atakan et al, 2012).
Cannabinoids act on the brain’s mesocorticolimbic dopamine system (Cheer et al, 2004), a neural pathway recently implicated in the temporal control of behavior (Narayanan et al, 2012). Such control can be assessed using fixed-interval (FI) schedules (Narayanan et al, 2012), which reinforce behavior on the basis of time (Dews, 1970). FI procedures produce a characteristic pattern of behavior known as a scallop. Immediately following reinforcement, responding slows before accelerating to a maximal terminal rate at the culmination of the interval (Dews, 1970). Cannabinoids, like other drugs that speed up the internal clock, produce an accelerated temporal response pattern (Cheng et al, 2007; Han and Robinson, 2001).
Although cannabinoids may speed up an internal clock, the underlying neural mechanisms remain unclear. In the present study, we hypothesized that WIN 55 212-2, a cannabinoid agonist with a high affinity for CB1 and CB2 receptors, would augment time-related patterns of dopamine as the temporal response pattern accelerates in an FI task. However, the pattern of accumbal dopamine release engendered by FI tasks is unknown. We therefore began by characterizing real-time dopamine release occurring in the nucleus accumbens of mice during FI responding, and found that dopamine release is inversely related to interval time while still phasically signaling the receipt of reward. Furthermore, we discovered that WIN 55 212-2 dose-dependently increased this pattern of dopamine release and accelerated the temporal response pattern. These results reveal that dopamine release accompanies the passage of time in FI tasks and that cannabinoid receptor activation amplifies release when interval timing is accelerated. To determine whether endogenous cannabinoid tone also modulates responding during FI tasks, we tested two distinct inhibitors of endocannabinoid enzymatic degradation. Elevations in the endocannabinoid 2-arachidonoylglycerol, but not anandamide, significantly accelerated the temporal response pattern in a manner akin to WIN 55 212-2.
MATERIALS AND METHODS
Experimental Subjects
All voltammetric recordings were preformed in freely moving male C57BL/6 mice. Animals were housed individually in a temperature-controlled environment on a 12 h light/dark schedule (0700–1900 h). Animals were maintained at 85% of their free-feeding body weight for the duration of experimentation. All experiments were conducted in the animals’ light cycle. The experimental protocol adhered to National Institutes of Health Animal Care guidelines and was performed in concordance with University of Maryland, Baltimore’s Institutional Animal Care and Use Committee (IACUC) protocols.
Electrode Fabrication
Chronically implantable electrodes were constructed as described previously (Clark et al, 2009). Briefly, a single carbon fiber was inserted into a 5 mm cut segment of fused silica while submerged in isopropyl alcohol. A seal was created by applying a two-part epoxy (Super Glue Corporation; TQs12 Epoxy) to one end of the silica tubing, while leaving the carbon fiber extending past the seal untouched. After allowing the epoxy seal time to dry, the carbon fiber was cut to a length of 150 μm. A silver connector was then attached to the carbon fiber on the opposite side of the silica tubing. Finally, a second coat of the two-part epoxy was applied to add insulation and structural support.
Surgery
Mice were anesthetized using isoflurane in O2 (4% induction and 1% maintenance) and implanted with a chronic voltammetry electrode aimed at the NAc core (+1.2 AP, +1.1 ML, −3.7 DV) and a contralateral Ag/AgCl reference electrode (0.5 mm diameter; Sigma-Aldrich). A stimulating electrode was lowered to the ipsilateral medial forebrain bundle for the duration of the surgery to allow for optimization of working electrode placement. The electrodes were attached to the voltammetric amplifier and lowered to the edge of the target region (−3.7 DV for the working and −3.8 DV for the stimulating). At this depth, a triangular voltammetric input waveform (initial ramp: −0.4 to 1.3 V, 400 V/s) was applied to the working electrode at 60 Hz for 30 min and then reduced to 10 Hz for the remainder of the surgery (Heien et al, 2003), while electrically evoked dopamine release was monitored. Electrical stimulation (60 biphasic pulses, 60 Hz, 300 μA, 2 ms per phase) was applied to the stimulating electrode (Plastics One) via a constant-current isolator (A-M Systems). Once electrically evoked dopamine release was detected in the nucleus accumbens core, a layer of dental cement was applied to secure the electrodes in place and allowed to dry. We focused on the core region of the NAc because of previous evidence suggesting that the core is critically involved in modulating cue-driven behavioral responses, whereas the shell is more involved in modulating context-driven behavioral responses (Bossert et al, 2007; Chaudhri et al, 2009; Ito and Hayen, 2011).
Dopamine Acquisition and Data Analysis
Dopamine was detected from fast-scan cyclic voltammograms collected at the carbon fiber electrode every 100 ms (initial waveform: −0.4 to 1.3 V, 400 V/s). Principal component regression was used to extract the dopamine component from the raw voltammetric data as described previously (Keithley et al, 2009). Aside from Figure 1, in which data were aligned to reinforcement, all data were aligned around the interval. Mean dopamine concentrations, local response rates, and time allotment data were extracted from each epoch and compared using a two-way repeated-measures ANOVA with Holm–Sidak post hoc analysis. Index of curvature comparisons were performed using a one-way repeated-measures ANOVA with Holm–Sidak post hoc analysis. Area under the curve analyses used to calculate index of curvature were performed using a SigmaPlot11 macro. Correlations are reported as a Pearson’s r. All statistics were performed using SigmaPlot11.
