Abstract
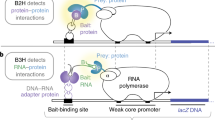
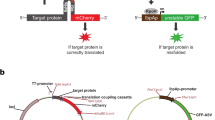
We have developed a general experimental strategy that enables the quantitative detection of dynamic protein–protein interactions in intact living cells, based on protein-fragment complementation assays (PCAs). In this method, protein interactions are coupled to refolding of enzymes from cognate fragments where reconstitution of enzyme activity acts as the detector of a protein interaction. We have described a number of assays with different reporter readouts, but of particular value to studies of protein interaction dynamics are assays based on enzyme reporters that catalyze the creation of products, thus taking advantage of the amplification of signal afforded. Here we describe protocols for one such PCA based on the enzyme TEM β-lactamase as a reporter in mammalian cells. The β-lactamase PCA consists of fusing complementary fragments of β-lactamase to two proteins of interest. If the proteins interact, the fragments are brought together and fold into active β-lactamase. Here we describe a protocol for this PCA that can be completed in a few hours, using two different substrates that are converted to fluorescent or colored products by β-lactamase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Remy, I., Campbell-Valois, F.X., Ghaddar, G., Aquin, S. & Michnick, S.W. Detection of protein interactions and library screening with protein-fragment complementation assays. in Protein–Protein Interactions: A Molecular Cloning Manual 2nd edn., Ch. 33, 637–672 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2005).
Remy, I. & Michnick, S.W. Clonal selection and in vivo quantitation of protein interactions with protein-fragment complementation assays. Proc. Natl. Acad. Sci. USA 96, 5394–5399 (1999).
Pelletier, J.N., Campbell-Valois, F.X. & Michnick, S.W. Oligomerization domain-directed reassembly of active dihydrofolate reductase from rationally designed fragments. Proc. Natl. Acad. Sci. USA 95, 12141–12146 (1998).
Remy, I. & Michnick, S.W. A cDNA library functional screening strategy based on fluorescent protein complementation assays to identify novel components of signaling pathways. Methods 32, 381–388 (2004).
Galarneau, A., Primeau, M., Trudeau, L.E. & Michnick, S.W. Beta-lactamase protein fragment complementation assays as in vivo and in vitro sensors of protein protein interactions. Nat. Biotechnol. 20, 619–622 (2002).
Wehrman, T., Kleaveland, B., Her, J.H., Balint, R.F. & Blau, H.M. Protein–protein interactions monitored in mammalian cells via complementation of beta-lactamase enzyme fragments. Proc. Natl. Acad. Sci. USA 99, 3469–3474 (2002).
Luker, K.E. et al. Kinetics of regulated protein–protein interactions revealed with firefly luciferase complementation imaging in cells and living animals. Proc. Natl. Acad. Sci. USA 101, 12288–12293 (2004).
Paulmurugan, R. & Gambhir, S.S. Monitoring protein–protein interactions using split synthetic Renilla luciferase protein-fragment-assisted complementation. Anal. Chem. 75, 1584–1589 (2003).
Paulmurugan, R., Umezawa, Y. & Gambhir, S.S. Noninvasive imaging of protein–protein interactions in living subjects by using reporter protein complementation and reconstitution strategies. Proc. Natl. Acad. Sci. USA 99, 15608–15613 (2002).
Ghosh, I., Hamilton, A.D. & Regan, L. Antiparallel leucine zipper-directed protein reassembly: application to the green fluorescent protein. J. Am. Chem. Soc. 122, 5658–5659 (2000).
Hu, C.D., Chinenov, Y. & Kerppola, T.K. Visualization of interactions among bZIP and Rel family proteins in living cells using bimolecular fluorescence complementation. Mol. Cell 9, 789–798 (2002).
Remy, I., Montmarquette, A. & Michnick, S.W. PKB/Akt modulates TGF-beta signalling through a direct interaction with Smad3. Nat. Cell Biol. 6, 358–365 (2004).
Zhang, J., Campbell, R.E., Ting, A.Y. & Tsien, R.Y. Creating new fluorescent probes for cell biology. Nat. Rev. Mol. Cell Biol. 3, 906–918 (2002).
Xu, X. et al. Imaging protein interactions with bioluminescence resonance energy transfer (BRET) in plant and mammalian cells and tissues. Proc. Natl. Acad. Sci. USA 104, 10264–10269 (2007).
Remy, I., Campbell-Valois, F.X. & Michnick, S.W. Detection of protein–protein interactions using a simple survival protein-fragment complementation assay (PCA) based on the enzyme dihydrofolate reductase. Nat. Protoc. 9, 2120–2125 (2007).
Zlokarnik, G. et al. Quantitation of transcription and clonal selection of single living cells with beta-lactamase as reporter. Science 279, 84–88 (1998).
Zlokarnik, G. Fusions to beta-lactamase as a reporter for gene expression in live mammalian cells. Methods Enzymol. 326, 221–244 (2000).
Acknowledgements
This work was supported by the Canadian Institute of Health Research (MOP-152556).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Remy, I., Ghaddar, G. & Michnick, S. Using the β-lactamase protein-fragment complementation assay to probe dynamic protein–protein interactions. Nat Protoc 2, 2302–2306 (2007). https://doi.org/10.1038/nprot.2007.356
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.356
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.