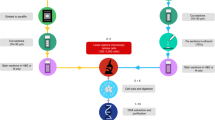
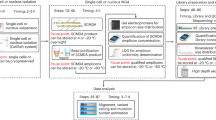
Abstract
A comprehensive genomic analysis of single cells is instrumental for numerous applications in tumor genetics, clinical diagnostics and forensic analyses. Here, we provide a protocol for single-cell isolation and whole genome amplification, which includes the following stages: preparation of single-cell suspensions from blood or bone marrow samples and cancer cell lines; their characterization on the basis of morphology, interphase fluorescent in situ hybridization pattern and antibody staining; isolation of single cells by either laser microdissection or micromanipulation; and unbiased amplification of single-cell genomes by either linker-adaptor PCR or GenomePlex library technology. This protocol provides a suitable template to screen for chromosomal copy number changes by conventional comparative genomic hybridization (CGH) or array CGH. Expected results include the generation of several micrograms of DNA from single cells, which can be used for CGH or other analyses, such as sequencing. Using linker-adaptor PCR or GenomePlex library technology, the protocol takes 72 or 30 h, respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Speicher, M.R. & Carter, N.P. The new cytogenetics: blurring the boundaries with molecular biology. Nat. Rev. Genet. 6, 782–792 (2005).
Telenius, H. et al. Cytogenetic analysis by chromosome painting using DOP-PCR amplified flow-sorted chromosomes. Genes Chromosomes Cancer 4, 257–263 (1992).
Speicher, M.R. et al. Molecular cytogenetic analysis of archived, paraffin embedded solid tumors by comparative genomic hybridization after universal PCR. Hum. Mol. Genet. 2, 1907–1914 (1993).
Speicher, M.R. et al. Correlation of microscopic phenotype with genotype in a formalin fixed, paraffin embedded testicular germ cell tumor using universal DNA amplification, comparative genomic hybridization and interphase cytogenetics. Am. J. Path. 146, 1332–1340 (1995).
Wiltshire, R.N. et al. Direct visualization of the clonal progression of primary cutaneous melanoma: application of tissue microdissection and comparative genomic hybridization. Cancer Res. 55, 3954–3957 (1995).
Klein, C.A. et al. Comparative genomic hybridization, loss of heterozygosity, and DNA sequence analysis of single cells. Proc. Natl. Acad. Sci. USA 96, 4494–4499 (1999).
Wells, D., Sherlock, J.K., Handyside, A.H. & Delhanty, J.D. Detailed chromosomal and molecular genetic analysis of single cells by whole genome amplification and comparative genomic hybridisation. Nucleic Acids Res. 27, 1214–1218 (1999).
Voullaire, L., Wilton, L., Slater, H. & Williamson, R. Detection of aneuploidy in single cells using comparative genomic hybridization. Prenat. Diagn. 19, 846–851 (1999).
Fragouli, E. et al. Comparative genomic hybridization analysis of human oocytes and polar bodies. Hum. Reprod. 21, 2319–2328 (2006).
Hu, D.G., Webb, G. & Hussey, N. Aneuploidy detection in single cells using DNA array-based comparative genomic hybridization. Mol. Hum. Reprod. 10, 283–289 (2004).
Gutierrez-Mateo, C. et al. Aneuploidy study of human oocytes first polar body comparative genomic hybridization and metaphase II fluorescence in situ hybridization analysis. Hum. Reprod. 19, 2859–2868 (2004).
Lizardi, P.M. et al. Mutation detection and single-molecule counting using isothermal rolling-circle amplification. Nat. Genet. 19, 225–232 (1998).
Zhong, X.B., Lizardi, P.M., Huang, X.H., Bray-Ward, P.L. & Ward, D.C. Visualization of oligonucleotide probes and point mutations in interphase nuclei and DNA fibers using rolling circle DNA amplification. Proc. Natl. Acad. Sci. USA 98, 3940–3945 (2001).
Lage, J.M. et al. Whole genome analysis of genetic alterations in small DNA samples using hyperbranched strand displacement amplification and array-CGH. Genome Res. 13, 294–307 (2003).
LeCaignec, C. et al. Single-cell chromosomal imbalances detection by array CGH. Nucleic Acids Res. 34, e68 (2006).
Spits, C. et al. Whole-genome multiple displacement amplification from single cells. Nat. Protoc. 1, 1965–1970 (2006).
Langer, S., Geigl, J.B., Gangnus, R. & Speicher, M.R. Sequential application of interphase-FISH and CGH to single cells. Lab. Invest. 85, 582–592 (2005).
Fiegler, H. et al. High resolution array-CGH analysis of single cells. Nucleic Acids Res. 35, e15 Epub 2006 Dec 18 (2007).
Gangnus, R., Langer, S., Breit, S., Pantel, K. & Speicher, M.R. Genomic profiling of viable and proliferative micrometastatic cells from early stage breast cancer patients. Clin. Cancer Res. 10, 3457–3464 (2004).
Thalhammer, S., Langer, S., Speicher, M.R., Heckl, W.M. & Geigl, J.B. Generation of chromosome painting probes from single chromosomes by laser microdissection and linker-adaptor PCR. Chromosome Res. 12, 337–343 (2004).
Fiegler, H., Redon, R. & Carter, N.P. Construction and use of spotted large-insert clone DNA microarrays for the detection of genomic copy number changes. Nat. Protoc. 2, 577–587 (2007).
Espina, A. et al. Laser-capture microdissection. Nat. Protoc. 1, 586–603 (2006).
Geigl, J.B., Uhrig, S. & Speicher, M.R. Multiplex-fluorescence in situ hybridization for chromosome karyotyping. Nat. Protoc. 1, 1172–1184 (2006).
Snijders, A.M. et al. Assembly of microarrays for genome-wide measurement of DNA copy number. Nat. Genet. 29, 263–264 (2001).
Acknowledgements
This work was supported by the European Commission (DISMAL project, contract no. LSHC-CT-2005-018911 and GENINCA project (2022309)).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Geigl, J., Speicher, M. Single-cell isolation from cell suspensions and whole genome amplification from single cells to provide templates for CGH analysis. Nat Protoc 2, 3173–3184 (2007). https://doi.org/10.1038/nprot.2007.476
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.476
This article is cited by
-
Development of single-cell-level microfluidic technology for long-term growth visualization of living cultures of Mycobacterium smegmatis
Microsystems & Nanoengineering (2021)
-
Atypical fibroxanthoma and pleomorphic dermal sarcoma harbor frequent NOTCH1/2 and FAT1 mutations and similar DNA copy number alteration profiles
Modern Pathology (2018)
-
Direct metabolomics for plant cells by live single-cell mass spectrometry
Nature Protocols (2015)
-
The dynamic range of circulating tumor DNA in metastatic breast cancer
Breast Cancer Research (2014)
-
Are morphological criteria sufficient for the identification of circulating tumor cells in renal cancer?
Journal of Translational Medicine (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.