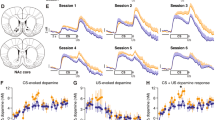
Changes in dopamine concentration aligned to reinforcement during the FI task. (a) The schematic shows all experimental events occurring in the FI task with a 10 s interval as they relate to the data presented below. The lever is always extended; reinforcement is contingent upon the first response that occurs after the 10 s interval. (b) A representative cumulative record from a mouse responding in the FI task (10 s interval). Vertical shifts in the cumulative record represent a response; horizontal shifts in the cumulative record represent time (s). (c) A representative dopamine concentration trace that corresponds to the above cumulative record. Inset shows the chemical signature for dopamine (ie, cyclic voltammogram) taken from the dopamine concentration trace at the vertical yellow line. (d) A representative pseudocolor plot shows the average pattern of dopamine transmission occurring in one FI session. The y axis represents the scan potential (EApp, V) applied to the electrode, the x axis represents time, and the z axis represents current. Inspection of the color plot allows for the identification of dopamine over time. Dopamine can be identified in the color plot by assessing for changes in current at the oxidation (+0.6 V) and reduction (−0.2 V) potentials for dopamine. The more prominent current shift at the oxidative potential for dopamine is highlighted by a horizontal yellow line. R represents the reinforced response. (e) Mean±SEM dopamine concentration trace from all fixed-interval (FI) sessions in which reinforcement was preceded by a 10 s interval plotted as a function of time. (f) The schematic shows all experimental events occurring in the FI task with a 30 s interval as they relate to the data presented below. (g and h) A representative cumulative record (top) and corresponding dopamine concentration trace (bottom) taken from a mouse responding in the FI task with a 30 s interval. (i) The representative pseudocolor plot shows mean dopamine (oxidative potential highlighted by horizontal yellow line) concentrations over time from a single FI session in which a 30 s interval preceded reinforcement availability. (j) Mean±SEM dopamine concentration trace from all FI sessions (30 s interval) plotted as a function of time.
Apparatus
All mice were tested in identical operant chambers (21.6 cm × 17.8 cm × 14 cm; Med Associates, St Albans, VT) housed within sound attenuating enclosures. Each chamber was equipped with two retractable levers (located 2 cm above the floor) and one LED stimulus light located above each lever (4.6 cm above the lever). An external food magazine was connected to a dispenser, centrally located between the two levers, that delivered chocolate flavored pellets (14 mg; Bio-Serv, Frenchtown, NJ). A houselight and a white-noise speaker (80 dB, masking noise background) were located on the opposite wall.
Behavioral Procedure
Separate groups of mice (n=5 for initial dopamine characterization; n=6 for WIN55 212-2, n=7 for JZL184, n=8 for URB597, n=8 for AM251) were initially trained under a fixed-ratio 1 schedule (10 s timeout) until stable responding was established (under 15% of variation in response rate across three consecutive sessions). Animals were then trained to respond under an FI schedule (10 s interval) that was adapted to accommodate our neurochemical study. Session onset was signaled to the animal by the illumination of the houselight. Thereafter, the first response made after 10 s had elapsed resulted in the delivery of a single food pellet. Responses made before this time were recorded but had no scheduled consequences. In addition, we added a 10 s waiting period after reinforcement in which the lever remained extended, but the house light dimmed. We added this period in an attempt to differentiate dopamine time-locked responses to reinforcement from those associated with the defined interval. Trial onset was signaled by the illumination of the houselight. Sessions ended after 1 h or 50 pellets, whichever came first. Dopamine concentrations were recorded after stable responding was established (∼4 sessions). Behavior was deemed stable if local rates of responding were greater in the final fifth of the interval for three consecutive sessions. Then, the interval was increased to 30 s; thus, the first response made after 30 s had elapsed, resulting in the delivery of a single food pellet. The onset of each trial, as well as the duration of the waiting period after food delivery, was similar to FI task with a 10 s interval.
Pharmacology
All injections, including vehicle, were administered intraperitoneally and assigned using a Latin square design with a minimum of 3 days between treatments. Pretreatment times were 30 min for all drugs injections except when AM251 injections preceded a primary drug treatment, in which case AM251 injections occurred 40 min before session onset. All drugs were prepared in a 1 : 1 : 18 vehicle consisting of emulphor, ethanol, and saline, respectively.
RESULTS
To investigate whether subsecond dopamine release events change as a function of time during periodically reinforced behavior, we measured fluctuations in their concentration in the nucleus accumbens core (n=5; Supplementary Figure S1), while mice responded for food in an FI task. Here, the lever was always extended into the experimental chamber, but a lever response was only reinforced after an experimenter-imposed interval. As we sought to assess separately dopamine concentrations during both food reinforcement and the experimenter-imposed interval, we added a 10 s postreinforcement period that was signaled to the animal by the dimming of a house light. Figure 1a and f show the experimental components of an individual trial with either a 10 or 30 s interval, respectively. We first measured release while mice responded for food in an FI task with a 10 s interval. Under these conditions, we observed a rapid increase in dopamine concentration time-locked to food reinforcement (R). This initial reinforcement-associated increase in dopamine release was then followed by a pause that preceded a second increase in dopamine concentration. This second concentration increase began before the initiation of lever pressing and then declined across time until the next reinforcement occurred. Figure 1b shows a cumulative record of behavioral responding and the corresponding representative pattern of dopamine release (Figure 1c; also see Supplementary Video S1). The scalloped behavioral response pattern persevered following the removal of all exteroceptive cues, suggesting that an interoceptive timing cue prompts the increase in response output throughout the interval (Supplementary Figure S2). To demonstrate the within-session consistency of this pattern of dopamine release, an average color plot from a representative mouse responding in one 30 min session is illustrated in Figure 1d. This pseudocolor plot illustrates the entire electrochemical profile of our representative data and thereby allows for the identification of subsecond dopamine concentrations from other electrochemical species. The y axis represents the voltage (V) applied to the electrode, the x axis time, and the z axis current. Dopamine can be identified in the color plot by assessing for changes in current at the oxidation (+0.6 V; dashed horizontal yellow line) and reduction (−0.2 V) potentials for dopamine. Figure 1e shows the mean change in dopamine concentration as a function of time for all mice.
We next investigated whether this pattern of dopamine release would reset and be sustained through a different interval by increasing the interval duration to 30 s. As predicted, an identical pattern occurred. Inspection of an individual trial (Figure 1h) shows dopamine: (a) increasing to reward delivery (R), (b) pausing, (c) increasing again before responding commenced, and (d) decreasing across the 30 s interval as response frequency increased (cf. Figure 1g and h). The observed patterns of dopamine concentration were consistent within (Figure 1e) and between (Figure 1f) sessions.
