Abstract
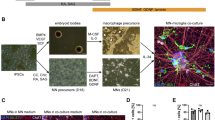
This protocol details methods to isolate and purify astrocytes and motoneurons (MNs) from the chick lumbar spinal cord. In addition, an approach to study the influences of astrocyte secreted factors on MNs is provided. Astrocytes are isolated between embryonic days 10 and 12 (E10–12), propagated in serum (2–3 h) and differentiated in chemically defined medium (3–4 h). When prepared according to this protocol, astrocyte cultures are more than 98% pure when assessed using the astrocyte-specific markers glial fibrillary acidic protein (GFAP) and S100β. MNs are isolated between E5.5 and 6.0 (3–4 h) using a procedure that takes selective advantage of the large size of these cells. These cultures can be maintained using individual trophic factors, target-derived factors or astrocyte-derived factors, the preparation of which is also described (5–6 h). All or part of these techniques can be used to investigate a variety of processes that occur during nervous system development and disease or after injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Nedergaard, M., Ransom, B. & Goldman, S.A. New roles for astrocytes: redefining the functional architecture of the brain. Trends Neurosci. 26, 523–530 (2003).
Vernadakis, A. Neuron-glia interrelations. Int. Rev. Neurobiol. 30, 149–224 (1988).
Svendsen, C.N. The amazing astrocyte. Nature 417, 29–32 (2002).
Ransom, B., Behar, T. & Nedergaard, M. New roles for astrocytes (stars at last). Trends Neurosci. 26, 520–522 (2003).
Maragakis, N.J. & Rothstein, J.D. Mechanisms of disease: astrocytes in neurodegenerative disease. Nat. Clin. Pract. Neurol. 2, 679–689 (2006).
Silver, J. & Miller, J.H. Regeneration beyond the glial scar. Nat. Rev. Neurosci. 5, 146–156 (2004).
McCarthy, K.D. & de Vellis, J. Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue. J. Cell Biol. 85, 890–902 (1980).
Qian, J.A., Bull, M.S. & Levitt, P. Target-derived astroglia regulate axonal outgrowth in a region-specific manner. Dev. Biol. 149, 278–294 (1992).
Schwartz, J.P. & Wilson, D.J. Preparation and characterization of type 1 astrocytes cultured from adult rat cortex, cerebellum, and striatum. Glia 5, 75–80 (1992).
Fischer, G. Growth requirements of immature astrocytes in serum-free hormonally defined media. J. Neurosci. Res. 12, 543–552 (1984).
Bottenstein, J. Growth and differentiation of neural cells in defined media. In Cell Culture in the Neurosciences (eds. Bottenstein, J. & Sato, G.) 3–43 (Plenum Pub Corp., New York, 1985).
Ang, L.C., Bhaumick, B., Munoz, D.G., Sass, J. & Juurlink, B.H. Effects of astrocytes, insulin and insulin-like growth factor I on the survival of motoneurons in vitro . J. Neurol. Sci. 109, 168–172 (1992).
Banker, G.A. Trophic interactions between astroglial cells and hippocampal neurons in culture. Science 209, 809–810 (1980).
Cassina, P. et al. Astrocyte activation by fibroblast growth factor-1 and motor neuron apoptosis: implications for amyotrophic lateral sclerosis. J. Neurochem. 93, 38–46 (2005).
Eagleson, K.L., Raju, T.R. & Bennett, M.R. Motoneurone survival is induced by immature astrocytes from developing avian spinal cord. Brain Res. 349, 95–104 (1985).
Elmariah, S.B., Oh, E.J., Hughes, E.G. & Balice-Gordon, R.J. Astrocytes regulate inhibitory synapse formation via Trk-mediated modulation of postsynaptic GABAA receptors. J Neurosci. 25, 3638–3650 (2005).
Pfrieger, F.W. & Barres, B.A. Synaptic efficacy enhanced by glial cells in vitro . Science. 277, 1684–1687 (1997).
