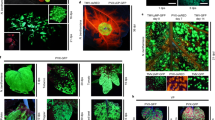
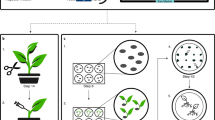
Abstract
Transient gene expression is an indispensable tool for studying functions of gene products. In the case of plants, transient introduction of genes by Agrobacterium infiltration is a method of choice for many species. However, this technique does not work efficiently in Arabidopsis leaf tissue, the most widely used model system for basic plant biology research. Here we present an optimized protocol for biolistic delivery of plasmid DNA into the epidermis of Arabidopsis leaves, which can be easily performed using the Bio-Rad Helios gene gun system. This protocol yields efficient and reproducible transient expression of diverse genes and is exemplified here for use in a functional assay of a transcription repressor and for the subcellular localization and cell-to-cell movement of plant viral movement protein. This protocol is suitable for studies of biological function and subcellular localization of the gene product of interest directly in planta by utilizing different types of activity-based assays. Using this procedure, the data are obtained after 2–4 d of work.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Clough, S.J. Floral dip: Agrobacterium-mediated germ line transformation. Methods Mol. Biol. 286, 91–102 (2005).
Clough, S.J. & Bent, A.F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana . Plant J. 16, 735–743 (1998).
Zhang, X. et al. Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat. Protoc. 1, 641–646 (2006).
Janssen, B.J. & Gardner, R.C. Localized transient expression of GUS in leaf discs following cocultivation with Agrobacterium . Plant Mol. Biol. 14, 61–72 (1990).
Abel, S. & Theologis, A. Transient transformation of Arabidopsis leaf protoplasts: a versatile experimental system to study gene expression. Plant J. 5, 421–427 (1994).
Yoo, S.D., Cho, Y.H. & Sheen, J. Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat. Protoc. 2, 1565–1572 (2007).
Yang, Y., Li, R. & Qi, M. In vivo analysis of plant promoters and transcription factors by agroinfiltration of tobacco leaves. Plant J. 22, 543–551 (2000).
Goodin, M.M. et al. pGD vectors: versatile tools for the expression of green and red fluorescent protein fusions in agroinfiltrated plant leaves. Plant J. 31, 375–383 (2002).
Lee, M.W. & Yang, Y. Transient expression assay by agroinfiltration of leaves. Methods Mol. Biol. 323, 225–229 (2006).
Burch-Smith, T.M., Anderson, J.C., Martin, G.B. & Dinesh-Kumar, S.P. Applications and advantages of virus-induced gene silencing for gene function studies in plants. Plant J. 39, 734–746 (2004).
Burch-Smith, T.M., Schiff, M., Liu, Y. & Dinesh-Kumar, S.P. Efficient virus-induced gene silencing in Arabidopsis . Plant Physiol. 142, 21–27 (2006).
Gelvin, S.B. Agrobacterium transformation of Arabidopsis thaliana roots: a quantitative assay. Methods Mol. Biol. 343, 105–114 (2006).
Levy, M., Rachmilevitch, S. & Abel, S. Transient Agrobacterium-mediated gene expression in the Arabidopsis hydroponics root system for subcellular localization studies. Plant Mol. Biol. Rep. 23, 179–184 (2005).
Wroblewski, T., Tomczak, A. & Michelmore, R. Optimization of Agrobacterium-mediated transient assays of gene expression in lettuce, tomato and Arabidopsis . Plant Biotech. J. 3, 259–273 (2005).
Taylor, N.J. & Fauquet, C.M. Microparticle bombardment as a tool in plant science and agricultural biotechnology. DNA Cell Biol. 21, 963–977 (2002).
Helenius, A. et al. Gene delivery into intact plants using the Helios™ gene gun. Plant Mol. Biol. Rep. 18, 287a–287l (2000).
Wydro, M., Kozubek, E. & Lehmann, P. Optimization of transient Agrobacterium-mediated gene expression system in leaves of Nicotiana benthamiana . Acta Biochim. Pol. 53, 289–298 (2006).
Krichevsky, A. et al. C2H2 zinc finger-SET histone methyltransferase is a plant-specific chromatin modifier. Dev. Biol. 303, 259–269 (2007).
Tzfira, T. et al. pSAT vectors: a modular series of plasmids for fluorescent protein tagging and expression of multiple genes in plants. Plant Mol. Biol. 57, 503–516 (2005).
Goderis, I.J. et al. A set of modular plant transformation vectors allowing flexible insertion of up to six expression units. Plant Mol. Biol. 50, 17–27 (2002).
Vergunst, A.C. et al. VirB/D4-dependent protein translocation from Agrobacterium into plant cells. Science 290, 979–982 (2000).
Lacroix, B., Vaidya, M., Tzfira, T. & Citovsky, V. The VirE3 protein of Agrobacterium mimics a host cell function required for plant genetic transformation. EMBO J. 24, 428–437 (2005).
Rivero-Lepinckas, L., Crist, D. & Scholl, R. Growth of plants and preservation of seeds. Methods Mol. Biol. 323, 3–12 (2006).
Haseloff, J. GFP variants for multispectral imaging of living cells. Methods Cell Biol. 58, 139–151 (1999).
Chen, M.H., Tian, G.W., Gafni, Y. & Citovsky, V. Effects of calreticulin on viral cell-to-cell movement. Plant Physiol. 138, 1866–1876 (2005).
Baluska, F., Samaj, J., Napier, R. & Volkmann, D. Maize calreticulin localizes preferentially to plasmodesmata in root apex. Plant J. 19, 481–488 (1999).
Laporte, C. et al. Involvement of the secretory pathway and the cytoskeleton in intracellular targeting and tubule assembly of Grapevine fanleaf virus movement protein in tobacco BY-2 cells. Plant Cell 15, 2058–2075 (2003).
Oparka, K.J. et al. Gating of epidermal plasmodesmata is restricted to the leading edge of expanding infection sites of tobacco mosaic virus (TMV). Plant J. 12, 781–789 (1997).
Acknowledgements
The work in our laboratory is supported by grants from NIH, NSF, NRI USDA CSREE, BSF and BARD to V.C., and from NIH (1 R01 AI066054-01A) to S.G.L.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ueki, S., Lacroix, B., Krichevsky, A. et al. Functional transient genetic transformation of Arabidopsis leaves by biolistic bombardment. Nat Protoc 4, 71–77 (2009). https://doi.org/10.1038/nprot.2008.217
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.217
This article is cited by
-
Genome-Wide Identification, In Silico Characterization of AtCOP1-Targeting Regulatory Proteins Network and their Expression Profiling in The COP1 Downregulated Arabidopsis thaliana
Journal of Plant Growth Regulation (2023)
-
Transient Gene Expression in Molecular Farming and Functional Genomics of Tea (Camellia sinensis): A Review
Journal of Plant Growth Regulation (2022)
-
An improved and efficient method of Agrobacterium syringe infiltration for transient transformation and its application in the elucidation of gene function in poplar
BMC Plant Biology (2021)
-
An improved biolistic delivery and analysis method for evaluation of DNA and CRISPR-Cas delivery efficacy in plant tissue
Scientific Reports (2021)
-
A modified transient gene expression protocol for subcellular protein localization analysis in rice
Plant Biotechnology Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.