We next sought to characterize directly how dopamine and response output change throughout the interval of the FI task. To analyze neurochemical and behavioral changes over time, we centered data on the interval, which we then divided into five equivalent epochs (Figure 2). Figure 2a depicts the schematic of a single trial of the FI task with a 30 s interval; the divisions of the 30 s interval are color coded throughout the remaining panels of the figure (Figure 2b–d). As occurs in a traditional FI schedule (Dews, 1970), the present FI task engendered a response pattern that accelerated throughout the interval (ie, scallop; Figure 2b). The raster plot (top) illustrates that responding reliably increases in frequency during individual trials, while the perievent histogram (middle) shows the aggregate response pattern from a single FI session. The corresponding mean pattern of dopamine concentration is shown below the behavioral data. To compare quantitatively how behavior and dopamine change over the interval, we calculated mean local response rates (defined as the response rate occurring in an individual epoch; Figure 2c) and mean local dopamine concentrations (defined as the dopamine concentration occurring in an individual epoch; Figure 2d). Over the course of the interval, responding increased (F(4,19)=4.265; p<0.05), while dopamine concentrations decreased (F(4,1199)=43.419; p<0.05).
Changes in dopamine concentration centered on a 30 s interval during the fixed-interval (FI) task. The 30 s interval is divided into five color-coded epochs (6 s per epoch). (a) A schematic shows all experimental events as they relate to the data presented below. (b) Patterns of responding lawfully increase in the FI task. (Top) The raster plot shows responses (black ticks) preceding food reinforcement (red triangle) across the 30 s interval. All trials are shown in chronological order as they occurred in a representative experimental session. (Middle) The perievent histogram shows the summation of responding. (Bottom) The corresponding concentration trace shows the mean pattern of dopamine transmission occurring through the representative session. Inset shows the cyclic voltammogram for dopamine taken at the yellow vertical line. (c) The rate of responding increases over the 30 s interval in the FI task. Bars represent mean±SEM response rates occurring in each 6 min epoch. (d) Dopamine concentrations decrease over the 30 s interval. Bars represent mean±SEM dopamine concentration occurring in each 6 min epoch. (e) Dopamine concentrations are inversely related to local response rates in the FI task. Mean dopamine concentrations and local response rates were derived for each of five equal epochs (2 s per epoch with 10 s interval; 6 s per epoch with 30 s interval) and then normalized. Mean dopamine concentrations in each epoch were divided by the initial dopamine concentration in the first epoch; local mean response rates in each epoch were divided by terminal rate in the last epoch. Dopamine concentrations (left, circles) decreased across the interval, regardless of the interval duration (10 s, crossed fill; 30 s, solid fill). Simultaneously, responding (right, triangles) increased across the interval, regardless of the interval duration (10 s, crossed fill; 30 s, solid fill).
To further assess the relationship between patterns of dopamine transmission and behavioral activity, we normalized the FI data collected in each epoch using both 10 and 30 s intervals and plotted the results as a function of the interval (Figure 2e). We calculated rate/terminal rate values by dividing the local response rate in each epoch by the terminal rate (defined as the response rate occurring in the final epoch) and then multiplying the result by 100. We calculated dopamine/initial dopamine values by dividing the mean local dopamine concentration in each epoch by the mean dopamine concentration observed in the first epoch and then multiplying the result by 100. As reported previously (Dews, 1970), the pattern of behavioral responding increased similarly under the FI schedule, regardless of the experimenter-defined interval. Unexpectedly, increased responding was inversely related to the pattern of dopamine release, which decreased across the interval in a parallel manner, regardless of the interval duration. Patterns of dopamine concentration were highly correlated (r(8)=0.986; p<0.01) between 10 and 30 s interval conditions, as were patterns of operant responding (r(8)=0.996; p<0.01). In contrast, response patterns were inversely related to dopamine concentration as the interval progressed (10 s rate vs 10 s dopamine: r(8)=−0.976, p<0.01; 10 s rate vs 30 s dopamine: r(8)=−0.948, p<0.01; 30 s rate vs 10 s dopamine: r(8)=−0.96, p<0.01; 30 s rate vs 30 s dopamine: r(8)=−0.920, p<0.01).
After characterizing the pattern of dopamine release engendered by FI tasks, we turned to our primary research question—whether cannabinoids alter the temporal response pattern while simultaneously increasing timing-related dopamine release. As predicted, the cannabinoid agonist WIN 55 212-2 accelerated the temporal response pattern and increased dopamine release during the interval (30 s) of the FI task. Each mouse was initially tested with vehicle and three doses of WIN 55 212-2 (n=6; 0.3–1 mg/kg, intraperitoneally), randomly assigned according to a latin-square design. Figure 3a and b illustrate the effects of the highest WIN 55 212-2 dosage used (1 mg/kg vs vehicle) on the scalloped response pattern observed in a representative animal. The raster plots (top) and corresponding perievent histograms (bottom) show that responding began to increase earlier in the interval following WIN 55 212-2 administration (cf. right vs left). The corresponding mean patterns of dopamine concentration from the representative animal are illustrated in Figure 3c and d. Figure 3e and f show the group’s mean behavioral response patterns and dopamine concentration traces, whereas Figure 4b and c show mean values with standard error for statistical purposes. WIN 55 212-2 dose-dependently produced a leftward shift in the behavioral response pattern and an upward shift in dopamine concentration occurring through the interval. The leftward shift in the scalloped response pattern (Figure 3e) was time-specific. Although we did observe a trend toward an overall increase in response rate, mean response rates were not significantly increased by WIN 55 212-2 treatment (F(4,24)=0.566; NS; Figure 4a). Rather, local response rates were increased in specific epochs (treatment × epoch interaction: F(16,149)=3.4535; p<0.01). Figure 4b illustrates that WIN 55 212-2 significantly increased local response rates in comparison to vehicle in the third epoch (1 mg/kg vs vehicle; p<0.01) and in the fourth epoch (0.6 and 1 mg/kg vs vehicle; p<0.01). Similarly, time-dependent increases in local dopamine concentration were observed through specific epochs of the interval (Figure 4c; treatment × epoch interaction: F(16,124)=9.454; p<0.01). WIN 55 212-2 dose-dependently increased dopamine concentrations in comparison to vehicle in the first (0.3–1 mg/kg vs vehicle; p<0.01), second (0.3–1 mg/kg vs vehicle; p<0.01), third (1 mg/kg vs vehicle; p<0.01), and fourth (1 mg/kg vs vehicle; p<0.01) epoch. Upon completion of the WIN 55 212-2 dose–response determination, all mice were pretreated with the cannabinoid CB1 receptor antagonist/inverse agonist (AM251, 0.75 mg/kg intraperitoenally) 10 min before WIN (1 mg/kg, intraperitoenally) administration to test whether cannabinoid-induced effects on the temporal response pattern and dopamine release were CB1 receptor dependent. AM251 pretreatment prevented the cannabinoid-induced acceleration in timing behavior and the increase in dopamine release through the interval (Figures 3e, f and 4b, c).