Henderson, C.E., Bloch-Gallego, E. & Camu, W. Purified embryonic motoneurons. In Nerve Cell Cultures: A Practical Approach (eds. Cohen, J. & Wilkin, G.) 69–81 (Oxford University, London, 1995).
Oppenheim, R.W. et al. Biological studies of a putative avian muscle-derived neurotrophic factor that prevents naturally occurring motoneuron death in vivo . J. Neurobiol. 24, 1065–1079 (1993).
Henderson, C.E. et al. Neurotrophins promote motor neuron survival and are present in embryonic limb bud. Nature 363, 266–270 (1993).
Houenou, L.J., Li, L., Lo, A.C., Yan, Q. & Oppenheim, R.W. Naturally occurring and axotomy-induced motoneuron death and its prevention by neurotrophic agents: a comparison between chick and mouse. Prog. Brain Res. 102, 217–226 (1994).
Wu, V.W. & Schwartz, J.P. Cell culture models for reactive gliosis: new perspectives. J. Neurosci. Res. 51, 675–681 (1998).
Bloch-Gallego, E. et al. Survival in vitro of motoneurons identified or purified by novel antibody-based methods is selectively enhanced by muscle-derived factors. Development 111, 221–232 (1991).
Arce, V. et al. Cardiotrophin-1 requires LIFRbeta to promote survival of mouse motoneurons purified by a novel technique. J. Neurosci. Res. 55, 119–126 (1999).
Sendtner, M., Pei, G., Beck, M., Schweizer, U. & Wiese, S. Developmental motoneuron cell death and neurotrophic factors. Cell Tissue Res. 301, 71–84 (2000).
Oppenheim, R.W. Neurotrophic survival molecules for motoneurons: an embarrassment of riches. Neuron 17, 195–197 (1996).
Taylor, A.R. et al. Astrocyte and muscle-derived secreted factors differentially regulate motoneuron survival. J. Neurosci. 27, 634–644 (2007).
Comella, J.X., Sanz-Rodriguez, C., Aldea, M. & Esquerda, J.E. Skeletal muscle-derived trophic factors prevent motoneurons from entering an active cell death program in vitro . J. Neurosci. 14, 2674–2686 (1994).
Dolcet, X., Egea, J., Soler, R.M., Martin-Zanca, D. & Comella, J.X. Activation of phosphatidylinositol 3-kinase, but not extracellular-regulated kinases, is necessary to mediate brain-derived neurotrophic factor-induced motoneuron survival. J. Neurochem. 73, 521–531 (1999).
Soler, R.M. et al. Receptors of the glial cell line-derived neurotrophic factor family of neurotrophic factors signal cell survival through the phosphatidylinositol 3-kinase pathway in spinal cord motoneurons. J. Neurosci. 19, 9160–9169 (1999).
Christopherson, K.S. et al. Thrombospondins are astrocyte-secreted proteins that promote CNS synaptogenesis. Cell 120, 421–433 (2005).
Acknowledgements
This work was supported by National Institutes of Health grant NS036081 and by funds from the WFUSM Brian White ALS Foundation (C.E.M.). The authors would like to thank David Prevette and William Safrit for their assistance with the generation of dissection images.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Taylor, A., Robinson, M. & Milligan, C. In vitro methods to prepare astrocyte and motoneuron cultures for the investigation of potential in vivo interactions. Nat Protoc 2, 1499–1507 (2007). https://doi.org/10.1038/nprot.2007.208
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.208
This article is cited by
-
Coculture of Primary Motor Neurons and Schwann Cells as a Model for In Vitro Myelination
Scientific Reports (2015)
-
Cytoprotective effects of urinary trypsin inhibitor on astrocytes injured by sustained compression
Molecular Biology Reports (2014)
-
Direct live monitoring of heterotypic axon-axon interactions in vitro
Nature Protocols (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.