Cannabinoid-induced changes in the patterns of behavior and dopamine release engendered by the FI task. (a and b) Cannabinoid administration increases the time of scallop onset. The raster plot and perievent histogram from a mouse show the scalloped behavioral response pattern occurring after vehicle (left) and WIN 55 212-2 treatment (right). (c and d) Cannabinoid administration augments the temporal pattern of dopamine release occurring through the interval in the FI task. The mean corresponding patterns of dopamine concentration occurring in the representative sessions are illustrated in average pseudocolor plots, with dopamine highlighted by the horizontal yellow line. (e) The cannabinoid-induced increase in scallop onset occurs in a dose-dependent manner. Mean behavioral response patterns following cannabinoid administration are plotted as a function of the interval duration. (f) The cannabinoid-induced increase in dopamine concentration occurs in a dose-dependent manner. Mean dopamine concentration traces for each drug treatment conditions are plotted as a function of the interval.
Cannabinoid-induced changes in mean local response rate and dopamine concentration. (a) Cannabinoid treatment did not significantly alter overall response rate. The effect of four cannabinoid drug doses is on response rate (rate per 6 s) is shown. (b) Local response rates were increased by WIN 55 212-2 in specific epochs. Normalized rate data are plotted as a function of the interval. (c) Local dopamine concentrations were increased by WIN 55 212-2 in specific epochs. Mean dopamine concentrations are plotted as a function of the interval. Data are expressed as mean (±SEM) and asterisks indicate a significant difference to vehicle (p⩽0.01).
To assess quantitatively the extent and direction of the cannabinoid-induced change in the temporal response pattern, we applied a previously characterized mathematical analysis known as the index of curvature (Fry et al, 1960; Narayanan et al, 2012). In the index of curvature analysis, a premature acceleration of responding will produce a negative index, while a slower acceleration of responding will produce a positive index. The index of curvature is calculated by dividing the difference between the area under a hypothetical linear response pattern (A=area of OYX) and the area under the observed temporal response pattern (A′= area of Oa′b ′c′YX) by the area under the hypothetical linear response pattern (I=(A−A′)/A; Figure 5a; Fry et al, 1960). WIN 55 212-2 significantly changed the index of curvature (F(5,29)=9.361; p<0.01), with dosages of 0.6 mg/kg (p=0.01) and 1 mg/kg (p<0.01) inducing a negative curvature index in comparison to vehicle. The negative curvature index produced by WIN 55 212-2 required CB1 receptor activation, as the effect was reversed by pretreatment with a low dose of the cannabinoid CB1 receptor antagonist/inverse agonist AM251 (0.75 mg/kg intraperitoneally).
Exogenous and endogenous cannabinoid induced changes in the index of curvature—a computational measure of the extent and direction of change in the temporal response pattern produced by fixed-ratio (FI) schedules. (a) A hypothetical depiction showing how the index of curvature is calculated. (b) The exogenous cannabinoid WIN 55 212-2 produced a negative index of curvature. (c and d) JZL184 induced increases in the endogenous cannabinoid 2-arachidonoylglycerol, but URB597-induced increases in the endogenous cannabinoid anandamide reproduced the WIN 55 212-2 effect by significantly shifting the index of curvature in a negative direction. (e) Although the effect failed to reach significance, the cannabinoid CB1 receptor antagonist AM251 produced a trend toward a positive index of curvature shift. Data are expressed as mean (±SEM) and asterisks indicate a significant difference to vehicle (p⩽0.01).
As the exogenous cannabinoid WIN 55 212-2 induced a significant negative shift in the curvature index, we next sought to investigate whether the endogenous cannabinoid system also has a role in modulating the temporal response pattern engendered by FI schedules. We began by pharmacologically increasing the two best-characterized endocannabinoids, 2-arachidonoylglycerol and anandamide by antagonizing their respective degradative enzymes, monoacylglycerol lipase (MAGL) and fatty acid amide hydrolase (FAAH) (Cravatt et al, 1996; Fegley et al, 2005; Long et al, 2008). JZL184 is a known MAGL inhibitor (Long et al, 2008), previously shown to increase 2-arachidonoylglycerol in the ventral tegmental area and augment cue-evoked dopamine concentrations while animals behave in cued operant tasks (Oleson et al, 2012). During FI responding, elevating 2-arachidonoylglycerol levels significantly modified the temporal response in a manner resembling WIN 55 212-2 (n=7; Figure 5c; F(6,27)=4.763; p=0.013), with 18 mg/kg (p=0.01) and 30 mg/kg (p<0.01) doses producing a negative curvature index in comparison to vehicle. As was the case for WIN 55 212-2, AM251 pretreatment (0.75 mg/kg intraperitoneally) normalized the negative curvature index induced by JZL184. In contrast, selectively increasing anandamide levels with the FAAH inhibitor URB597 (n=8; 0.1, 0.3. 0.6, and 1.0 mg/kg intraperitoneally) failed to alter the temporal response pattern (Figure 5d; NS). Finally, we tested the effects of independently administering a range of AM251 doses on FI responding. AM251 produced a trend toward a positive curvature index, although this effect was not significant (n=8; Figure 5e; NS). We also assessed changes in the postreinforcement pause for all drug treatments (Supplementary Figure S3). WIN 55 212-2 and JZL184 produced a trend toward decreasing the postreinforcement pause, while AM251 produced a trend toward increasing the postreinforcement pause, although none of these effects were statistically significant. It is possible that an FI task with a longer interval would be more conducive to postreinforcement pause analyses.
In an attempt to better understand how cannabinoid manipulations affect the complete behavioral profile produced by FI schedules, we also recorded responses on the inactive lever during the endocannabinoid portion of our study. In addition to producing a scalloped temporal response pattern on the active lever, the interval between reinforced responses in FI schedules produces a critical range of intermittency that fosters irrelevant or adjunctive behavioral responses (Falk, 1971, 1998). James Olds (1982) previously suggested that inactive lever responses observed during FI responding represents such a manifestation of adjunctive behavior. As previously reported with other forms of adjunctive behavior (Killeen, 1975), inactive lever responses during the FI task initially increased before declining through the remainder of the interval (Figure 6a and d). We therefore proceeded to analyze inactive lever responses as a form of adjunctive behavior, and calculated the percentage of time allocated to the inactive lever relative to reinforcement-directed (ie, active lever) responses. Interestingly, both JZL184 and AM251 decreased the percentage of time devoted to the inactive lever, while URB597 failed to change inactive lever responses (data not shown). The 6 mg/kg dose of AM251 was omitted from time-allotment analyses because both active and inactive lever responses were greatly reduced, suggesting that this particular dosage of AM251 generally suppressed behavior. To illustrate the change in time allotted to the inactive lever over time, Figure 6b and e show smooth polynomial functions (ie, splines) curve fitted to mean data for JZL184 and AM251 drug treatment conditions. A two-way repeated-measures ANOVA revealed significant dose by epoch interactions for both JZL184 (Figure 6c; F(16,174)=2.440; p<0.01) and AM251 (Figure 6e; F(8,119)=3.070; p<0.01). Post hoc analysis revealed that in the first epoch of the interval, 30 mg/kg JZL184 significantly decreased (p<0.01) the percentage of time devoted to inactive lever in comparison to vehicle. In the second epoch, where adjunctive behavior was most pronounced, 18 and 30 mg/kg JZL184 significantly decreased time allotted to the inactive lever (p<0.01 vs vehicle), while 1 and 3 mg/kg AM251 also significantly decreased time allotted to the inactive lever (p<0.01 vs vehicle). The JZL184-induced decrease in time devoted to the inactive lever was normalized by pretreatment with a low dose (0.75 mg/kg) of AM251.
Endocannabinoid-induced changes in adjunctive behavior, as assessed by responses on an inactive lever. (a and d) Mean responses on the inactive lever initially increase before declining through the interval. (b and e) Splines model the mean percentage of time devoted to responding on the inactive vs the active lever over the course of the interval following JZL184 (left) and AM251 (right) treatment. (c and f) JZL184 (left) and AM251 (right) significantly decrease the percentage of time spent responding on the inactive lever. Data are expressed as mean (±SEM) and asterisks indicate a significant difference to vehicle (p⩽0.01).
DISCUSSION
An altered perception of time is one of the most commonly reported effects of cannabinoids—occurring in 95% of cannabis users (Tart, 1970). Although subjects report that cannabis makes time pass very slowly (Moreau de Tours, 1845; Tart, 1970), the majority of clinical studies on the subject reveal that cannabinoids increase the rate at which subjective time passes relative to clock time (Atakan et al, 2012; Hicks et al, 1984; O'Leary et al, 2003). This cannabinoid-induced acceleration of an internal clock likely contributes to subjects increasing self-timed behaviors, such as counting and tapping, and overestimating periods of elapsed time (Anderson et al, 2010; Hicks et al, 1984; Lieving et al, 2006; O'Leary et al, 2003). Similar observations are observed in preclinical studies. In accordance with Han and Robinson (2001), in the present study we found that treating rodents with the cannabinoid receptor agonist WIN 55 212-2 shortened the response interval in an FI task, effectively leading animals to show a premature acceleration in the temporal response pattern.
Although it has been suggested that cannabinoids might modulate timing behavior by acting on the mesocorticolimbic dopamine system (Pattij et al, 2008), it remains unclear precisely how cannabinoids modulate dopamine release during timing behavior, and for that matter, how subsecond dopamine release is related to time in FI tasks. To investigate these questions, we measured subsecond dopamine release events in the nucleus accumbens core as animals responded for food under an FI schedule. Our data reveal that changes in accumbal dopamine concentration are inversely related to time when behavior is maintained under an FI schedule. Before reinforcement, we observed a decrease in dopamine concentration that coincided with the interval and lawfully reset to different durations (10 vs 30 s). Importantly, the decrease in dopamine concentration corresponded to an increase in local response rate, suggesting that a decline in dopamine concentration is related to the accelerating response pattern that characterizes the FI scallop. Taken together, these data suggest that during behavior maintained in the FI task, dopamine is principally related to the dominant cue predicting reinforcement availability: time. However, we acknowledge that it remains unclear whether dopamine is exclusively encoding time, amplifying an action sequence using temporal information, or both.
The historical psychopharmacology and behavioral neuroscience literatures suggest that dopamine has a complex role during behavior measured under conditions of periodic reinforcement. Early psychopharmacology studies demonstrated that indirect dopamine agonists, depending on the dosage used, selectively increase response rates under FI schedules without changing, and sometimes even decreasing response rates under fixed-ratio (FR) schedules (Kelleher and Morse, 1968; Morse and Kelleher, 1970; Barrett, 1976; McKearney, 1974, 1982; Spealman et al, 1977). Interestingly, pharmacologically increasing dopamine concentrations using indirect dopamine agonists was reported to accelerate exclusively responding in the early portions of the interval (Smith, 1964), thereby causing the FI-engendered temporal response pattern to appear more linear in nature. As the increase in local response rate occurs at a time when competing adjunctive behaviors are most prevalent (Falk, 1971; Killeen, 1975), these data may suggest that high concentration surges in dopamine release promote reinforcement-driven responding over reinforcement-irrelevant behaviors when reinforcement is periodically available. Similarly, stimulating high-concentration dopamine release events promotes drug self-administration while reducing stereotypic behavioral responses (Phillips et al, 2003). However, disrupting accumbal dopamine neurotransmission also promotes reinforcement-driven responses over adjunctive behaviors (Bakshi and Kelley, 1991; Robbins and Koob, 1980). Indeed, ablating dopamine terminals or antagonizing dopamine receptors in the NAc resulted in animals devoting more time toward obtaining primary reinforcement over competing adjunctive responses (Robbins and Koob, 1980; Bakshi and Kelley, 1991; Baldo et al, 2002). Taken together, these seemingly paradoxical data suggest that high and/or low dopamine concentrations promote reinforcement-driven behavior, while intermediate dopamine concentrations promote adjunctive behavior during a critical period of intermittency produced by periodic reinforcement (Falk, 1971).
When considered in the context of the existing literature, our real-time neurochemical observations may suggest that a declining pattern of dopamine release associated with interval duration in FI schedules may switch the animal’s primary motivation from reinforcement directed in nature to competing adjunctive responses before ultimately ceasing to regulate motivated behavior at the interval terminus. This interpretation is congruous with the canonical view of the basal ganglia in the generation of motoric responses (Albin et al, 1989; DeLong, 1990), a theory that received recent validation using modern neuroscientific and computational techniques (Dreyer et al, 2010; Kravitz et al, 2012; but see Cui et al, 2013). According to this theory, high concentration surges in dopamine release activate striatal medium spiny neurons comprising the direct pathway to promote directed action sequences (eg, reinforcement-directed active lever response), whereas moderate dopamine concentrations activate striatal medium spiny neurons comprising the indirect pathway to inhibit directed action sequences, and possibly switch the motivational drive of the animal to seek out alternative options (Dreyer et al, 2010; Kravitz et al, 2012). It is also important to note that these distinct dopaminergic responses are unlikely to mediate directly a behavioral response, but rather modulate converging cortical, hippocampal, and amygdalar input into the striatum (Brady and O’Donnell, 2004; Floresco et al, 2001). Thus, we propose that dopamine is related to the FI-engendered behavioral repertoire as follows: (1) a high concentration surge of dopamine release to food reinforcement promotes a directed action sequence of food retrieval via a direct pathway-dependent mechanism, (2) the initial high increase in dopamine concentration at the onset of the interval promotes the initiation of regulated active lever responding via a direct pathway-dependent mechanism, (3) as the interval elapses, dopamine concentrations decline to a moderate level and the animal is motivated to seek out alternative options via indirect pathway activation; here, dopamine-dependent adjunctive behaviors (Robbins and Koob, 1980) occur at a maximal level and compete against primary reinforcement-directed responses, and (4) as dopamine concentrations decline further toward the end of the interval, dopamine modulation of the indirect pathway is removed, thereby eliminating the indirect pathway promotion of adjunctive behavior. Now dopaminergic modulation of the converging input into the accumbens is lost, and an unchecked high response pattern of reinforcement-directed behavior predominates. We would like to acknowledge that our speculative model only describes rapid dopamine concentration changes observed in the nucleus accumbens core, and it is likely that divergent patterns of dopamine concentration may be observed along different timescales in distinct neural substrates and that dopamine levels below our limit of detection may also have a role (Narayanan et al, 2012). According to our model, if dopamine concentrations were enhanced at interval onset, yet still sufficiently decreased at the interval terminus, competing forces over reinforcement-directed behavior would be minimized and a behavioral repertoire that is dominated by obtaining primary reinforcement through the interval would emerge. An accelerated temporal response pattern and a negative index of curvature would characterize responding on the active lever in such a scenario.
In accordance with our proposed model, we found that the synthetic cannabinoid WIN 55 212-2 dose-dependently increased local accumbal dopamine concentrations observed during the interval in a manner that coincided with an accelerated temporal response pattern on the active lever. Theoretically, the increased dopamine concentration observed at the interval onset motivates primary reinforcement-directed responses via a direct pathway-dependent mechanism (Dreyer et al, 2010; Kravitz et al, 2012). Competing motivation for adjunctive behavior is minimized as direct pathway activation is maintained before a rapid decline in dopamine concentration toward the terminal segment of the interval. This results in an accelerated temporal response pattern, an effect that was revealed by the occurrence of a negative curvature index.
We previously demonstrated that the endocannabinoid 2-arachidonoylglycerol, but not anandamide, facilitates dopamine release and cue-motivated behavior (Oleson et al, 2012). In the present study, we replicated this distinction by showing that increased 2-arachidonoylglycerol, but not anandamide, concentrations are sufficient to accelerate the temporal response pattern and induced a negative curvature index in a manner similar to WIN 55 212-2. Surprisingly, we found that both increasing 2-arachidonoylglycerol levels and disrupting endocannabinoids signaling using the CB1 receptor antagonist/inverse agonist AM251 reduced adjunctive behavior despite producing opposite trends in the curvature index. These data suggest that endocannabinoids might have a modulatory role in promoting behavioral fitness by motivating behaviors directed at both obtaining primary reinforcement and alternative options. Within an ethological context, it might be advantageous for an animal’s motivation to switch from feeding to foraging during periods of time in which the primary food source is unavailable. Our endocannabinoid data suggest that ongoing tone might be required to modulate the incentive to forage, while increased concentrations of 2-arachidonoylglycerol might promote the incentive to obtain the primary food source. On the basis of previous data (Melis et al, 2004; Oleson et al, 2012), we speculate that in the latter case, high-frequency firing of dopamine neurons would increase the on-demand synthesis of 2-arachidonoylglycerol, which would then function to amplify dopamine release and ultimately motivate behavior aimed at obtaining the primary food source.
Previous accounts of interval timing proposed that the brain contains a neural timer that resets to zero by the occurrence of reinforcement (Gallistel and Gibbon, 2000; Gibbon, 1977) and that dopamine is critically involved in second-to-second estimation (Bromberg-Martin et al, 2010; Buhusi and Meck, 2005; Coull et al, 2010; Coull et al, 2012; Matell and Meck, 2000; Meck et al, 2008; Narayanan et al, 2012; Wittmann, 2013). Consistent with this theory, we observed a decrease in subsecond dopamine concentrations throughout the interval of an FI task that resets after each reinforcement-evoked dopamine event. These data support the view that dopamine transmission has multiple roles during adaptive behavior (Bromberg-Martin et al, 2010), and further demonstrate that the particular patterns of dopamine transmission that are recruited during operant behavior are greatly influenced by the rules that dictate reinforcement availability (ie, the reinforcement schedule). Cannabinoids capably accelerate this mechanism, in part, by augmenting time-related patterns of dopamine release in a modulatory manner.
FUNDING AND DISCLOSURE
This work was supported by NIH Grant R01DA025890 (JFC). The authors declare no conflict of interest.
References
Albin RL, Young AB, Penney JB (1989). The functional anatomy of basal ganglia disorders. Trends Neurosci 12: 366–375.
Anderson BM, Rizzo M, Block RI, Pearlson GD, O'Leary DS (2010). Sex, drugs, and cognition: effects of marijuana. J Psychoactive Drugs 42: 413–424.
Atakan Z, Morrison P, G Bossong M, Martin-Santos R, A Crippa J (2012). The effect of cannabis on perception of time: a critical review. Curr Pharm Des 18: 4915–4922.
Baldo BA, Sadeghian K, Basso AM, Kelley AE (2002). Effects of selective dopamine D1 or D2 receptor blockade within nucleus accumbens subregions on ingestive behavior and associated motor activity. Behav Brain Res 137: 165–177.
Bakshi VP, Kelley AE (1991). Dopaminergic regulation of feeding behavior: I. Differential effects of haloperidol microinfusion into three striatal subregions. Psychobiology 19: 223–232.
Barrett JE (1976). Effects of alcohol, chlordiazepoxide, cocaine and pentobarbital on responding maintained under fixed-interval schedules of food or shock presentation. J Pharmacol Exp Ther 196: 605–615.
Bossert JM, Poles GC, Wihbey KA, Koya E, Shaham Y (2007). Differential effects of blockade of dopamine D1-family receptors in nucleus accumbens core or shell on reinstatement of heroin seeking induced by contextual and discrete cues. J Neurosci 27: 12655–12663.
Brady AM, O’Donnell P (2004). Dopaminergic modulation of prefrontal cortical input to nucleus accumbens neurons in vivo. J Neurosci 24: 1040–1049.
Bromberg-Martin ES, Matsumoto M, Hikosaka O (2010). Dopamine in motivational control: rewarding, aversive, and alerting. Neuron 68: 815–834.
Bromberg-Martin ES, Matsumoto M, Nakahara H, Hikosaka O (2010). Multiple timescales of memory in lateral habenula and dopamine neurons. Neuron 67: 499–510.
Buhusi CV, Meck WH (2005). What makes us tick? Functional and neural mechanisms of interval timing. Nat Rev Neurosci 6: 755–765.
Chaudhri N, Sahuque LL, Schairer WW, Janak PH (2009). Separable roles of the nucleus accumbens core and shell in context-and cue-induced alcohol-seeking. Neuropsychopharmacology 35: 783–791.
Cheer JF, Wassum KM, Heien ML, Phillips PE, Wightman RM (2004). Cannabinoids enhance subsecond dopamine release in the nucleus accumbens of awake rats. J Neurosci 24: 4393–4400.
Cheng R-K, Hakak OL, Meck WH (2007). Habit formation and the loss of control of an internal clock: inverse relationship between the level of baseline training and the clock-speed enhancing effects of methamphetamine. Psychopharmacology 193: 351–362.
Clark JJ, Sandberg SG, Wanat MJ, Gan JO, Horne EA, Hart AS et al (2009). Chronic microsensors for longitudinal, subsecond dopamine detection in behaving animals. Nat Methods 7: 126–129.
Coull JT, Cheng R-K, Meck WH (2010). Neuroanatomical and neurochemical substrates of timing. Neuropsychopharmacology 36: 3–25.
Coull JT, Hwang HJ, Leyton M, Dagher A (2012). Dopamine precursor depletion impairs timing in healthy volunteers by attenuating activity in putamen and supplementary motor area. J Neurosci 32: 16704–16715.
Cravatt BF, Giang DK, Mayfield SP, Boger DL, Lerner RA, Gilula NB (1996). Molecular characterization of an enzyme that degrades neuromodulatory fatty-acid amides. Nature 384: 83–87.
Cui G, Jun SB, Jin X, Pham MD, Vogel SS, Lovinger DM et al (2013). Concurrent activation of striatal direct and indirect pathways during action initiation. Nature 494: 238–242.
DeLong MR (1990). Primate models of movement disorders of basal ganglia origin. Trends Neurosci 13: 281–285.
Dews PB (1970). The theory of fixed-interval responding. In: Schoenfeld WN (ed). The Theory of Reinforcement Schedules. Appleton Century Crofts: New York, NY. pp 43–61.
Dreyer JK, Herrik KF, Berg RW, Hounsgaard JD (2010). Influence of phasic and tonic dopamine release on receptor activation. J Neurosci 30: 14273–14283.
Falk JL (1971). The nature and determinants of adjunctive behavior. Physiol Behav 6: 577–588.
Falk JL (1998). Drug abuse as an adjunctive behavior. Drug Alcohol Depend 52: 91–98.
Fegley D, Gaetani S, Duranti A, Tontini A, Mor M, Tarzia G et al (2005). Characterization of the fatty acid amide hydrolase inhibitor cyclohexyl carbamic acid 3′-carbamoyl-biphenyl-3-yl ester (URB597): effects on anandamide and oleoylethanolamide deactivation. J Pharmacol Exp Ther 313: 352–358.
Floresco SB, Todd CL, Grace AA (2001). Glutamatergic afferents from the hippocampus to the nucleus accumbens regulate activity of ventral tegmental area dopamine neurons. J Neurosci 21: 4915–4922.
Fry W, Kelleher RT, Cook L (1960). A mathematical index of performance on fixed-interval schedules of reinforcement. J Exp Anal Behav 3: 193–199.
Gallistel CR, Gibbon J (2000). Time, rate, and conditioning. Psychol Rev 107: 289.
Gibbon J (1977). Scalar expectancy theory and Weber’s law in animal timing. Psychol Rev 84: 279.
Han C, Robinson JK (2001). Cannabinoid modulation of time estimation in the rat. Behav Neurosci 115: 243.
Heien MLAV, Phillips PEM, Stuber GD, Seipel AT, Wightman RM (2003). Overoxidation of carbon-fiber microelectrodes enhances dopamine adsorption and increases sensitivity. Analyst 128: 1413–1419.
Hicks RE, Gualtieri CT, Mayo J Jr, Perez-Reyes M (1984). Cannabis, atropine, and temporal information processing. Neuropsychobiology 12: 229–237.
Ito R, Hayen A (2011). Opposing roles of nucleus accumbens core and shell dopamine in the modulation of limbic information processing. J Neurosci 31: 6001–6007.
Keithley RB, Mark Wightman R, Heien ML (2009). Multivariate concentration determination using principal component regression with residual analysis. Trends Anal Chem 28: 1127–1136.
Kelleher RT, Morse WH (1968). Determinants of the specificity of behavioral effects of drugs. Rev Physiol Biochem Exp Pharmacol 60: 1–56.
Killeen PR (1975). On the temporal control of behavior. Psychol Rev 82: 89–115.
Kravitz AV, Tye LD, Kreitzer AC (2012). Distinct roles for direct and indirect pathway striatal neurons in reinforcement. Nat Neurosci 15: 816–818.
Lieving LM, Lane SD, Cherek DR, Tcheremissine OV (2006). Effects of marijuana on temporal discriminations in humans. Behav Pharmacol 17: 173–183.
Long JZ, Li W, Booker L, Burston JJ, Kinsey SG, Schlosburg JE et al (2008). Selective blockade of 2-arachidonoylglycerol hydrolysis produces cannabinoid behavioral effects. Nat Chem Biol 5: 37–44.
Matell MS, Meck WH (2000). Neuropsychological mechanisms of interval timing behavior. BioEssays 22: 94–103.
McKearney JW (1974). Effects of d-amphetamine, morphine and chlorpromazine on responding under fixed-interval schedules of food presentation or electric shock presentation. J Pharmacol Exp Ther 190: 141–153.
McKearney JW (1982). Effects of dopamine uptake inhibitors on schedule-controlled behavior in the squirrel monkey. Psychopharmacology 78: 377–379.
Meck WH, Penney TB, Pouthas V (2008). Cortico-striatal representation of time in animals and humans. Curr Opin Neurobiol 18: 145–152.
Melis M, Pistis M, Perra S, Muntoni AL, Pillolla G, Gessa GL (2004). Endocannabinoids mediate presynaptic inhibition of glutamatergic transmission in rat ventral tegmental area dopamine neurons through activation of CB1 receptors. J Neurosci 24: 53–62.
Moreau de Tours J-J (1845) Du hachisch et de l'aliénation mentale. Études Psychologiques: Paris, France.
Morse WH, Kelleher RT (1970). Schedules as fundamental determinants of behavior. In: Schoenfeld WN (ed). The Theory of Reinforcement Schedules. Appleton Century Crofts: New York, NY. pp 139–185.
Narayanan NS, Land BB, Solder JE, Deisseroth K, DiLeone RJ (2012). Prefrontal D1 dopamine signaling is required for temporal control. Proc Natl Acad Sci 109: 20726–20731.
Olds ME (1982). Reinforcing effects of morphine in the nucleus accumbens. Brain Res 237: 429–440.
Oleson EB, Beckert MV, Morra JT, Lansink CS, Cachope R, Abdullah RA et al (2012). Endocannabinoids shape accumbal encoding of cue-motivated behavior via CB1 receptor activation in the ventral tegmentum. Neuron 73: 360–373.
O'Leary DS, Block RI, Turner BM, Koeppel J, Magnotta VA, Ponto LB et al (2003). Marijuana alters the human cerebellar clock. NeuroReport 14: 1145–1151.
Pattij T, Wiskerke J, Schoffelmeer AN (2008). Cannabinoid modulation of executive functions. Eur J Pharmacol 585: 458–463.
Phillips PE, Stuber GD, Heien ML, Wightman RM, Carelli RM (2003). Subsecond dopamine release promotes cocaine seeking. Nature 422: 614–618.
Robbins TW, Koob GF (1980). Selective disruption of displacement behaviour by lesions of the mesolimbic dopamine system. Nature 285: 409–412.
Smith CB (1964). Effects of d-amphetamine upon operant behavior of pigeons: enhancement by reserpine. J Pharmacol Exp Ther 146: 167–174.
Spealman RD, Goldberg SR, Kelleher RT, Goldberg DM, Charlton JP (1977). Some effects of cocaine and two cocaine analogs on schedule-controlled behavior of squirrel monkeys. J Pharmacol Exp Ther 202: 500–509.
Tart CT (1970). Marijuana intoxication: common experiences. Nature 226: 701–704.
Tinklenberg JR, Roth WT, Kopell BS (1976). Marijuana and ethanol: differential effects on time perception, heart rate, and subjective response. Psychopharmacology 49: 275–279.
Wittmann M (2013). The inner sense of time: how the brain creates a representation of duration. Nat Rev Neuroscience 14: 217–223.
Acknowledgements
We thank Dr Lindsey Hamilton for helpful comments in the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Neuropsychopharmacology website
Rights and permissions
About this article
Cite this article
Oleson, E., Cachope, R., Fitoussi, A. et al. Cannabinoid Receptor Activation Shifts Temporally Engendered Patterns of Dopamine Release. Neuropsychopharmacol 39, 1441–1452 (2014). https://doi.org/10.1038/npp.2013.340
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2013.340
Keywords
This article is cited by
-
Clinical, Cognitive, and Neurobiological Correlates of Impaired Timing Abilities Associate to Cannabis Use: a Systematic Review
International Journal of Mental Health and Addiction (2023)
-
Dopamine, behavior, and addiction
Journal of Biomedical Science (2021)
-
Inhibition of endocannabinoid degradation rectifies motivational and dopaminergic deficits in the Q175 mouse model of Huntington’s disease
Neuropsychopharmacology (2018)
-
Persistent effects of chronic Δ9-THC exposure on motor impulsivity in rats
Psychopharmacology (2015)